Abstract
Subtle cognitive impairment can occur early in the course of Parkinson’s disease (PD) and may manifest under different forms of executive dysfunction such as impaired cognitive flexibility. The precise contribution of nigrostriatal dopaminergic neurodegeneration to these non-motor features of the disease is poorly known. Whether such cognitive impairment associated with the disease process may also predate and contribute to the development of neuropsychiatric side-effects following dopamine replacement therapy remains largely unknown. To address these issues, we investigated the respective contributions of nigrostriatal degeneration and chronic treatment with the dopamine D3-preferring agonist pramipexole on behavioral flexibility in a rat model of PD. Flexible, intermediate and inflexible rats were identified based on baseline assessment of behavioral flexibility using an operant set-shifting task. Nigrostriatal degeneration was induced by bilateral viral-mediated expression of A53T mutated human α-synuclein in the substantia nigra pars compacta and behavioral flexibility was assessed after induction of nigrostriatal degeneration, and during chronic pramipexole treatment. Nigrostriatal degeneration impaired behavioral flexibility in flexible but not in inflexible rats. Pramipexole induced a decrease of behavioral flexibility that was exacerbated in lesioned rats and in the most flexible individuals. Furthermore, the deficits induced by pramipexole in lesioned rats affected different components of the task between flexible and inflexible individuals. This study demonstrates that nigrostriatal degeneration and pramipexole unequally impair behavioral flexibility, suggesting that the susceptibility to develop non-motor impairments upon treatment initiation could primarily depend on premorbid differences in behavioral flexibility.
Similar content being viewed by others
Introduction
Non-motor features of Parkinson’s disease (PD) and non-motor side effects of dopamine replacement therapy are important factors contributing to the decreased quality of life of patients1,2,3. Within the large spectrum of non-motor symptoms, subtle cognitive impairment can occur early during the course of the disease in one third of patients and may manifest under different aspects of executive dysfunction, such as attentional, memory deficits, as well as decreased cognitive flexibility4,5,6.
Dopamine replacement therapy with dopaminergic D2/D3 agonists is widely used to relieve motor symptoms but can be associated with compulsive behaviors designed as impulse control disorders (ICD) in a significant proportion of patients7,8,9,10. ICD are defined by the inability to refrain the urge to repeatedly engage in behaviors leading to harmful personal, social or financial consequences such as pathological gambling, hypersexuality, binge eating or compulsive shopping10. The fact that ICD affect only a subset of PD patients suggests a significant role for an underlying individual vulnerability as a predisposing factor3.
Executive functions contribute to develop adapted goal-directed behaviors and several clinical studies have highlighted impairments in PD patients with ICD11,12,13. Particularly, working memory, visual-spatial planning, reward processing and cognitive as well as behavioral flexibility (set-shifting, task switching) are impaired11,13,14,15. Cognitive flexibility represents the ability to switch thinking between concepts and behavioral flexibility is defined as the ability to adapt behavior according to environmental changes. As tests assessing cognitive flexibility require behavioral responses, both constructs are intimately associated16. Whether such type of cognitive impairment associated with the disease process may predate and/or contribute to the development of ICD remains largely unknown.
The pathophysiology of executive dysfunction involves dopaminergic and non-dopaminergic (cholinergic, noradrenergic and serotonergic) systems, leading to the dysfunction of frontostriatal circuits17. Interestingly, frontostriatal dysfunction has been identified in PD patients with ICD12,18, suggesting some pathophysiological overlap between non-motor symptoms of PD and non-motor side effects of dopamine replacement therapy. The occurrence of these non-motor features early in the course of the disease further suggest a possible contribution of nigral dopaminergic neurons per se.
Here, we used a rat model of PD induced by viral-mediated expression of α-synuclein in the substantia nigra pars compacta (SNc)19,20 to probe the potential contribution of nigrostriatal degeneration to behavioral flexibility and its possible implications for developing further impairments under dopamine replacement therapy. To this end, we investigated interindividual responses to nigrostriatal degeneration on behavioral flexibility in a set-shifting task, as well as the effects of pramipexole (PPX), one of the most widely used dopaminergic treatment that is also implicated in ICD.
Results
Characterization of the population based on basal inflexibility score
Upon completion of baseline sessions (Fig. 1a, b), the analysis based on the inflexibility score allowed segregating three different groups (Kruskal–Wallis H = 45.50, df = 2, p < 0.0001, Fig. 1c): FLEX, INT and INFLEX (FLEX vs INT: p < 0.01, INT vs INFLEX: p < 0.01, FLEX vs INFLEX p < 0.0001). Baseline error rate was higher for the egocentric dimension than for the visual dimension in all groups (F1, 100 = 47.02, p < 0.0001; FLEX: p < 0.05, INT: p < 0.001, INFLEX: p < 0.001, Fig. 1d) indicating better performances when the active lever was indicated by a visual cue. Moreover, INFLEX animals made more errors than FLEX animals both in the visual and egocentric dimensions (F2, 100 = 14.15, p < 0.0001; visual: p < 0.05, egocentric: p < 0.001, Fig. 1d).
a Experiment timeline. Food restricted animals were trained on the set-shifting task (16 days) in order to screen Flexible (FLEX), Intermediate (INT) and Inflexible (INFLEX) rats. Bilateral lesion of the substantia nigra pars compacta was achieved via viral-mediated bilateral overexpression of α-synuclein (AAV- α-syn). 9 weeks after the surgery, animals were re-tested in the set-shifting task (16 days) to evaluate the effect of nigrostriatal degeneration on behavioral flexibility. Behavioral flexibility was then tested (3 × 4 days) during 21 days of pramipexole (PPX) treatment (0.3 mg/kg/day, i.p). Motor function was assessed with the stepping test at baseline, 4 and 9 weeks after surgery, and during PPX treatment. b Presentation of the set shifting task. In the visual rule, the position of the light cue indicates which lever is rewarded (shown in green). In the egocentric rule, one lever (either left or right) is rewarded and the position of the light cue is random. c Inflexibility scores of FLEX, INT and INFLEX rats at baseline. d Baseline error rates in the visual and egocentric rules for FLEX, INT and INFLEX rats. All data are presented as mean ± SEM. *p < 0.05, **p < 0.01, ***p < 0.001.
Alpha-synuclein-induced nigrostriatal degeneration and motor deficits
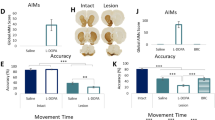
Stereological counts showed that viral-mediated expression of α-synuclein led to a significant loss of TH-positive neurons in the SNc (−42.56%, t = 9.898; df = 51; p < 0.0001 Fig. 2a). The extent of the lesion was independent of baseline flexibility and each group that underwent viral-mediated expression of α-synuclein presented a similar loss of SNc neurons compared to their sham counterparts (Fig. 2b).Viral-mediated expression of α-synuclein in the SNc induced progressive bilateral motor deficits with a significant effect of the lesion (F1, 204 = 316.8, p < 0.0001), time (F3, 204 = 77.04, p < 0.0001) and lesion X time interaction (F3, 204 = 45.23, p < 0.0001, Fig. 2c). Motor deficits were significant at 4 weeks post-AAV (p < 0.0001), worsened at 9 weeks (p < 0.0001) and were improved by PPX (p < 0.0001). The magnitude of stepping deficits was similar between all lesioned rats regardless of the initial inflexibility score (Fig. 2d).
a Stereological counts of tyrosine hydroxylase (TH) positive neurons in the SNc and representative images of the SNc in sham (top) and lesioned rats (bottom). Scale bar = 1 mm. b Magnitude of neurodegeneration in FLEX, INT and INFLEX rats (***p < 0.0001, ****p < 0.0001 vs corresponding sham FLEX, INT and INFLEX rats). c Motor impairments induced by viral mediated expression of alpha-synuclein in the SNc were significant at 4 weeks (****p < 0.0001 vs baseline) and 9 weeks post-AAV injection (****p < 0.0001 vs 4 weeks), and were alleviated by pramipexole (PPX, ****p < 0.0001 vs 9 weeks). d Magnitude of motor impairments at 9 weeks post-AAV injection in FLEX, INT and INFLEX rats (****p < 0.0001 vs corresponding sham FLEX, INT and INFLEX rats). All data are presented as mean ± SEM.
Effects of dopaminergic neurodegeneration and chronic treatment with pramipexole on behavioral flexibility
When considering the entire rat population with no distinction among FLEX, INT and INFLEX groups, there was a significant effect of the lesion (F1, 153 = 48.88, p < 0.0001), time (F2, 153 = 60.75, p < 0.0001) and lesion X time interaction (F2, 153 = 12.75, p < 0.0001, Fig. 3a). Viral-mediated expression of α-synuclein had no significant effect on inflexibility score when compared with baseline performances, but lesioned rats displayed worse performances than their sham counterparts after AAV surgery (p < 0.001). Chronic treatment with PPX induced a significant worsening of behavioral flexibility in both sham and lesioned animals compared with post-AAV (p < 0.0001, Fig. 3a). However, this deleterious effect of PPX was more pronounced in lesioned than in sham animals (p < 0.0001, Fig. 3a).
a Inflexibility score of whole sham and lesioned populations at Baseline, post-AAV injection and during the last four days of chronic PPX treatment (PPX). b Color plot illustrating the evolution of the inflexibility score for sham (GFP) and lesioned (α-syn) groups throughout the course of the experiment, each colored rectangle representing a single animal. Flexibility performances are represented at Baseline, post-AAV injection and during the last four days of chronic PPX treatment (PPX): dark blue indicates a low inflexibility score and red a high inflexibility score. Inflexibility scores for sham vs lesioned group at Baseline, post-AAV injection and during the last four days of chronic PPX treatment (PPX) for FLEX (c), INT (d) and INFLEX (e) groups. All data are presented as mean ± SEM.
When considering initial inflexibility score, this parameter was differentially affected both by the nigrostriatal lesion and by PPX (Fig. 3b, individual animal’s inflexibility scores are represented by color-coded rectangles aligned vertically for each experimental time point). Three-way ANOVA indicated a significant effect of the lesion (F1, 141 = 59.87, p < 0.0001), flexibility (F2, 141 = 13.48, p < 0.0001) time (F2, 141 = 68.67, p < 0.0001), lesion X time interaction (F2, 141 = 15.61, p < 0.0001) and flexibility X time interaction (F4, 141 = 4.18, p < 0.01).
Further analysis within each flexibility group revealed a significant effect of the lesion (F1, 48 = 18.50, p < 0.0001), time (F2, 48 = 32.68, p < 0.0001) and lesion X time interaction (F2, 48 = 6.01, p < 0.001, Fig. 3c) in FLEX animals. Nigrostriatal lesion induced a significant deterioration of set-shifting compared with baseline (p < 0.05). PPX treatment worsened the inflexibility score both in sham (p < 0.01) and lesioned FLEX rats (p < 0.05), and this deleterious effect was more pronounced in lesioned than in sham animals (p < 0.01). In INT rats, there was a significant effect of the lesion (F1, 45 = 38.08, p < 0.0001), time (F2, 45 = 60.50, p < 0.0001) and lesion X time interaction (F2, 45 = 8.43, p < 0.001, Fig. 3d). Whereas baseline performances were not affected by the dopaminergic denervation, PPX induced an increase of the inflexibility score in both sham and lesioned rats (p < 0.0001), and this deleterious effect was higher in lesioned than in sham rats (p < 0.0001). In INFLEX rats, there was a significant effect of the lesion (F1, 48 = 15.32, p < 0.001), time (F2, 48 = 13.45, p < 0.0001) and lesion X time interaction (F2, 48 = 4.68, p < 0.05, Fig. 3e). Post hoc analysis indicated no difference between baseline and post-AAV in sham or lesioned rats, no differences between sham and lesioned rats at post-AAV, and only lesioned rats were affected by chronic PPX treatment (p < 0.001 vs post-AAV, baseline and vs sham PPX, Fig. 3e).
These results indicate that α-synuclein induced nigral degeneration impairs behavioral flexibility in FLEX animals. In addition, the lesion potentiated the deleterious effect of PPX on behavioral flexibility in all animals. Moreover, the magnitude of the deleterious effect of PPX on flexibility negatively correlated with the inflexibility score before PPX treatment both in sham (r = −0.5841, p < 0.001, Fig. 4a) and lesioned animals (r = −0.4777, p < 0.05, Fig. 4b), indicating that in both groups the most flexible animals were the most impacted by PPX.
Effects of dopaminergic neurodegeneration and PPX on each dimension of the set-shifting task
As the set-shifting task involves rules based on two distinct perceptual dimensions (visual and egocentric), we assessed how each dimension was affected by the lesion and by PPX. The visual error rate was not impaired by the dopaminergic lesion or by PPX in FLEX and INT rats (Fig. 5a, b). However, in INFLEX rats, there was a significant effect of the lesion (F1, 48 = 5.25, p < 0.05) as well as lesion X time interaction (F2, 48 = 7.18, p < 0.01), and PPX increased the visual error rate in INFLEX lesioned animals compared to their sham counterparts (p < 0.001, Fig. 5c). Regarding the egocentric dimension, there was a significant effect of the lesion (F1, 48 = 9.64, p < 0.01) and time (F2, 48 = 40.83, p < 0.0001) in FLEX animals that displayed a significant deficit after PPX compared to post-AAV (sham: p < 0.001, lesioned: p < 0.0001, Fig. 5d). In addition, this deficit induced by PPX was greater in lesioned than in sham rats (p < 0.01 Fig. 5d). In INT rats, there was a significant effect of the lesion (F1, 45 = 4.83, p < 0.05), time (F2, 45 = 19.46, p < 0.0001) and lesion X time interaction (F2, 45 = 3.29, p < 0.05). The error rates of INT sham and lesioned animals during the egocentric rule were also significantly higher after PPX treatment compared with post-AAV (p < 0.01, Fig. 5e). Conversely, the performances of INFLEX sham and lesioned animals in the egocentric dimension were unaffected by the lesion or by PPX (Fig. 5f).
Error rate in the visual rule at Baseline, after AAV injection (post-AAV), and during the last four days of chronic PPX treatment (PPX) for sham and lesioned animals for FLEX (a), INT (b) and INFLEX (c) groups. Error rate in egocentric rule at Baseline, after AAV injection (post-AAV), and during the last four days of chronic PPX treatment (PPX) for sham and lesioned animals for FLEX (d), INT (e) and INFLEX (f) groups. Number of completed rules at Baseline, after AAV injection (post-AAV), and during the last four days of PPX treatment (PPX) for sham and lesioned animals for FLEX (g), INT (h) and INFLEX (i) groups. All data are presented as mean ± SEM. *p < 0.05; **p < 0.01; ***p < 0.001; ****p < 0.0001.
We also assessed the effect of the dopaminergic lesion and PPX on the number of completed rules during the task. In the FLEX group, there was a significant effect of the lesion (F1, 48 = 12.59, p < 0.001), time (F2, 48 = 12.61, p < 0.001) and lesion X time interaction (F2, 48 = 4.16, p < 0.05). FLEX lesioned rats displayed a smaller number of completed rules at post-AAV compared with their sham counterparts (p < 0.05, Fig. 5g) that further decreased after PPX (p < 0.05). A similar effect was observed in INT rats (lesion: F1, 45 = 25.8, p < 0.0001, time: F2, 45 = 0.0001 and lesion X time: F2, 45 = 9.6, p < 0.001), with lesioned rats displaying less completed rules post-AAV compared with shams (p < 0.01, Fig. 5h). The number of completed rules further decreased after PPX (sham: p < 0.05, lesioned: p < 0.01, lesioned vs sham: p < 0.0001, Fig. 5h). In INFLEX rats, the main effect of the lesion (F1, 48 = 16.12, p < 0.001) and time (F2, 48 = 5.68, p < 0.01) were evidenced only as a decreased number of completed rules after PPX in lesioned rats (p < 0.01, Fig. 5i).
Discussion
Cognitive impairment is a frequent non-motor feature of PD affecting up to 30% of newly diagnosed PD patients21, and can manifest under different forms including executive dysfunction such as cognitive and behavioral flexibility deficits2. Although the pathophysiology of these non-motor symptoms is still incompletely understood, executive dysfunction is at least partially associated with a dopamine-dependent dysfunction of fronto-striatal circuits2. Interestingly, altered activity of fronto-striatal circuits, decreased cognitive flexibility and set-shifting have also been reported in PD patients with ICD12,13,21,22, suggesting that interindividual differences in executive function prior to treatment initiation could contribute to the increased vulnerability of some patients to develop ICD.
This study aimed at investigating the contribution of nigrostriatal degeneration and dopamine replacement therapy with PPX on interindividual differences in behavioral flexibility. Using a validated set-shifting task23,24 performed at the premorbid stage, after nigrostriatal degeneration and following chronic treatment with PPX in a rat model of PD, we demonstrate that nigrostriatal dopaminergic loss unequally affects behavioral flexibility depending on baseline performances. We further show that the deleterious effects of PPX on behavioral flexibility are increased in rats with nigrostriatal degeneration. It should be acknowledged that this work included only male rats and that future studies are needed to determine if these findings also extend to females.
Our results provide experimental evidence that nigrostriatal dopaminergic neurons contribute to set-shifting abilities and they demonstrate that nigrostriatal degeneration has differential implications for set-shifting abilities depending on baseline performances. Indeed, a deleterious effect of nigral neurodegeneration was only observed in FLEX rats, whereas the performances of INT and INFLEX animals were unaffected. Stereological counts of nigral neurons demonstrated a similar level of denervation between FLEX, INT and INFLEX rats, indicating that this effect could not be attributed to a different magnitude of the lesion. These results are in agreement with clinical studies showing that PD patients display cognitive flexibility deficits as shown by an impaired ability to ignore irrelevant/distracting information, increased perseveration and learned irrelevance5,25. Even though the precise mechanisms underlying this involvement of the nigrostriatal pathway on set-shifting remain to be elucidated, there is evidence pointing toward a role for interindividual differences in dopamine homeostasis, as suggested by studies showing associations between polymorphisms in the catechol-O-methyltransferase gene and behavioral flexibility, both in the general population26 and in PD patients27. An association between polymorphisms in the dopamine transporter gene, fronto-striatal activation and set-shifting performances in PD patients further supports a role for interindividual differences in dopamine homeostasis as a substrate underlying behavioral flexibility28.
Frontostriatal loops are implicated in behavioral flexibility and can be modulated by dopaminergic pathways at multiple levels. This modulation has been mostly studied in the prefrontal cortex and ventral striatum in rodents, and local infusion of dopamine agonists or antagonists can alter different aspects of behavioral flexibility. In the prefrontal cortex, local infusion of D1 or D2 antagonists impair set-shifting whereas D1 or D2 agonists have no effect29. In the nucleus accumbens, blockade of D1 or increased stimulation of D2 receptors both impair set-shifting, suggesting that a critical balance between activation of D1 and D2 receptors is required for optimal performances30. Here, we provide evidence for a contribution of D3 receptor activation by demonstrating that systemic administration of PPX decreases behavioral flexibility. However, at the dose used in this preclinical study or in PD patients, PPX will also not only act on the D3 but also on D2 receptors and the combined stimulation of both receptors could contribute to an imbalance in dopamine receptor subtype activation promoting impaired behavioral flexibility as previously shown for D1 and D2 receptors30. Importantly, our results indicate that nigrostriatal degeneration acts as an aggravating factor since this deleterious effect of PPX was markedly exacerbated in lesioned rats. In non-lesioned rats, PPX was found to differentially undermine set-shifting abilities depending on basal performances, with FLEX and INT animals being impaired whereas INFLEX animals were unaffected. Conversely, all lesioned rats were impaired by PPX regardless of their basal flexibility status. Regardless of the lesion, performances in the visual and egocentric dimensions of the task were differentially affected by PPX depending on the baseline flexibility status. Indeed, the ability to follow a relevant visual cue was preserved in both sham and lesioned FLEX and INT rats, while their capacity to rely on the egocentric dimension was undermined. Conversely, in INFLEX lesioned rats, the ability to follow the egocentric strategy was preserved but this was at the expense of following the visual cue. This suggest that visual and egocentric strategies rely on distinct substrates that are differentially impacted by PPX depending on baseline flexibility trait.
This deleterious effect of a D3 preferring agonist on set shifting abilities in rats with nigrostriatal degeneration is in agreement with previous work showing that a D3 antagonist was able to improve performances in an attentional set-shifting task in parkinsonian monkeys31. Our results showing a pro-inflexible effect of PPX are also in line with previous findings showing that D3 knock-out mice display better performances in a set-shifting task32.
Even though executive impairment is common in PD, with one third of patients displaying some degree of executive dysfunction at the time of diagnosis33, there is a noticeable clinical heterogeneity that may reflect the contribution of genetic factors (as discussed above), specific patterns of neurodegeneration and disease progression, as well as premorbid interindividual differences in cognitive performances6. In this regard, our preclinical results help understanding such clinical heterogeneity by demonstrating that nigrostriatal degeneration leads to contrasting outcomes on behavioral flexibility depending on premorbid performance levels. Therefore, individuals who were the most flexible prior to nigral neurodegeneration experienced a drastic alteration of their behavioral flexibility, whereas poorly flexible individuals were much less affected. These findings also have implications for the pathophysiology of ICD in PD since clinical studies have reported impairment in cognitive flexibility and set-shifting in ICD13,15. Indeed, our preclinical results indicate that impairments in behavioral flexibility are triggered by PPX in all subjects but also by the neurodegenerative process in some individuals. Accordingly, longitudinal assessment of cognitive flexibility in PD patients may help understanding how disease-induced and drug-induced impairments may interact to promote the emergence of ICD in some patients.
By demonstrating that a loss of nigrostriatal neurons leading to homogenous motor impairments in a rat model of PD unequally impairs behavioral flexibility depending on premorbid performances, this study highlights non-motor outcomes of nigrostriatal neurodegeneration and helps understanding the bases of inter-individual variability of cognitive impairments in PD. Our results with PPX further highlight that the deleterious effect of this drug is not only exacerbated by the loss of nigrostriatal neurons but involves impairments in different dimensions of the task between flexible and inflexible individuals, suggesting that behavioral processes subserving flexibility are differentially altered by the drug depending on individual baseline performances. Taken together, our results suggest that the vulnerability to develop behavioral flexibility impairments upon nigrostriatal neurodegeneration and treatment initiation could be linked to pre-morbid interindividual differences.
Methods
Subjects
All experiments were approved by the local ethical committee (Comethea Poitou-Charentes C2EA-84, study approval #11562-2017092810291542) and performed under the European Directive (2010/63/EU) on the protection of animals used for scientific purposes. Male Sprague Dawley rats (n = 52, 175 g, Janvier, France) were housed on a reversed 12 h cycle. After seven days of habituation, they were food restricted to 15 g per day one week before behavioral studies. Water was available ad libitum.
Flexibility procedure
This procedure was performed in operant chambers (Med Associates, Fairfax, VT, USA) equipped with two identical levers, each surmounted by a light cue. Before assessment of set-shifting abilities, five consecutive 1-h training sessions were performed to train the rats to press the levers and to collect the reward in the magazine. Behavioral flexibility was then assessed before surgery (baseline, 16 sessions), 9 weeks after nigrostriatal degeneration (post-AAV surgery, 16 sessions) and 12 sessions during PPX treatment (days 1–4, days 10–13 and days 18–21), with a set-shifting task requiring discriminating between two rules based on two distinct perceptual dimensions: visual and egocentric (Fig. 1a)23,24,34. Experiments were performed daily during 45 min sessions. During the visual rule, the active lever was determined by the position of the light cue above it. During the egocentric rule, only one lever (right or left) was active, independently of the position of the light cue (Fig. 1b). For each rule, the position of the light cue was randomly determined and could occur above the same lever only for two consecutive trials. The visual and egocentric dimensions alternated during test sessions and were presented in a pseudo random order and rats were exposed to all four possible combinations in each test session (an example of the schedule of a test session is shown on supplementary Fig. 1).
A good lever choice led to obtain a 45 mg sucrose pellet reward (TestDiet, Saint-Louis, MO, USA). Otherwise, a sound was triggered, and the house-light was turned on for twenty seconds (time-out). Rules alternated after ten consecutive correct responses. Each dimension (visual and egocentric) was presented at most twice per session. Each session ended after all four rules were successfully completed or after 45 min if the four rules were not completed. For each session, the percentage of incorrect responses (error percentage) and the number of completed rules were quantified. An inflexibility score was determined for each rat as the mean error percentage of the last four test sessions minus the sum of completed rules over the last four sessions. Rats were clustered in tertiles, based on their baseline inflexibility score: Flexible (FLEX, n = 17), Intermediate (INT, n = 17) and Inflexible (INFLEX, n = 18).
AAV- α-synuclein-mediated lesion
Upon completion of baseline testing, FLEX, INT and INFLEX rats were randomly assigned to the sham or lesioned group. Rats were anesthetized with isoflurane (2% at 1.5 l/min) and placed in a stereotaxic frame (Kopf Instruments®, Tujunga, CA, USA). Vitamin A was applied on the eyes to prevent corneal dryness and 1 mL of saline solution was subcutaneous injected to prevent dehydration. Pre-surgical analgesia was performed with ketoprofen (10 mg/kg, i.p.) and local analgesia with 2% xylocaine gel after disinfection with vetedine®. Two bilateral injections were performed in the SNc (in millimeters from bregma and dura and according to the Paxinos and Watson rat brain atlas, AP: −5.1, ML: +/−2.2, DV: −8 and AP: −5.6, ML: +/−2; DV: −8) with 1 µL of AAV2 expressing human A53T α-synuclein (CMVie/SynP-synA53T-WPRE, 5.2 1013 gcp/mL) or AAV2 expressing GFP (green fluorescent protein, CMVie/SynP-GFPdegron-WPRE, 3.7 1013 gcp/mL) at 0.2 µL/min as previously described19,35. Post-surgical analgesia was performed with ketoprofen (10 mg/kg/day, i.p.) the day after surgery. Akinesia was estimated twice a day during four consecutive days using the stepping test as previously described36 at baseline, 4 and 9 weeks after surgery, and following chronic PPX treatment.
Treatment
After completion of the post-AAV surgery assessment of behavioral flexibility, all animals received PPX (Sequoia Research Products, Berkshire, UK) prepared daily in sterile saline and administrated subcutaneously at a dose of 0,3 mg/kg/day for 21 days. Behavioral flexibility was tested 45 min after PPX injection to avoid interference with the early decreased locomotion occurring during the first 30 min after injection37.
Tissue processing and histopathological analysis
Brain collection was performed after the last flexibility session. Sodium pentobarbital (120 mg/kg, i.p.) was injected prior to an intracardiac perfusion with 200 mL 0,9% NaCl followed by 200 mL ice-cold 4% paraformaldehyde. Overnight post-fixation in the same fixative and cryoprotection in H2O/20% sucrose at 4 °C were realized. Brains were frozen in isopentane at −40 °C and stored at −80 °C. Serial 50 µm coronal free-floating sections were collected and stored at −20 °C in a cryoprotectant solution. After 3 washes in Tris Buffer Saline 1X (TBS, 15 min each), sections were incubated 10 min in a peroxidase blocking solution (S2023, Agilent Technologies, Santa Clara, CA, USA). After 3 washes, an incubation of 90 min in a blocking solution containing 3% BSA (bovin serum albumin) and 0.3% Triton in TBS 1X was performed. Sections were then incubated 18 h at 4 °C with mouse anti-TH (1/5000, MAB318, Sigma-Aldrich, Saint Louis, MO, USA) in blocking solution. Sections were washed 3 times in TBS 1X, and incubated 1 h at room temperature (RT) with EnVision HRP system anti-mouse (K400111-2, Agilent Technologies, Santa Clara, CA, USA) in blocking solution. Then, sections were washed three times in TBS 1X and immunoreactions were revealed with DAB peroxidase substrate (K346811-2, Agilent Technologies, Santa Clara, CA, USA). Finally, sections were mounted on gelatin-coated slides and coverslipped with DePeX.
TH positive neurons were counted using the optical fractionator method on every 6th section of the SNc as previously described35. Systematic random sampling was performed with the Mercator Pro V6.5 (Explora Nova, La Rochelle, France) software coupled with a Leica 5500B microscope. Following delineation of the SNc with x5 objective, counting was done with x40 objective.
Statistical analysis
Data are expressed as mean ± Standard Error of the Mean (SEM) and analyzed using GraphPad Prism 9.3 and R. Normality was tested with the Kolmogorov–Smirnov test. Data having a Gaussian distribution were analyzed using two-way analysis of variance (ANOVA) followed by Tukey’s post-hoc multiple comparisons test, except when comparing only two groups where a Student’s t test was performed. Kruskal–Wallis test followed by Dunn’s post-hoc test, or Mann–Whitney test were applied when data did not follow a normal distribution. The following variables were analyzed: lesion (sham vs lesioned), time (baseline, post-AAV, PPX) and flexibility status (FLEX, INT, INFLEX). Correlations between inflexibility scores were performed using Pearson correlation. For all analyzes, a p value < 0.05 was considered significant.
Reporting summary
Further information on research design is available in the Nature Research Reporting Summary linked to this article.
Data availability
The data that support the findings of this study are available from the corresponding author upon request.
References
Chaudhuri, K. R., Healy, D. G. & Schapira, A. H. National Institute for Clinical, E. Non-motor symptoms of Parkinson’s disease: diagnosis and management. Lancet Neurol. 5, 235–245 (2006).
Schapira, A. H. V., Chaudhuri, K. R. & Jenner, P. Non-motor features of Parkinson disease. Nat. Rev. Neurosci. 18, 435–450 (2017).
Voon, V. et al. Impulse control disorders and levodopa-induced dyskinesias in Parkinson’s disease: an update. Lancet Neurol. 16, 238–250 (2017).
Barone, P. et al. The PRIAMO study: a multicenter assessment of nonmotor symptoms and their impact on quality of life in Parkinson’s disease. Mov. Disord. 24, 1641–1649 (2009).
Cools, R., Barker, R. A., Sahakian, B. J. & Robbins, T. W. Mechanisms of cognitive set flexibility in Parkinson’s disease. Brain J. Neurol. 124, 2503–2512 (2001).
Kehagia, A. A., Barker, R. A. & Robbins, T. W. Neuropsychological and clinical heterogeneity of cognitive impairment and dementia in patients with Parkinson’s disease. Lancet Neurol. 9, 1200–1213 (2010).
Antonini, A. et al. ICARUS study: prevalence and clinical features of impulse control disorders in Parkinson’s disease. J. Neurol. Neurosurg. Psychiatry 88, 317–324 (2017).
Martini, A., Dal Lago, D., Edelstyn, N. M. J., Grange, J. A. & Tamburin, S. Impulse control disorder in Parkinson’s disease: a meta-analysis of cognitive, affective, and motivational correlates. Front. Neurol. 9, https://doi.org/10.3389/fneur.2018.00654 (2018).
Voon, V. et al. Chronic dopaminergic stimulation in Parkinson’s disease: from dyskinesias to impulse control disorders. Lancet. Neurol. 8, 1140–1149 (2009).
Weintraub, D. et al. Impulse control disorders in Parkinson disease: a cross-sectional study of 3090 patients. Arch. Neurol. 67, https://doi.org/10.1001/archneurol.2010.65 (2010).
Djamshidian, A. et al. Risk and learning in impulsive and nonimpulsive patients with Parkinson’s disease. Mov. Disord. 25, 2203–2210 (2010).
Santangelo, G. et al. Cognitive dysfunctions and pathological gambling in patients with Parkinson’s disease. Mov. Disord. 24, 899–905 (2009).
Vitale, C. et al. Comparative neuropsychological profile of pathological gambling, hypersexuality, and compulsive eating in Parkinson’s disease. Mov. Disord. 26, 830–836 (2011).
Cools, R., Barker, R. A., Sahakian, B. J. & Robbins, T. W. l-Dopa medication remediates cognitive inflexibility, but increases impulsivity in patients with Parkinson’s disease. Neuropsychologia 41, 1431–1441 (2003).
Santangelo, G., Raimo, S. & Barone, P. The relationship between Impulse Control Disorders and cognitive dysfunctions in Parkinson’s Disease: a meta-analysis. Neurosci. Biobehav. Rev. 77, 129–147 (2017).
Uddin, L. Q. Cognitive and behavioural flexibility: neural mechanisms and clinical considerations. Nat. Rev. Neurosci. 22, 167–179 (2021).
Jellinger, K. A. In International Review of Neurobiology Vol. 133, 13–62 (Elsevier, 2017).
Cilia, R. et al. Pathological gambling in patients with Parkinson’s disease is associated with fronto-striatal disconnection: a path modeling analysis. Mov. Disord. 26, 225–233 (2011).
Bourdenx, M. et al. Lack of additive role of ageing in nigrostriatal neurodegeneration triggered by α-synuclein overexpression. Acta Neuropathol. Commun. 3, https://doi.org/10.1186/s40478-015-0222-2 (2015).
Dehay, B. & Fernagut, P.-O. Alpha-synuclein-based models of Parkinson’s disease. Revue Neurol. 172, 371–378 (2016).
Santangelo, G. et al. Mild Cognitive Impairment in newly diagnosed Parkinson’s disease: a longitudinal prospective study. Parkinsonism Rel. Disord. 21, 1219–1226 (2015).
Carriere, N., Lopes, R., Defebvre, L., Delmaire, C. & Dujardin, K. Impaired corticostriatal connectivity in impulse control disorders in Parkinson disease. Neurology 84, 2116–2123 (2015).
Darrah, J. M., Stefani, M. R. & Moghaddam, B. Interaction of N-methyl-D-aspartate and group 5 metabotropic glutamate receptors on behavioral flexibility using a novel operant set-shift paradigm. Behav. Pharmacol. 19, 225–234 (2008).
Istin, M., Thiriet, N. & Solinas, M. Behavioral flexibility predicts increased ability to resist excessive methamphetamine self-administration: Flexibility and METH addiction. Addict. Biol. 22, 958–966 (2017).
Fallon, S. J., Hampshire, A., Barker, R. A. & Owen, A. M. Learning to be inflexible: enhanced attentional biases in Parkinson’s disease. Cortex 82, 24–34 (2016).
Schulz, S. et al. When control fails: influence of the prefrontal but not striatal dopaminergic system on behavioural flexibility in a change detection task. Neuropharmacology 62, 1028–1033 (2012).
Fallon, S. J. et al. Differential optimal dopamine levels for set-shifting and working memory in Parkinson’s disease. Neuropsychologia 77, 42–51 (2015).
Habak, C. et al. Dopamine transporter SLC6A3 genotype affects cortico-striatal activity of set-shifts in Parkinson’s disease. Brain 137, 3025–3035 (2014).
Floresco, S. B., Magyar, O., Ghods-Sharifi, S., Vexelman, C. & Tse, M. T. Multiple dopamine receptor subtypes in the medial prefrontal cortex of the rat regulate set-shifting. Neuropsychopharmacology 31, 297–309 (2006).
Haluk, D. M. & Floresco, S. B. Ventral striatal dopamine modulation of different forms of behavioral flexibility. Neuropsychopharmacology 34, 2041–2052 (2009).
Millan, M. J. et al. The dopamine D3 receptor antagonist, S33138, counters cognitive impairment in a range of rodent and primate procedures. Int. J. Neuropsychopharmacol. 13, 1035–1051 (2010).
Glickstein, S. B., Desteno, D. A., Hof, P. R. & Schmauss, C. Mice lacking dopamine D2 and D3 receptors exhibit differential activation of prefrontal cortical neurons during tasks requiring attention. Cereb Cortex 15, 1016–1024 (2005).
Decourt, M., Jimenez-Urbieta, H., Benoit-Marand, M. & Fernagut, P. O. Neuropsychiatric and cognitive deficits in Parkinson’s disease and their modeling in rodents. Biomedicines 9 https://doi.org/10.3390/biomedicines9060684 (2021).
Brady, A. M. & Floresco, S. B. Operant procedures for assessing behavioral flexibility in rats. J. Vis. Exp., https://doi.org/10.3791/52387 (2015).
Engeln, M. et al. Multi-facetted impulsivity following nigral degeneration and dopamine replacement therapy. Neuropharmacology 109, 69–77 (2016).
Olsson, M., Nikkhah, G., Bentlage, C. & Björklund, A. Forelimb akinesia in the rat Parkinson model: differential effects of dopamine agonists and nigral transplants as assessed by a new stepping test. J. Neurosci. 15, 3863–3875 (1995).
Chang, W. L., Breier, M. R., Yang, A. & Swerdlow, N. R. Disparate effects of pramipexole on locomotor activity and sensorimotor gating in Sprague-Dawley rats. Pharmacol. Biochem. Behav. 99, 634–638 (2011).
Acknowledgements
The Université de Poitiers and the Institut National de la Santé et de la Recherche Médicale provided infrastructural support. This work was supported by Fondation de France (grant #00086205), région Nouvelle Aquitaine CPER 2015–2020 and FEDER 2014–2020 programs. The authors thank Jean-Emmanuel Longueville for his help with R and Dr Erwan Bezard for useful comments and suggestions on the manuscript. This study has benefited from the facilities and expertise of PREBIOS core facility at the University of Poitiers. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Author information
Authors and Affiliations
Contributions
conceptualization: M.B.M., M.S., P.O.F. Data acquisition: M.D., E.B., M.F., M.B.M., P.O.F. Statistical analysis: M.D., M.B.M., P.O.F. Writing of the first draft: M.D., M.B.M., P.O.F. Manuscript review and editing: M.D., E.B., M.F., M.S., M.B.M., P.O.F. M.B.M. and P.O.F. contributed equally as last authors.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Decourt, M., Balado, E., Francheteau, M. et al. Premorbid performances determine the deleterious effects of nigrostriatal degeneration and pramipexole on behavioural flexibility. npj Parkinsons Dis. 9, 31 (2023). https://doi.org/10.1038/s41531-023-00475-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41531-023-00475-3