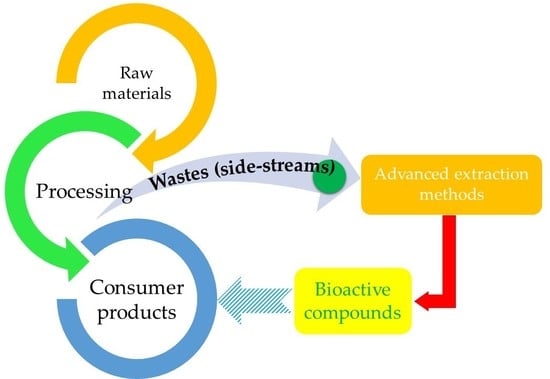
Recovery of Natural Antioxidants from Agro-Industrial Side Streams through Advanced Extraction Techniques
Abstract
:1. Introduction
2. Recovery of Antioxidants from Agro-Industrial Side Streams
2.1. Recent Advances in Recovery of Antioxidants from Agro-Industrial Side Streams
2.2. Recovery of Antioxidant Compounds from Edible Oil Industry Wastes
2.3. Recovery of Antioxidant Compounds from Fruits and Vegetable Wastes
2.3.1. Fruits Wastes
2.3.2. Vegetable Wastes
2.4. Recovery of Antioxidants Compounds from other Different Industries
3. Potential Applications of Antioxidants Recovered from Food Waste and by-Products
4. Conclusions and Perspectives
Author Contributions
Funding
Conflicts of Interest
References
- Baiano, A. Recovery of biomolecules from food wastes—A review. Molecules 2014, 19, 14821–14842. [Google Scholar] [CrossRef] [PubMed]
- Michal, J.; Andrea, Š.; Anna, M.; Jozef, Š. Extraction of value-added components from food industry based and agro-forest biowastes by deep eutectic solvents. J. Biotechnol. 2018, 282, 46–66. [Google Scholar]
- Sladkova, A.; Benedekova, M.; Stopka, J.; Surina, I.; Haz, A.; Strizincová, P.; Cizova, K.; Skulcova, A.; Burcova, Z.; Kreps, F.; et al. Yield of polyphenolic substances extracted from spruce (Picea abies) bark by microwave-assisted extraction. BioResources 2016, 11, 9912–9921. [Google Scholar] [CrossRef]
- Kinsella, Z.; Stewart, V.E.H.; Trombly, J. The Environmental Paper Listening Study. Chapter Four: Tree Free Paper. Available online: http://www.conservatree.org/paperlisteningstudy/TreeFreePaperLS.pdf (accessed on 2 September 2019).
- Reddy, N.; Yang, Y. Biofibers from agricultural byproducts for industrial applications. Trends Biotechnol. 2005, 23, 22–27. [Google Scholar] [CrossRef] [PubMed]
- Nunes, M.A.; Pimentel, F.B.; Costa, A.S.; Alves, R.C.; Oliveira, M.B.P. Olive by-products for functional and food applications: Challenging opportunities to face environmental constraints. Innov. Food Sci. Emerg. Technol. 2016, 35, 139–148. [Google Scholar] [CrossRef]
- Ferrentino, G.; Asaduzzaman, M.; Scampicchio, M.M. Current technologies and new insights for the recovery of high valuable compounds from fruits by-products. Crit. Rev. Food Sci. Nutr. 2018, 58, 386–404. [Google Scholar] [CrossRef] [PubMed]
- Sindhu, R.; Gnansounou, E.; Rebello, S.; Binod, P.; Varjani, S.; Thakur, I.S.; Nair, R.B.; Pandey, A. Conversion of food and kitchen waste to value-added products. J. Environ. Manag. 2019, 241, 619–630. [Google Scholar] [CrossRef] [PubMed]
- Nasini, L.; De Luca, G.; Ricci, A.; Ortolani, F.; Caselli, A.; Massaccesi, L.; Regni, L.; Gigliotti, G.; Proietti, P. Gas emissions during olive mill waste composting under static pile conditions. Int. Biodeterior. Biodegrad. 2016, 107, 70–76. [Google Scholar] [CrossRef]
- Sharholy, M.; Ahmad, K.; Mahmood, G.; Trivedi, R.C. Municipal solid waste management in Indian cities—A review. Waste Manag. 2008, 28, 459–467. [Google Scholar] [CrossRef]
- Bunaciu, A.A.; Danet, A.F.; Fleschin, S.; Aboul- Enein, H.Y. Recent applications for in vitro antioxidant activity assay. Crit. Rev. Anal. Chem. 2016, 46, 389–399. [Google Scholar] [CrossRef]
- Gunes, R.; Palabiyik, I.; Toker, O.S.; Konar, N.; Kurultay, S. Incorporation of defatted apple seeds in chewing gum system and phloridzin dissolution kinetics. J. Food Eng. 2019, 255, 9–14. [Google Scholar] [CrossRef]
- Tremocoldi, M.A.; Rosalen, P.L.; Franchin, M.; Massarioli, A.P.; Denny, C.; Daiuto, E.R.; Paschoal, J.A.R.; Melo, P.S.; de Alencaret, S.M. Exploration of avocado by-products as natural sources of bioactive compounds. PLoS ONE 2018, 13, e0192577. [Google Scholar] [CrossRef] [PubMed]
- Zago, E.; Lecomte, J.; Barouh, N.; Aouf, C.; Carre, P.; Fine, F.; Villeneuve, P. Influence of rapeseed meal treatments on its total phenolic content and composition in sinapine, sinapic acid and canolol. Ind. Crops Prod. 2015, 76, 1061–1070. [Google Scholar] [CrossRef]
- Li, B.B.; Smith, B.; Hossain, M.M. Extraction of phenolics from citrus peels II: Enzyme-assisted extraction method. Sep. Purif. Technol. 2006, 48, 189–196. [Google Scholar] [CrossRef]
- Rodrigues, S.; Pinto, G.A.S.; Fernandes, F.A.N. Optimization of ultrasound extraction of phenolic compounds from coconut (Cocos nucifera) shell powder by response surface methodology. Ultrason. Sonochem. 2008, 15, 95–100. [Google Scholar] [CrossRef]
- Castrica, M.; Rebucci, R.; Giromini, C.; Tretola, M.; Cattaneo, D.; Baldi, A. Total phenolic content and antioxidant capacity of agri-food waste and by-products. Italian J. Anim. Sci. 2018, 18, 336–341. [Google Scholar] [CrossRef]
- Puganen, A.; Kallio, H.P.; Schaich, K.M.; Suomela, J.P.; Yang, B. Red/green currant and sea buckthorn berry press residues as potential sources of antioxidants for food use. J. Agric. Food Chem. 2018, 66, 3426–3434. [Google Scholar] [CrossRef]
- Liu, Q.; Tang, G.Y.; Zhao, C.N.; Feng, X.L.; Xu, X.Y.; Cao, S.Y.; Meng, X.; Li, S.; Gan, R.Y.; Li, H.B. Comparison of antioxidant activities of different grape varieties. Molecules 2018, 23, 2432. [Google Scholar] [CrossRef]
- Erşan, S.; Güçlü Üstündağ, Ö.; Carle, R.; Schweiggert, R.M. Subcritical water extraction of phenolic and antioxidant constituents from pistachio (Pistacia vera L.) hulls. Food Chem. 2018, 253, 46–54. [Google Scholar] [CrossRef]
- Dos Reis, L.C.R.; Facco, E.M.P.; Flores, S.H.; Rios, A.D.O. Stability of functional compounds and antioxidant activity of fresh and pasteurized orange passion fruit (Passiflora caerulea) during cold storage. Food Res. Int. 2018, 106, 481–486. [Google Scholar] [CrossRef]
- Stoll, T.; Schweiggert, U.; Schieber, A.; Carle, R. Process for the recovery of a carotene-rich functional food ingredient from carrot pomace by enzymatic liquefaction. Innov. Food Sci. Emerg. Technol. 2003, 4, 415–423. [Google Scholar] [CrossRef]
- Choudhari, S.M.; Ananthanarayan, L. Enzyme aided extraction of lycopene from tomato tissues. Food Chem. 2007, 102, 77–81. [Google Scholar] [CrossRef]
- Santos, F.T.; Goufo, P.; Santos, C.; Botelho, D.; Fonseca, J.; Queirós, A.; Costa, M.S.; Trindade, H. Comparison of five agro-industrial waste-based composts as growing media for lettuce: Effect on yield, phenolic compounds and vitamin C. Food Chem. 2016, 209, 293–301. [Google Scholar] [CrossRef] [PubMed]
- Srikanth, S.; Swathi, M.; Tejaswini, M.; Sharmila, G.; Muthukumaran, C.; Jaganathan, M.K.; Tamilarasan, K. Statistical optimization of molasses based exopolysaccharide and biomass production by Aureobasidium pullulans MTCC 2195. Biocatal. Agric. Biotechnol. 2014, 3, 7–12. [Google Scholar] [CrossRef]
- Pestana-Bauer, V.R.; Zambiazi, R.C.; Mendonça, C.R.; Beneito-Cambra, M.; Ramis-Ramos, G. γ-Oryzanol and tocopherol contents in residues of rice bran oil refining. Food Chem. 2012, 134, 1479–1483. [Google Scholar] [CrossRef] [PubMed]
- Galanakis, C.M.; Tornberg, E.; Gekas, V. Recovery and preservation of phenols from olive waste in ethanolic extracts. J. Chem. Technol. Biotechnol. 2010, 85, 1148–1155. [Google Scholar] [CrossRef]
- Jumah, R.; Al-Asheh, S.; Banat, F.; Al-Zoubi, K. Electro-osmotic dewatering of tomato paste suspension under AC electric field. Dry. Technol. 2005, 23, 1465–1475. [Google Scholar] [CrossRef]
- Koubala, B.B.; Mbome, L.I.; Kansci, G.; Mbiapo, F.T.; Crepeau, M.J.; Thibault, J.F.; Ralet, M.C. Physicochemical properties of pectins from ambarella peels (Spondias cytherea) obtained using different extraction conditions. Food Chem. 2008, 106, 1202–1207. [Google Scholar] [CrossRef]
- Galanakis, C.M. Recovery of high added-value components from food wastes: Conventional, emerging technologies and commercialized applications. Trends Food Sci. Technol. 2012, 26, 68–87. [Google Scholar] [CrossRef]
- Sowbhagya, H.B.; Chitra, V.N. Enzyme-assisted extraction of flavourings and colorants from plant materials. Crit. Rev. Food Sci. Nutr. 2010, 50, 146–161. [Google Scholar] [CrossRef]
- Tsakona, S.; Galanakis, C.M.; Gekas, V. Hydro-ethanolic mixtures for the recovery of phenols from Mediterranean plant materials. Food Bioproc. Technol. 2012, 5, 1384–1393. [Google Scholar] [CrossRef]
- Strati, I.F.; Oreopoulou, V. Effect of extraction parameters on the carotenoid recovery from tomato waste. Int. J. Food Sci. Technol. 2011, 46, 23–29. [Google Scholar] [CrossRef]
- Fierascu, R.C.; Ortan, A.; Avramescu, S.M.; Fierascu, I. Phyto-nanocatalysts: Green synthesis, characterization, and applications. Molecules 2019, 24, 3418. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sarkis, J.R.; Boussetta, N.; Tessaro, I.C.; Marczak, L.D.F.; Vorobiev, E. Application of pulsed electric fields and high voltage electrical discharges for oil extraction from sesame seeds. J. Food Eng. 2015, 153, 20–27. [Google Scholar] [CrossRef]
- Roselló-Soto, E.; Barba, F.J.; Parniakov, O.; Galanakis, C.M.; Lebovka, N.; Grimi, N.; Vorobiev, E. High voltage electrical discharges, pulsed electric field, and ultrasound assisted extraction of protein and phenolic compounds from olive kernel. Food Bioproc. Technol. 2015, 8, 885–894. [Google Scholar] [CrossRef]
- Ghafoor, K.; Park, J.; Choi, Y.H. Optimization of supercritical fluid extraction of bioactive compounds from grape (Vitis labrusca B.) peel by using response surface methodology. Innov. Food Sci. Emerg. Technol. 2011, 11, 485–490. [Google Scholar] [CrossRef]
- Périno-Issartier, S.; Huma, Z.; Abert-Vian, M.; Chemat, F. Solvent free microwave-assisted extraction of antioxidants from sea buckthorn (Hippophae rhamnoides) food by-products. Food Bioproc. Technol. 2011, 4, 1020–1028. [Google Scholar] [CrossRef]
- Naviglio, D.; Scarano, P.; Ciaravolo, M.; Gallo, M. Rapid Solid-Liquid Dynamic Extraction (RSLDE): A powerful and greener alternative to the latest solid-liquid extraction techniques. Foods 2019, 8, 245. [Google Scholar] [CrossRef] [Green Version]
- Posadino, A.; Biosa, G.; Zayed, H.; Abou-Saleh, H.; Cossu, A.; Nasrallah, G.; Giordo, R.; Pagnozzi, D.; Porcu, M.C.; Pretti, L.; et al. Protective effect of cyclically pressurized solid–liquid extraction polyphenols from Cagnulari grape pomace on oxidative endothelial cell death. Molecules 2018, 23, 2105. [Google Scholar] [CrossRef] [Green Version]
- Gallego, R.; Bueno, M.; Herrero, M. Sub- and supercritical fluid extraction of bioactive compounds from plants, food-by-products, seaweeds and microalgae – An update. TrAC Trends Anal. Chem. 2019, 116, 198–213. [Google Scholar] [CrossRef]
- Alonso, E. The role of supercritical fluids in the fractionation pretreatments of a wheat bran-based biorefinery. J. Supercrit. Fluids 2018, 133, 603–614. [Google Scholar] [CrossRef]
- Barba, F.J.; Zhu, Z.; Koubaa, M.; Sant’Ana, A.S.; Orlien, V. Green alternative methods for the extraction of antioxidant bioactive compounds from winery wastes and by-products: A review. Trends Food Sci. Technol. 2016, 49, 96–109. [Google Scholar] [CrossRef]
- Nguyen, T.T.; Barber, A.R.; Corbin, K.; Zhang, W. Lobster processing by-products as valuable bioresource of marine functional ingredients, nutraceuticals, and pharmaceuticals. Bioresour. Bioprocess 2017, 4, 27. [Google Scholar] [CrossRef] [PubMed]
- Kazan, A.; Celiktas, M.S.; Sargin, S.; Yesil-Celiktas, O. Bio-based fractions by hydrothermal treatment of olive pomace: Process optimization and evaluation. Energy Convers. Manag. 2015, 103, 366–373. [Google Scholar] [CrossRef]
- Ko, M.J.; Kwon, H.L.; Chung, M.S. Pilot-scale subcritical water extraction of flavonoids from satsuma Mandarin (Citrus unshiu Markovich) peel. Innov. Food Sci. Emerg. Technol. 2016, 38, 175–181. [Google Scholar] [CrossRef]
- Vergara-Salinas, J.R.; Vergara, M.; Altamirano, C.; Gonzalez, Á.; Pérez-Correa, J.R. Characterization of pressurized hot water extracts of grape pomace: Chemical and biological antioxidant activity. Food Chem. 2015, 171, 62–69. [Google Scholar] [CrossRef]
- Mariotti-Celis, M.S.; Martínez-Cifuentes, M.; Huamán-Castilla, N.; Pedreschi, F.; Iglesias-Rebolledo, N.; Pérez-Correa, J.R. Impact of an integrated process of hot pressurised liquid extraction-macroporous resin purification over the polyphenols, hydroxymethylfurfural and reducing sugars content of Vitis vinifera ‘Carménère’ pomace extracts. Int. J. Food Sci. Technol. 2018, 53, 1072–1078. [Google Scholar] [CrossRef]
- Pereira, D.T.V.; Tarone, A.G.; Cazarin, C.B.B.; Barbero, G.F.; Martínez, J. Pressurized liquid extraction of bioactive compounds from grape marc. J. Food Eng. 2019, 240, 105–113. [Google Scholar] [CrossRef]
- Alexandre, E.M.C.; Silva, S.; Santos, S.A.O.; Silvestre, A.J.D.; Duarte, M.F.; Saraiva, J.A.; Pintado, M. Antimicrobial activity of pomegranate peel extracts performed by high pressure and enzymatic assisted extraction. Food Res. Int. 2019, 115, 167–176. [Google Scholar] [CrossRef] [Green Version]
- Da Porto, C.; Natolino, A.; Decorti, D. The combined extraction of polyphenols from grape marc: Ultrasound assisted extraction followed by supercritical CO2 extraction of ultrasound-raffinate. LWT Food Sci. Technol. 2015, 61, 98–104. [Google Scholar] [CrossRef] [Green Version]
- Chen, H.; Fu, X.; Luo, Z. Properties and extraction of pectin-enriched materials from sugar beet pulp by ultrasonic-assisted treatment combined with subcritical water. Food Chem. 2015, 168, 302–310. [Google Scholar] [CrossRef]
- Sumere, B.R.; de Souza, M.C.; dos Santos, M.P.; Bezerra, R.M.N.; da Cunha, D.T.; Martinez, J.; Rostagno, M.A. Combining pressurized liquids with ultrasound to improve the extraction of phenolic compounds from pomegranate peel (Punica granatum L.). Ultrason. Sonochem. 2018, 48, 151–162. [Google Scholar] [CrossRef]
- Boukroufa, M.; Boutekedjiret, C.; Petigny, L.; Rakotomanomana, N. Bio-refinery of orange peels waste: A new concept based on integrated green and solvent free extraction processes using ultrasound and microwave techniques to obtain essential oil, polyphenols and pectin. Ultrason. Sonochem. 2015, 24, 72–79. [Google Scholar] [CrossRef]
- Grassino, A.N.; Brnčić, M.; Vikić-Topić, D.; Roca, S.; Dent, M.; Rimac Brnčić, S. Ultrasound assisted extraction and characterization of pectin from tomato waste. Food Chem. 2016, 198, 93–100. [Google Scholar] [CrossRef]
- Minjares-Fuentes, R.; Femenia, A.; Garau, M.C.; Meza-Velázquez, J.A.; Simal, S.; Rosselló, C. Ultrasound-assisted extraction of pectins from grape pomace using citric acid: A response surface methodology approach. Carbohydr. Polym. 2014, 106, 179–189. [Google Scholar] [CrossRef]
- Routray, W.; Orsat, V. Microwave-assisted extraction of flavonoids: A review. Food Bioproc. Technol. 2012, 5, 409–424. [Google Scholar] [CrossRef]
- Şahin, S. A novel technology for extraction of phenolic antioxidants from Mandarin (Citrus deliciosa Tenore) leaves: Solvent-free microwave extraction. Korean J. Chem. Eng. 2015, 32, 950–957. [Google Scholar] [CrossRef]
- Llompart, M.; Celeiro, M.; Dagnac, T. Microwave-assisted extraction of pharmaceuticals, personal care products and industrial contaminants in the environment. TrAC Trends Anal. Chem. 2019, 116, 136–150. [Google Scholar] [CrossRef]
- Phongthai, S.; Lim, S.T.; Rawdkuen, S. Optimization of microwave-assisted extraction of rice bran protein and its hydrolysates properties. J. Cereal Sci. 2016, 70, 146–154. [Google Scholar] [CrossRef]
- Rodsamran, P.; Sothornvit, R. Microwave heating extraction of pectin from lime peel: Characterization and properties compared with the conventional heating method. Food Chem. 2019, 278, 364–372. [Google Scholar] [CrossRef]
- Kumar, M.; Dahuja, A.; Sachdev, A.; Kaur, C.; Varghese, E.; Saha, S.; Sairam, K.V.S.S. Evaluation of enzyme and microwave-assisted conditions on extraction of anthocyanins and total phenolics from black soybean (Glycine max L.) seed coat. Int. J. Biol. Macromol. 2019, 135, 1070–1081. [Google Scholar] [CrossRef]
- Dávila, I.; Remón, J.; Gullón, P.; Labidi, J.; Budarin, V. Production and characterization of lignin and cellulose fractions obtained from pretreated vine shoots by microwave assisted alkali treatment. Biores. Technol. 2019, 289, 121726. [Google Scholar] [CrossRef]
- Puértolas, E.; Barba, F.J. Electrotechnologies applied to valorization of by-products from food industry: Main findings, energy and economic cost of their industrialization. Food Bioprod. Process. 2016, 100, 172–184. [Google Scholar] [CrossRef]
- Barba, F.J.; Puértolas, E.; Brncic, M.; Panchev, I.N.; Dimitrov, D.A.; Athes-Dutour, V.; Mousaa, M.; Souchon, I. Emerging extraction. In Food Waste Recovery Processing Technologies and Industrial Techniques, 1st ed.; Galanakis, C.M., Ed.; Academic Press, Elsevier: Oxford, UK, 2015; pp. 249–272. [Google Scholar]
- Boussetta, N.; Soichi, E.; Lanoiselle, J.L.; Vorobiev, E. Valorization of oilseed residues: Extraction of polyphenols from flaxseed hulls by pulsed electric fields. Ind. Crops Prod. 2014, 52, 347–353. [Google Scholar] [CrossRef]
- Parniakov, O.; Barba, F.J.; Grimi, N.; Lebovka, N.; Vorobiev, E. Extraction assisted by pulsed electric energy as a potential tool for green and sustainable recovery of nutritionally valuable compounds from mango peels. Food Chem. 2015, 196, 842–848. [Google Scholar] [CrossRef]
- Rajha, H.N.; Boussetta, N.; Louka, N.; Maroun, R.G.; Vorobiev, E. Mechanical and non-mechanical effects of high-voltage electrical discharges on polyphenol extraction from vine shoots. In Proceedings of the Electroporation-based Technologies for Biorefinery Workshop, Compiègne, France, 27–28 January 2015. [Google Scholar]
- Luengo, E.; Álvarez, I.; Raso, J. Improving the pressingextraction of polyphenols of orange peel by pulsed electric fields. Innov. Food Sci. Emerg. Technol. 2013, 17, 79–84. [Google Scholar] [CrossRef]
- Ferri, F.; Bertina, L.; Scoma, A.; Marchetti, L.; Fava, F. Recovery of low molecular weight phenols through solid-phase extraction. Chem. Eng. J. 2011, 166, 994–1001. [Google Scholar] [CrossRef]
- Fernández-Bolaños, J.; Heredia, A.; Rodríguez, G.; Rodríguez, R.; Guillén, R.; Jiménez, A. Method for Obtaining Purified Hydroxytyrosol from Products and By-Products Derived from the Olive Tree. Patent No. WO/2002/064537, 8 February 2002. [Google Scholar]
- González, M.I.; Alvarez, S.; Riera, F.A.; Álvarez, R. Lactic acid recovery from whey ultrafiltrate fermentation broths and artificial solutions by nanofiltration. Desalination 2008, 228, 84–96. [Google Scholar] [CrossRef]
- Doyen, A.; Beaulieu, L.; Saucier, L.; Pouliot, Y.; Bazinet, L. Impact of ultrafiltration membrane material on peptide separation from a snow crab byproduct hydrolysate by electrodialysis with ultrafiltration membranes. J. Agricult. Food Chem. 2011, 59, 1784–1792. [Google Scholar] [CrossRef]
- Nayak, A.; Bhushan, B. An overview of the recent trends on the waste valorization techniques for food wastes. J. Environ. Manag. 2019, 233, 352–370. [Google Scholar] [CrossRef]
- Awalludin, M.F.; Othman, S.; Rokiah, H.; Nadhari, W.N.A.W. An overview of the oil palm industry in Malaysia and its waste utilization through thermochemical conversion, specifically via liquefaction. Renew. Sus. Energ. Rev. 2015, 50, 1469–1484. [Google Scholar] [CrossRef]
- Hadi, A.N.; Ng, H.M.; Choo, Y.; Hashim, A.M.; Jayakumar, S.N. Performance of choline-based deep eutectic solvents in the extraction of tocols from crude palm oil. J. Am. Oil. Chem. Soc. 2015, 92, 1709–1716. [Google Scholar] [CrossRef]
- Ahmad, F.B.; Zhang, Z.; Doherty, W.O.S.; O’Hara, I.M. The outlook of the production of advanced fuels and chemicals from integrated oil palm biomass biorefinery. Renew. Sus. Energ. Rev. 2019, 109, 386–411. [Google Scholar] [CrossRef]
- Aditiya, H.B.; Chong, W.T.; Mahlia, T.M.I.; Sebayang, A.H.; Berawi, M.A.; Nur, H. Second generation bioethanol potential from selected Malaysia’s biodiversity biomasses: A review. Waste Manag. 2016, 47, 46–61. [Google Scholar] [CrossRef]
- Cardenas-Toro, F.P.; Alcazar-Alay, S.C.; Coutinho, J.P.; Godoy, H.T.; Forster-Carneiro, T.; Meireles, M.A.A. Pressurized liquid extraction and low-pressure solvent extraction of carotenoids from pressed palm fiber: Experimental and economical evaluation. Food Bioprod. Process. 2015, 94, 90–100. [Google Scholar] [CrossRef]
- Dal Prá, V.; Soares, J.F.; Monego, D.L.; Vendruscolo, R.G.; Freire, D.M.G.; Alexandri, M.; Koutinas, A.; Wagner, R.; Mazutti, M.A.; da Rosa, M.B. Extraction of bioactive compounds from palm (Elaeis guineensis) pressed fiber using different compressed fluids. J. Supercrit. Fluids 2016, 112, 51–56. [Google Scholar] [CrossRef]
- Dal Prá, V.; Lunelli, F.C.; Vendruscolo, R.G.; Martins, R.; Wagner, R.; Lazzaretti, A.P., Jr.; Freire, D.M.G.; Alexandri, M.; Koutinas, A.; Mazutti, M.A.; et al. Ultrasound-assisted extraction of bioactive compounds from palm pressed fiber with high antioxidant and photoprotective activities. Ultrason. Sonochem. 2017, 36, 362–366. [Google Scholar] [CrossRef]
- Lee, Z.S.; Chin, S.Y.; Lim, J.W.; Witoon, T.; Cheng, C.K. Treatment technologies of palm oil mill effluent (POME) and olive mill wastewater (OMW): A brief review. Environ. Technol. Innov. 2019, 15, 100377. [Google Scholar] [CrossRef]
- Rodríguez-Gutiérrez, G.; Lama-Muñoz, A.; Ruiz-Méndez, M.V.; Rubio-Senent, F.; Fernández-Bolaños, J. New olive-pomace oil improved by hydrothermal pre-treatments. In Olive Oil—Constituents, Quality, Health Properties and Bioconversions; Boskou, D., Ed.; InTech: Rijeka, Croatia, 2012; pp. 249–266. [Google Scholar]
- García, A.; Rodríguez-Juan, E.; Rodríguez-Gutiérrez, G.; Rios, J.J.; Fernández-Bolaños, J. Extraction of phenolic compounds from virgin olive oil by deep eutectic solvents (DESs). Food Chem. 2016, 197, 554–561. [Google Scholar] [CrossRef]
- Lama-Muñoz, A.; Del Mar Contreras, M.; Espínola, F.; Moya, M.; de Torres, A.; Romero, I.; Castro, E. Extraction of oleuropein and luteolin-7-O-glucoside from olive leaves: Optimization of technique and operating conditions. Food Chem. 2019, 293, 161–168. [Google Scholar] [CrossRef]
- Martínez-Patiño, J.C.; Gullón, B.; Romero, I.; Ruiz, E.; Brnčić, M.; Šic Žlabur, J.; Castro, E. Optimization of ultrasound-assisted extraction of biomass from olive trees using response surface methodology. Ultrason. Sonochem. 2019, 51, 487–495. [Google Scholar] [CrossRef] [PubMed]
- Xie, P.; Huang, L.; Zhang, C.; Xiaoji, Y.D.; Cheng, W.J. Enhanced extraction of hydroxytyrosol, maslinic acid and oleanolic acid from olive pomace: Process parameters, kinetics and thermodynamics, and greenness assessment. Food Chem. 2019, 276, 662–674. [Google Scholar] [CrossRef] [PubMed]
- Issaoui, A.; Ksibib, H.; Ksibi, M. Supercritical fluid extraction of triterpenes and aliphatic hydrocarbons from olive tree derivatives. Arab. J. Chem. 2017, 10, S3967–S3973. [Google Scholar] [CrossRef] [Green Version]
- Wang, Z.; Wang, C.; Zhang, C.; Li, W. Ultrasound-assisted enzyme catalyzed hydrolysis of olive waste and recovery of antioxidant phenolic compounds. Innov. Food Sci. Emerg. Technol. 2017, 44, 224–234. [Google Scholar] [CrossRef]
- Martínez-Force, E.; Dunford, N.T.; Salas, J.J. Sunflower: Chemistry, Production, Processing, and Utilization; Academic Press and AOCS Press: Urbana, IL, USA, 2015. [Google Scholar]
- Weisz, G.M.; Kammerer, D.R.; Carle, R. Identification and quantification of phenolic compounds from sunflower (Helianthus annuus L.) kernels and shells by HPLC-DAD/ESI-MSn. Food Chem. 2009, 115, 758–765. [Google Scholar] [CrossRef]
- Fuentes-Gandara, F.; Torres, A.; Fernández-Ponce, M.T.; Casas, L.; Mantell, C.; Varela, R.; Martínez de la Ossa-Fernández, E.J.; Macías, F.A. Selective fractionation and isolation of allelopathic compounds from Helianthus annuus L. leaves by means of high-pressure techniques. J. Supercrit. Fluids 2019, 143, 32–41. [Google Scholar] [CrossRef]
- Purkayastha, M.D.; Dutta, N.; Kalita, D.; Mahanta, C. Exploratory analysis for characterization of solvent-treated products (meal and extract) from rapeseed press-cake: Preliminary investigation using principal component analysis. Waste Biomass Valoriz. 2014, 5, 835–846. [Google Scholar] [CrossRef]
- Grübel, K.; Kuglarz, M.; Wacławek, S.; Padil, V.V.T.; Černík, M.; Varma, R.S. Microwave-assisted sustainable co-digestion of sewage sludge and rapeseed cakes. Energ. Convers. Manag. 2019, 199, 112012. [Google Scholar] [CrossRef]
- Barba, F.J.; Boussetta, N.; Vorobiev, E. Emerging technologies for the recovery of isothiocyanates, protein and phenolic compounds from rapeseed and rapeseed press-cake: Effect of high voltage electrical discharges. Innov. Food Sci. Emerg. Technol. 2015, 31, 67–72. [Google Scholar] [CrossRef]
- Patel, S. Pumpkin (Cucurbita sp.) seeds as nutraceutic: A review on status quo and scopes. Med. J. Nutrition Metab. 2013, 6, 183–189. [Google Scholar] [CrossRef]
- Ferreira, D.F.; Barin, J.S.; Binello, A.; Veselov, V.V.; Cravotto, G. Highly efficient pumpkin-seed extraction with the simultaneous recovery of lipophilic and hydrophilic compounds. Food Bioprod. Process. 2019, 117, 224–230. [Google Scholar] [CrossRef]
- Zuk, M.; Richter, D.; Matuła, J.; Szopa, J. Linseed, the multipurpose plant. Ind. Crops Prod. 2015, 75, 165–177. [Google Scholar] [CrossRef]
- Chang, J.I.; Tsai, J.J.; Wu, K.H. Composting of vegetable waste. Waste Manag. Res. 2006, 24, 354–362. [Google Scholar] [CrossRef] [PubMed]
- Plazzotta, S.; Manzocco, L.; Nicoli, M.C. Fruit and vegetable waste management and the challenge of fresh-cut salad. Trends Food Sci. Technol. 2017, 63, 51–59. [Google Scholar] [CrossRef]
- Food and Agriculture Organization of the United Nations. Available online: http://www.fao.org/faostat/en/#data (accessed on 2 September 2019).
- Coman, V.; Teleky, B.E.; Mitrea, L.; Martău, G.A.; Szabo, K.; Călinoiu, L.F.; Vodnar, D.C. Bioactive potential of fruit and vegetable wastes. Adv. Food Nutr. Res. 2019, in press. [Google Scholar] [CrossRef]
- Food and Agriculture Organization of the United Nations. Global Food Losses and Waste. Extent, Causes and Prevention. Available online: http://www.fao.org/docrep/014/mb060e/mb060e00.pdf (accessed on 2 September 2019).
- Porat, R.; Lichter, A.; Terry, L.A.; Harker, R.; Buzby, J. Postharvest losses of fruit and vegetables during retail and in consumers’ homes: Quantifications, causes, and means of prevention. Postharvest Biol. Technol. 2018, 139, 135–149. [Google Scholar] [CrossRef] [Green Version]
- De Laurentiis, V.; Corrado, S.; Sala, S. Quantifying household waste of fresh fruit and vegetables in the EU. Waste Manag. 2018, 77, 238–251. [Google Scholar] [CrossRef]
- Gouw, V.P.; Jung, J.; Zhao, Y. Functional properties, bioactive compounds, and in vitro gastrointestinal digestion study of dried fruit pomace powders as functional food ingredients. LWT Food Sci. Technol. 2017, 80, 136–144. [Google Scholar] [CrossRef]
- Tarko, T.; Duda-Chodak, A.; Bebak, A. Aktywność biologiczna wybranych wytłoków owocowych oraz warzywnych. Żywność-Nauka Technologia Jakość 2012, 4, 55–65. [Google Scholar]
- Majerska, J.; Michalsk, A.; Figiel, A. A review of new directions in managing fruit and vegetable processing by-products. Trends Food Sci. Technol. 2019, 88, 207–219. [Google Scholar] [CrossRef]
- Johnson, M.H.; De Mejia, E.G.; Fan, J.; Lila, M.A.; Yousef, G.G. Anthocyanins and proanthocyanidins from blueberry–blackberry fermented beverages inhibit markers of inflammation in macrophages and carbohydrate-utilizing enzymes in vitro. Mol. Nutr. Food Res. 2013, 57, 1182–1197. [Google Scholar] [CrossRef]
- Juan, C.; Jianquan, K.; Junni, T.; Zijian, C.; Ji, L. The profile in polyphenols and volatile compounds in alcoholic beverages from different cultivars of mulberry. J. Food Sci. 2012, 77, C430–C436. [Google Scholar] [CrossRef]
- Shahidi, F.; Ambigaipalan, P. Phenolics and polyphenolics in foods, beverages and spices: Antioxidant activity and health effects—A review. J. Funct. Foods 2015, 18, 820–897. [Google Scholar] [CrossRef]
- Alhamad, M.N.; Rababah, T.M.; Al-u’datt, M.; Ereifej, K.; Esoh, R.; Feng, H.; Yang, W. The physicochemical properties, total phenolic, antioxidant activities, and phenolic profile of fermented olive cake. Arab. J. Chem. 2017, 10, 136–140. [Google Scholar] [CrossRef] [Green Version]
- Nile, S.H.; Nile, A.; Liu, J.; Kim, D.H.; Kai, G. Exploitation of apple pomace towards extraction of triterpenic acids, antioxidant potential, cytotoxic effects, and inhibition of clinically important enzymes. Food Chem. Toxicol. 2019, 131, 110563. [Google Scholar] [CrossRef]
- Chandrasekar, V.; San Martín-González, M.F.; Hirst, P.; Ballard, T.S. Optimizing microwave-assisted extraction of phenolic antioxidants from Red Delicious and Jonathan apple pomace. J. Food Process. Eng. 2015, 38, 571–582. [Google Scholar] [CrossRef]
- Del Pilar Sánchez-Camargo, A.; Gutiérrez, L.F.; Milena Vargas, S.; Martinez-Correa, H.A.; Parada-Alfonso, F.; Narváez-Cuenca, C.E. Valorisation of mango peel: Proximate composition, supercritical fluid extraction of carotenoids, and application as an antioxidant additive for an edible oil. J. Supercrit. Fluids 2019, 152, 104574. [Google Scholar] [CrossRef]
- Ozturk, B.; Winterburn, J.; Gonzalez-Miquel, M. Orange peel waste valorisation through limonene extraction using bio-based solvents. Biochem. Eng. J. 2019, 151, 107298. [Google Scholar] [CrossRef]
- Jessop, P.G. Searching for green solvents. Green Chem. 2011, 13, 1391–1398. [Google Scholar] [CrossRef]
- Klamt, A.; Eckert, F. COSMO-RS: A novel and efficient method for the a priori prediction of thermophysical data of liquids. Fluid Phase Equilib. 2000, 172, 43–72. [Google Scholar] [CrossRef] [Green Version]
- Grillo, G.; Boffa, L.; Binello, A.; Mantegna, S.; Cravotto, G.; Chemat, F.; Dizhbite, T.; Lauberte, L.; Telysheva, G. Cocoa bean shell waste valorisation; extraction from lab to pilot-scale cavitational reactors. Food Res. Int. 2019, 115, 200–208. [Google Scholar] [CrossRef]
- Okiyama, D.G.C.; Soares, I.D.; Cuevas, M.S.; Crevelin, E.J.; Moraes, L.A.B.; Melo, M.P.; Oliveira, A.L.; Rodrigues, C.E.C. Pressurized liquid extraction of flavanols and alkaloids from cocoa bean shell using ethanol as solvent. Food Res. Int. 2018, 114, 20–29. [Google Scholar] [CrossRef]
- Yilmaz, T.; Kumcuoglu, S.; Tavman, S. Ultrasound-assisted extraction of lycopene and β-carotene from 1242 tomato-processing wastes. Italian J. Food Sci. 2016, 29, 186–194. [Google Scholar]
- Lu, Z.; Wang, J.; Gao, R.; Ye, F.; Zhao, G. Sustainable valorisation of tomato pomace: A comprehensive review. Trends Food Sci. Technol. 2019, 86, 172–187. [Google Scholar] [CrossRef]
- Coelho, M.; Pereira, R.; Rodrigues, A.S.; Teixeira, J.A.; Pintado, M.E. Extraction of tomato by-products’ bioactive compounds using ohmic technology. Food Bioprod. Process. 2019, 117, 329–339. [Google Scholar] [CrossRef] [Green Version]
- Kim, S.W.; Ko, M.J.; Chung, M.S. Extraction of the flavonol quercetin from onion waste by combined treatment with intense pulsed light and subcritical water extraction. J. Clean. Prod. 2019, 231, 1192–1199. [Google Scholar] [CrossRef]
- Oms-Oliu, G.; Aguiló-Aguayo, I.; Martín-Belloso, O.; Soliva-Fortuny, R. Effects of pulsed light treatments on quality and antioxidant properties of fresh-cut mushrooms (Agaricus bisporus). Postharvest Biol. Technol. 2010, 56, 216–222. [Google Scholar] [CrossRef]
- Derrien, M.; Aghabararnejad, M.; Gosselin, A.; Desjardins, Y.; Angersa, P.; Boumghar, Y. Optimization of supercritical carbon dioxide extraction of lutein and chlorophyll from spinach by-products using response surface methodology. LWT Food Sci. Technol. 2018, 93, 79–87. [Google Scholar] [CrossRef]
- Goula, A.M.; Ververi, M.; Adamopoulou, A.; Kaderides, K. Green ultrasound-assisted extraction of carotenoids from pomegranate wastes using vegetable oils. Ultrason. Sonochem. 2017, 34, 821–830. [Google Scholar] [CrossRef]
- Food and Agriculture Organization of the United Nations—Food and Agriculture Data. Available online: http://www.fao.org/faostat/en/ (accessed on 5 September 2019).
- Kaspar, A.A.; Reichert, J.M. Future directions for peptide therapeutics development. Drug Discov. Today 2013, 18, 807–817. [Google Scholar] [CrossRef]
- Stenmarck, A.; Jensen, C.; Quested, T.; Moates, G.; Buksti, M.; Cseh, B.; Juul, S.; Parry, A.; Polotano, A.; Redlinghofer, B.; et al. Estimates of European Food Waste Levels; IVL Swedish Environmental Research Institute: Stockholm, Sweden, 2016. [Google Scholar]
- Tonini, D.; Albizzati, P.F.; Astrup, T.F. Environmental impacts of food waste: Learnings and challenges from a case study on UK. Waste Manag. 2018, 76, 744–766. [Google Scholar] [CrossRef]
- Bai, C.; Wei, Q.; Ren, X. Selective extraction of collagen peptides with high purity from cod skins by deep eutectic solvents (DESs). ACS Sustain. Chem. Eng. 2017, 5, 7220–7227. [Google Scholar] [CrossRef]
- Marcet, I.; Alvarez, C.; Paredes, B.; Diaz, M. The use of sub-critical water hydrolysis for the recovery of peptides and free amino acids from food processing wastes. Review of sources and main parameters. Waste Manag. 2016, 49, 364–371. [Google Scholar] [CrossRef]
- Udenigwe, C.C.; Girgih, A.T.; Mohan, A.; Gong, M.; Malomo, S.A.; Aluko, R.E. Antihypertensive and bovine plasma oxidation-inhibitory activities of spent hen meat protein hydrolysates. J. Food Biochem. 2017, 41, e12378. [Google Scholar] [CrossRef]
- Yu, W.; Field, C.J.; Wu, J. Purification and identification of anti-inflammatory peptides from spent hen muscle proteins hydrolysate. Food Chem. 2018, 253, 101–107. [Google Scholar] [CrossRef]
- Maikhunthod, B.; Intarapichet, K.O. Heat and ultrafiltration extraction of broiler meat carnosine and its antioxidant activity. Meat Sci. 2005, 71, 364–374. [Google Scholar] [CrossRef]
- Golberg, A.; Sack, M.; Teissie, J.; Pataro, G.; Pliquett, U.; Saulis, G.; Stefan, T.; Miklavcic, D.; Vorobiev, E.; Frey, W.; et al. Energy efficient biomass processing with pulsed electric fields for bioeconomy and sustainable development. Biotechnol. Biofuels 2016, 9, 94. [Google Scholar] [CrossRef] [Green Version]
- Marcet, I.; Alvarez, C.; Paredes, B.; Diaz, M. Inert and oxidative subcritical water hydrolysis of insoluble egg yolk granular protein, functional properties, and comparison to enzymatic hydrolysis. J. Agric. Food Chem. 2014, 62, 8179–8186. [Google Scholar] [CrossRef]
- Asaduzzaman, A.K.M.; Chun, B.S. Recovery of functional materials with thermally stable antioxidative properties in squid muscle hydrolyzates by subcritical water. J. Food Sci. Technol. 2015, 52, 793–802. [Google Scholar] [CrossRef] [Green Version]
- Zhu, G.Y.; Zhu, X.; Wan, X.L.; Fan, Q.; Ma, Y.H.; Qian, J.; Liu, X.L.; Shen, Y.J.; Jiang, J.H. Hydrolysis technology and kinetics of poultry waste to produce amino acids in subcritical water. J. Anal. Appl. Pyrol. 2010, 88, 187–191. [Google Scholar] [CrossRef]
- Pereira, R.N.; Rodrigues, R.M.; Ramos, O.L.; Malcata, F.X.; Teixeira, J.A.; Vicente, A.A. Production of whey protein-based aggregates under ohmic heating. Food Bioprocess Technol. 2016, 9, 576–587. [Google Scholar]
- Ghosh, S.; Gillis, A.; Sheviryov, J.; Levkov, K.; Golberg, A. Towards waste meat biorefinery: Extraction of proteins from waste chicken meat with non-thermal pulsed electric fields and mechanical pressing. J. Clean. Prod. 2019, 208, 220–231. [Google Scholar] [CrossRef]
- Mussatto, S.I. Biotechnological potential of brewing industry by-products. In Biotechnology for Agro-Industrial Residues Utilisation: Utilisation of Agro-Residues; Singhnee’Nigam, P., Pandey, A., Eds.; Springer Netherlands: Dordrecht, The Netherlands, 2009; pp. 313–326. [Google Scholar]
- Chae, H.J.; Joo, H.; In, M.J. Utilization of brewer’s yeast cells for the production of food-grade yeast extract. Part 1: Effects of different enzymatic treatments on solid and protein recovery and flavor characteristics. Bioresour. Technol. 2001, 76, 253–258. [Google Scholar] [CrossRef]
- Callemien, D.; Collin, S. Structure, organoleptic properties, quantification methods, and stability of phenolic compounds in beer—A review. Food Rev. Int. 2010, 26, 1–84. [Google Scholar] [CrossRef]
- Barbosa-Pereira, L.; Bilbao, A.; Vilches, P.; Angulo, I.; LLuis, J.; Fité, B.; Paseiro-Losada, P.; Cruz, J.M. Brewery waste as a potential source of phenolic compounds: Optimisation of the extraction process and evaluation of antioxidant and antimicrobial activities. Food Chem. 2014, 145, 191–197. [Google Scholar] [CrossRef] [PubMed]
- Trivedi, J.; Aila, M.; Bangwal, D.P.; Kaul, S.; Garg, M.O. Algae based biorefinery—How to make sense? Renew. Sustain. Energy Rev. 2015, 47, 295–307. [Google Scholar] [CrossRef]
- Plaza, M.; Santoyo, S.; Jaime, L.; García-Blairsy Reina, G.; Herrero, M.; Señoráns, F.J.; Ibáñez, E. Screening for bioactive compounds from algae. J. Pharm. Biomed. Anal. 2010, 51, 450–455. [Google Scholar] [CrossRef]
- Rodriguez-Jasso, R.M.; Mussatto, S.I.; Pastrana, L.; Aguilar, C.N.; Teixeira, J.A. Microwave-assisted extraction of sulfated polysaccharides (fucoidan) from brown edible algae. Carbohydr. Polym. 2011, 86, 1137–1144. [Google Scholar] [CrossRef] [Green Version]
- Quitain, A.T.; Kai, T.; Sasaki, M.; Goto, M. Microwave–hydrothermal extraction and degradation of fucoidan from supercritical carbon dioxide deoiled Undaria pinnatifida. Ind. Eng. Chem. Res. 2013, 52, 7940–7946. [Google Scholar] [CrossRef]
- Men’shova, R.V.; Lepeshkin, F.D.; Ermakova, S.P.; Pokrovskii, O.I.; Zvyagintseva, T.N. Effect of pretreatment conditions of brown algae by supercritical fluids on yield and structural characteristics of fucoidans. Chem. Nat. Comp. 2013, 48, 923–926. [Google Scholar] [CrossRef]
- He, Z.; Chen, Y.; Chen, Y.; Liu, H.; Yuan, G.; Fan, Y.; Chen, K. Optimization of the microwave-assisted extraction of phlorotannins from Saccharina japonica Aresch and evaluation of the inhibitory effects of phlorotannin-containing extracts on HepG2 cancer cells. Chin. J. Ocean. Limnol. 2013, 31, 1045–1054. [Google Scholar] [CrossRef]
- Xiao, X.H.; Yuan, Z.Q.; Li, G.K. Preparation of phytosterols and phytol from edible marine algae by microwave-assisted extraction and high-speed counter-current chromatography. Sep. Purif. Technol. 2013, 104, 284–289. [Google Scholar] [CrossRef]
- Durmaz, Y.; Monteiro, M.; Bandarra, N.; Gökpinar, Ş.; Işik, O. The effect of low temperature on fatty acid composition and tocopherols of the red microalga, Porphyridium cruentum. J. Appl. Phycol. 2007, 19, 223–227. [Google Scholar] [CrossRef]
- Grimi, N.; Dubois, A.; Marchal, L.; Jubeau, S.; Lebovka, N.I.; Vorobiev, E. Selective extraction from microalgae Nannochloropsis sp. using different methods of cell disruption. Biores. Technol. 2014, 153, 254–259. [Google Scholar] [CrossRef]
- Wang, J.; Zhang, M.; Fang, Z. Recent development in efficient processing technology for edible algae: A review. Trends Food Sci. Technol. 2019, 88, 251–259. [Google Scholar] [CrossRef]
- Faustino, M.; Veiga, M.; Sousa, P.; Costa, E.M.; Silva, S.; Pintado, M. Agro-food byproducts as a new source of natural food additives. Molecules 2019, 24, 1056. [Google Scholar] [CrossRef] [Green Version]
- Mahato, N.; Sharma, K.; Sinha, M.; Cho, M.H. Citrus waste derived nutra-/pharmaceuticals for health benefits: Current trends and future perspectives. J. Funct. Foods 2018, 40, 307–316. [Google Scholar] [CrossRef]
- Lavecchia, R.; Zuorro, A. Evaluation of olive pomace as a source of phenolic antioxidants for the production of functional cosmetics. Int. J. Appl. Eng. Res. 2015, 10, 34405–34409. [Google Scholar]
- Pacifico, S.; Piccolella, S.; Veneziano, R. New sustainable cosmetic products from food waste: A joined-up approach between design and food chemistry. In Designing Sustainability for All, Proceedings of the 3rd LeNS World Distributed Conference (3rd volume), Milano, Mexico City, Beijing, Bangalore, Curitiba, Cape Town, 3–5 April 2019; Ambrosio, M., Vezzoli, C., Eds.; Edizioni POLI.design: Milano, Italy, 2019; pp. 975–979. [Google Scholar]
- Han, Y.; Yu, M.; Wang, L. Bio-based films prepared with soybean by-products and pine (Pinus densiflora) bark extract. J. Clean. Prod. 2018, 187, 1–8. [Google Scholar] [CrossRef]
- Han, Y.; Yu, M.; Wang, L. Preparation and characterization of antioxidant soy protein isolate films incorporating licorice residue extract. Food Hydrocoll. 2018, 75, 13–21. [Google Scholar] [CrossRef]
- Maryam, Z.A.; Jamilaha, A.B.; Nur Hanania, Z.A. Functional and antioxidant properties of protein-based films incorporated with mango kernel extract for active packaging. Food Hydrocoll. 2018, 74, 207–218. [Google Scholar] [CrossRef]
- Ahn, J.; Grun, I.; Fernando, L. Antioxidant properties of natural plant extracts containing polyphenolic compounds in cooked ground beef. J. Food Sci. 2002, 67, 1364–1369. [Google Scholar] [CrossRef]
- Azman, N.; Skowyra, M.; Muhammad, K.; Gallego, M.; Almajano, M. Evaluation of the antioxidant activity of Betula pendula leaves extract and its effects on model foods. Pharmaceut. Biol. 2017, 55, 912–919. [Google Scholar] [CrossRef] [PubMed]
- Carpenter, R.; O’Grady, M.; O’Callaghan, Y.; O’Brien, N.; Kerry, J. Evaluation of the antioxidant potential of grape seed and bearberry extracts in raw and cooked pork. Meat Sci. 2007, 76, 604–610. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.; Reed, J.; Richards, M. Ability of various polyphenolic classes from cranberry to inhibit lipid oxidation in mechanically separated Turkey and cooked ground pork. J. Muscle Foods. 2001, 67, 248–266. [Google Scholar] [CrossRef]
- Karre, L.; Lopez, K.; Getty, K. Natural antioxidants in meat and poultry products. Meat Sci. 2013, 94, 220–227. [Google Scholar] [CrossRef] [PubMed]
- Raghavan, S.; Richards, M. Partitioning and inhibition of lipid oxidation in mechanically separated Turkey by components of cranberry press cake. J. Agricult. Food Chem. 2006, 54, 6403–6408. [Google Scholar] [CrossRef]
- Zhang, H.; Wu, J.; Guo, X. Effects of antimicrobial and antioxidant activities of spice extracts on raw chicken meat quality. Food Sci. Human Wellness 2016, 5, 39–48. [Google Scholar] [CrossRef] [Green Version]
- Casarotti, S.; Jorge, N. Antioxidant activity of rosemary extract in soybean oil under thermoxidation. J. Food Process. Preserv. 2014, 38, 136–145. [Google Scholar] [CrossRef]
- Mohdaly, A.; Sarhan, M.; Mahmoud, A.; Ramadan, M.; Smetanska, I. Antioxidant efficacy of potato peels and sugar beet pulp extracts in vegetable oils protection. Food Chem. 2010, 123, 1019–1026. [Google Scholar] [CrossRef]
- Peng, X.; Ma, J.; Cheng, K.W.; Jiang, Y.; Chen, F.; Wang, M. The effects of grape seed extract fortification on the antioxidant activity and quality attributes of bread. Food Chem. 2010, 119, 49–53. [Google Scholar] [CrossRef]
- Reddy, V.; Urooj, A.; Kumar, A. Evaluation of antioxidant activity of some plant extracts and their application in biscuits. Food Chem. 2005, 90, 317–321. [Google Scholar] [CrossRef] [Green Version]
- Vasileva, I.; Denkova, R.; Chochkov, R.; Teneva, D.; Denkova, Z.; Dessev, T.; Denev, P.; Slavov, A. Effect of lavender (Lavandula angustifolia) and melissa (Melissa officinalis) waste on quality and shelf life of bread. Food Chem. 2018, 253, 13–21. [Google Scholar] [CrossRef] [PubMed]
- Adilah, A.N.; Jamilah, B.; Noranizan, M.A.; NurHanani, Z.A. Utilization of mango peel extracts on the biodegradable films for active packaging. Food Pack. Shelf Life 2018, 16, 1–7. [Google Scholar] [CrossRef]
- Hanani, Z.A.N.; Yee, F.C.; Nor-Khaizura, M.A.R. Effect of pomegranate (Punica granatum L.) peel powder on the antioxidant and antimicrobial properties of fish gelatin films as active packaging. Food Hydrocoll. 2019, 89, 253–259. [Google Scholar] [CrossRef]
- Souza, B.; Larroza Nunes, I.; Vargas Pereira, F.; Druzian, J. Desenvolvimento e avaliação da eficácia de filmes biodegradáveis de amido de mandioca com nanocelulose como reforço e com extrato de erva-mate como aditivo antioxidante. Ciência Rural 2012, 42, 2085–2091. [Google Scholar]
- Santana, M.; Machado, B.; Silva, T.; Nunes, I.; Druzian, J. Incorporação de urucum como aditivo antioxidante em embalagens biodegradáveis a base de quitosana. Ciencia Rural 2013, 43, 544–550. [Google Scholar] [CrossRef] [Green Version]
- Knapp, M.A.; dos Santos, D.F.; Pilatti-Riccio, D.; Deon, V.G.; dos Santos, G.H.F.; Pinto, V.Z. Yerba mate extract in active starch films: Mechanical and antioxidant properties. J. Food Process. Preserv. 2019, 43, e13897. [Google Scholar] [CrossRef]
- Díaz Vela, J.; Totosaus, A.; Pérez Chabela, M.L. Integration of agroindustrial co-products as functional food ingrdents: Cactus pear (Opuntia ficus indica) flour and pineapple. J. Food Process. Perserv. 2015, 39, 2630–2638. [Google Scholar]
- Serna-Cock, L.; García-Gonzales, E.; Torres-León, C. Agro-industrial potential of the mango peel based on its nutritional and functional properties. Food Rev. Int. 2016, 32, 346–376. [Google Scholar] [CrossRef]
- Corbo, M.R.; Bevilacqua, A.; Petruzzi, L.; Casanova, F.P.; Sinigaglia, M. Functional beverages: The emerging side of functional foods commercial trends, research, and health implications. Compr. Rev. Food Sci. Food Saf. 2014, 13, 1192–1206. [Google Scholar] [CrossRef]
- Battino, M.; Beekwilder, J.; Denoyes-Rothan, B.; Laimer, M.; McDougall, G.J.; Mezzetti, B. Bioactive compounds in berries relevant to human health. Nutr. Rev. 2009, 67, S145–S150. [Google Scholar] [CrossRef] [PubMed]
- Shahidi, F.; De Camargo, A.C. Tocopherols and tocotrienols in common and emerging dietary sources: Occurrence, applications, and health benefits. Int. J. Mol. Sci. 2016, 17, 1745. [Google Scholar] [CrossRef] [PubMed]
- Lee, E.; Ryu, G.R.; Ko, S.H.; Ahn, Y.B.; Yoon, K.H.; Ha, H.; Song, K.H. Antioxidant treatment may protect pancreatic beta cells through the attenuation of islet fibrosis in an animal model of type 2 diabetes. Biochem. Biophys. Res. Commun. 2011, 414, 397–402. [Google Scholar] [CrossRef]
- Kim, T.; Yan, Q. Peroxisome-proliferator-activated receptors regulate redox signaling in the cardiovascular system. World J. Cardiol. 2013, 5, 164–174. [Google Scholar] [CrossRef]
- Fang, F.; Kang, Z.; Wong, C. Vitamin E tocotrienols improve insulin sensitivity through activating peroxisome proliferator-activated receptors. Mol. Nutr. Food Res. 2010, 54, 345–352. [Google Scholar] [CrossRef]
- Bajaj, S.; Khan, A. Antioxidants and diabetes. Indian J. Endocrinol Metab. 2012, 16, S267–S271. [Google Scholar]
- Afrin, S.; Giampieri, F.; Gasparrini, M.; Forbes-Hernandez, T.Y.; Varela-López, A.; Quiles, J.L.; Mezzetti, B.; Battino, M. Chemopreventive and therapeutic effects of edible berries: A focus on colon cancer prevention and treatment. Molecules 2016, 21, 169. [Google Scholar] [CrossRef] [Green Version]
- Baby, B.; Antony, P.; Vijayan, R. Antioxidant and anticancer properties of berries. Crit. Rev. Food Sci. Nutr. 2017, 58, 2491–2507. [Google Scholar] [CrossRef]
- De Leonardis, A.; Macciola, V.; Di Rocco, A. Oxidative stabilization of cold-pressed sunflower oil using phenolic compound of the same seed. J. Sci. Food Agricult. 2003, 83, 523–528. [Google Scholar] [CrossRef]
- Ciannamea, E.M.; Stefani, P.M.; Ruseckaite, R.A. Properties and antioxidant activity of soy protein concentrate films incorporated with red grape extract processed by casting and compression molding. LWT Food Sci. Technol. 2016, 74, 353–362. [Google Scholar] [CrossRef]
- Muíño, I.; Díaz, M.T.; Apeleo, E.; Pérez-Santaescolástica, C.; Rivas-Cañedo, A.; Pérez, C.; Cañeque, V.; Lauzuric, S.; de la Fuente, J. Valorisation of an extract from olive oil waste as a natural antioxidant for reducing meat waste resulting from oxidative processes. J. Clean. Prod. 2017, 140, 924–932. [Google Scholar] [CrossRef]
- Araújo, M.; Pimentel, F.B.; Alvesa, R.C.; Beatriz, M.; Oliveira, P.P. Phenolic compounds from olive mill wastes: Health effects, analytical approach and application as food antioxidants. Trends Food Sci. Technol. 2015, 45, 200–211. [Google Scholar] [CrossRef]
- Vu, H.T.; Scarlett, C.J.; Vuong, Q.V. Phenolic compounds within banana peel and their potential uses: A review. J. Funct. Foods 2018, 40, 238–248. [Google Scholar] [CrossRef]
- Shetty, S.B.; Mahin-Syed-Ismail, P.; Varghese, S.; Thomas-George, B.; Kandathil Thajuraj, P.; Baby, D. Antimicrobial effects of Citrus sinensis peel extracts against dental caries bacteria: An in vitro study. J. Clin. Exp. Dent. 2016, 8, 70–77. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Apraj, V.D.; Pandita, N.S. Evaluation of skin antiaging potential of Citrus reticulata blanco peel. Pharmacog. Res. 2016, 8, 160–168. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Choi, H.S. Lipolytic effects of citrus peel oils and their components. J. Sci. Food Agric. 2006, 54, 3254–3258. [Google Scholar] [CrossRef]
- Lateef, A.; Azeez, M.A.; Asafa, T.B.; Yekeen, T.A.; Akinboro, A.; Oladipo, I.C.; Azeez, L.; Ojo, S.A.; Gueguim-Kana, E.B.; Beukes, L.S. Cocoa pod husk extract-mediated biosynthesis of silver nanoparticles: Its antimicrobial, antioxidant and larvicidal activities. J. Nanostruct. Chem. 2016, 6, 159–169. [Google Scholar] [CrossRef] [Green Version]
- De Souza de Azevedo, P.O.; Aliakbarian, B.; Casazza, A.A.; LeBlanc, J.G.; Perego, P.; de Souza Oliveira, R.P. Production of fermented skim milk supplemented with different grape pomace extracts: Effect on viability and acidification performance of probiotic cultures. PharmaNutrition 2018, 6, 64–68. [Google Scholar] [CrossRef]
- Martins, N.; Ferreira, I.C.F.R. Wastes and by-products: Upcoming sources of carotenoids for biotechnological purposes and health-related applications. Trends Food Sci. Technol. 2017, 62, 33–48. [Google Scholar] [CrossRef]
- Kushwaha, R.; Kumar, V.; Vyas, G.; Kaur, J. Optimization of different variable for eco-friendly extraction of betalains and phytochemicals from beetroot pomace. Waste Biomass Valoriz. 2018, 9, 1485–1494. [Google Scholar] [CrossRef]
- Sharma, K.D.; Karki, S.; Thakur, N.S.; Attri, S. Chemical composition, functional properties and processing of carrot-A review. J. Food Sci. Technol. 2012, 49, 22–32. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kallel, F.; Ellouz Chaabouni, S. Perspective of garlic processing wastes as low-cost substrates for production of high-added value products: A review. Environ. Progr. Sustain. Energy 2017, 36, 1765–1777. [Google Scholar] [CrossRef]
- Larrosa, M.; Llorach, R.; Espın, J.C.; Tomas-Barberan, F.A. Increase of antioxidant activity of tomato juice upon functionalisation with vegetable byproduct extracts. LWT Food Sci. Technol. 2002, 35, 532–542. [Google Scholar] [CrossRef]
- Amofa-Diatuo, T.; Anang, D.M.; Barba, F.J.; Tiwari, B.K. Development of new apple beverages rich in isothiocyanates by using extracts obtained from ultrasound treated cauliflower by-products: Evaluation of physical properties and consumer acceptance. J. Food Compos. Anal. 2017, 61, 73–81. [Google Scholar] [CrossRef]
- Li, N.; Zhang, Q.; Song, J. Toxicological evaluation of fucoidan extracted from Laminaria japonica in Wistar rats. Food Chem. Toxicol. 2005, 43, 421–426. [Google Scholar] [CrossRef]
- Mayer, M.A.; Finlayson, G.; Fischman, D.; de Paz, C.; Telleriarte, M.R.; Ferrero, A.J.; Bobillo, C.; Fernández, B.E. Evaluation of the satiating properties of a nutraceutical product containing Garcinia cambogia and Ascophyllum nodosum extracts in healthy volunteers. Food Funct. 2014, 5, 773–779. [Google Scholar] [CrossRef]
- Veeruraj, A.; Liu, L.; Zheng, J.; Wu, J.; Arumugam, M. Evaluation of astaxanthin incorporated collagen film developed from the outer skin waste of squid Doryteuthis singhalensis for wound healing and tissue regenerative applications. Mat. Sci. Eng. C 2019, 95, 29–42. [Google Scholar] [CrossRef]
- Xu, J.; Wei, R.; Jia, Z.; Song, R. Characteristics and bioactive functions of chitosan/gelatin-based film incorporated with ε-polylysine and astaxanthin extracts derived from by-products of shrimp (Litopenaeus vannamei). Food Hydrocoll. 2019, 100, 105436. [Google Scholar] [CrossRef]
- Pattanaik, S.S.; Sawant, P.B.; Xavier, K.A.M.; Dube, K.; Srivastava, P.P.; Dhanabalan, V.; Chadha, N.K. Characterization of carotenoprotein from different shrimp shell waste for possible use as supplementary nutritive feed ingredient in animal diets. Aquaculture 2019, 515, 734594. [Google Scholar] [CrossRef]
Sample Availability: Samples are not available from the authors. |

| Compound Group | Source | Extracted Compounds | Ref. |
|---|---|---|---|
| Phenolic compounds | Apple seeds | Phloridzin, ellagic acid, caffeic acid, ferrulic acid, protocatechuic acid, gallic acid | [12] |
| Phenolic compounds | Avocado seeds | Procyanidin B2, epicatechin, rans-5-O-caffeoyl-d-quinic acid, procyanidin B1, catechin | [13] |
| Phenolic compounds | Rapeseed cake | Sinapine, sinapic acid and canolol | [14] |
| Phenolic compounds | Citrus peel | Total phenolic content | [15] |
| Phenolic compounds | Coconut shell | Total phenolic content | [16] |
| Phenolic compounds | Grape marc, Orange peel, Strawberry, Citrus pulp Camelina cake | Total phenolic content | [17] |
| Phenolic compounds | Black currant Sea buckthorn | Delphinidin 3-O-rutinoside, delphinidin 3-O-glucoside, cyanidin 3-O-rutinoside, cyaniding-3-O-glucoside, ellagitannins, proanthocyanidins, p-coumaric acid, caffeic acid-hexosides, coumaroylquinic acid-hexosides, vanillic acid-hexoside, (+)-Catechin, (−)-epicatechin, Quercetin 3-O-rutinoside, 3-O-glucoside, and 3-O-(6′′-malonyl)-glucoside | [18] |
| Phenolic acids, flavonoids | Grape skin | Gallic acid, caffeic acid, epicatechin, p-coumaric acid, rutin, catechin gallate | [19] |
| Flavonols | Pistachio hulls | Gallic acid, penta-O-galloyl-β-d-glucose, anacardic acid | [20] |
| Flavonoids, carotenoids | Passion fruit peel | β-carotene, provitamin A, quercetin, lycopene | [21] |
| Carotene | Carrot pomace | α- and β-carotene | [22] |
| Lycopene | Tomato peel | Lycopene | [23] |
| Non phenolic compounds | Lettuce | Ascorbic acid | [24] |
| Non phenolic compounds | Sugarcane molasses | Pullulan | [25] |
| Non phenolic compounds | Rice bran oil | Tocopherol | [26] |
| Waste | Extraction Method | Optimized Extraction Conditions | Obtained Compounds | Antioxidant Assay | Ref. |
|---|---|---|---|---|---|
| Flaxseed hulls | PEF | Electrode area (cm2)—95 pulse length (µs)—10 Temperature (°C)—20 Electric field (kV/cm)—20 | Tocopherols, polyphenols, phytosterols | - | [62] |
| Palm pressed fibers | PLE, Sx, Pc | Temperature (°C)—35, 35, 78.4 Flow rate (g/min) 2.4 Pressure (Mpa)—0.1; 0.1; 4 | Carotenoids | - | [79] |
| Palm pressed fiber | PLE | Solvents: CO2 and compressed liquefied petroleum gas Temperature (°C)—60 Pressure (MPa)—25.0 | β-sitosterol, α-tocopherol, squalene | HPX/XOD | [80] |
| Palm pressed fiber | UAE | Ultrasound intensity (W.cm−2)—120 Pulse cycle 0.4 Temperature (°C)—20 | β-sitosterol, α-tocopherol, squalene, | DPPH ABTS | [81] |
| Olive leaves | ASE | Temperature (°C)—190 Leaf moisture content (%)—5 Aqueous ethanol concentration (%)—80 | Oleuropein, Luteolin-7-O-glucoside | DPPH | [85] |
| Olive tree pruning biomass Olive mill leaves | UAE | Power (W)—400 Frequency (kHz)—24. Liquid/solid ratio of extraction (v/w)—20 mL/g. | Phenolic compounds Flavonoids | DPPH, ABTS, FRAP | [86] |
| Olive pomace | UAE, MAE, Se | Ethanol concentration (%)—90, Temperature (°C)—50, Time (min)—5 Liquid /solid ratio (mL/g)—30 Ultrasound intensity (W/cm2)—135.6 Ultrasound frequency (kHz)—60 | Hydroxytyrosol, maslinic acid, oleanolic acid | - | [87] |
| Olive leaves and tree bark | SCe | Temperature (°C)—60, Pressure (bar)—300 | α-tocopherol, squalene | - | [88] |
| Olive waste | UAEH | Cellulase, pectinase Frequency (kHz)—40 Power (W)—200 | Phenolic compounds | DPPH, ABTS, FRAP | [89] |
| Sunflower leaves | PLE, ESE | CO2 and mixture of solvents (ethanol in water from 0 to 100%) Pressure (bar)—400 Temperature (°C)—55 | Diterpenoids, flavonoids | - | [92] |
| Rapeseed press-cake | HVED | High voltage pulsed power (kV)—40 Intensity (kA)—10 Needle diameter (mm)—10 | Protein, polyphenols and isothiocyanates | TEAC | [95] |
| Pumpkin seeds | UAE MAE | Frequency (GHz)—2.45 Ethanol concentration (%)—60 Time (min)—20 UAE-EtOH—60% UAE-hex/EtOH/ H2O—30:49:21% | Phenolic compounds | DPPH | [97] |
| Waste | Extraction Method | Optimized Extraction Conditions | Obtained Compounds | Antioxidant Assay | Ref. |
|---|---|---|---|---|---|
| Apple pomace | Cec | Methanol, ethanol and ethyl acetate | Phenolic compounds and triterpenic acids | DPPH, FRAP, ABTS | [113] |
| Apple pomace | MAE | Solvent—70% acetone and 60% ethanol, Microwave power (W)—735, Solvent volume to sample ratio (mL/g)—5.65 Time (s)—149 | Phenolic compounds | DPPH | [114] |
| Mango peels | ScE | Pressure (MPa)—25.0 Temperature (°C)—60 Solvent—15% w/w ethanol | Carotene | - | [115] |
| Orange peel | LSE | Solvent: cyclopentyl methyl ether, ethyl lactate, isopropyl alcohol, polyethylene glycol 300, isopropyl acetate, dimethyl carbonate, methyl ethyl ketone, 2-methyl-tetrahydrofuran and ethyl acetate Temperature (°C)—70 Time (min)—150 Solid -liquid ratio—1:10 | Limonene | - | [116] |
| Cocoa shells | UAE, HC | Hexane, hydro-alcoholic solution (70:30 EtOH/H2O) ternary mixture (30:49:21 Hex/EtOH/H2O) cycle number 47.1, cycle time (s)—5 residence time (s)—5 total residence time (min)—3.93 | Catechins epicatechins theobromine caffeine | DPPH | [119] |
| Tomato seeds | UAE | Power (W)—90 hexane-acetone-ethanol 2-1-1 | Lycopene | - | [121] |
| Tomato seeds | MAE, OT | Temperature (°C)—70 Time (min)—15 Solvent—70% ethanol | Rutin | - | [123] |
| Onion waste | SbWE(PT) | Temperature (°C)—145 Time (min)—15 intense pulsed light (V)—1200 Time (s)—60 | Quercitin | - | [124] |
| Pomegra-nate waste | UAE | Temperature (°C)—51.5; Amplitude level—58.8% Solvent—sunflower oil | Carotenoids | - | [127] |
| Waste | Extraction Method | Optimized Extraction Conditions | Obtained Compounds | Antioxidant Assay | Ref. |
|---|---|---|---|---|---|
| Squid muscle | SbWE | Temperature (°C)—250 for aminoacids; 160 for peptides | Amino acids Peptides | ABTS | [139] |
| Poultry wastes | SbWE | Temperature (K) 533 Reaction time (min)—28 H2SO4 concentration in reactant system 0.02%. | Amino acids | - | [140] |
| Waste chicken breast muscle | OT | Sets of high voltage short pulses and by low voltage long pulses Energy (J/g)—38.4 ± 1.2 | Proteins | DPPH ABTS | [142] |
| Fucus vesiculosus | MAE | Pressure (psi)—120 Time (min)—1 1g alga/25mL water | Fucoidan | - | [149] |
| Saccharina japonica Aresch | MAE | Solvent: 55% ethanol Irradiation power (W)—400 solid/solvent ratio 1:8; Time (min)—25 | Phlorotannins | - | [152] |
| Undaria pinnatifida and Sargassum fusiforme | MAE coupled with HSCCC | Solvent: ethanolic KOH solution (1.5 mol/L) Irradiation power (W)—500 Liquid/solid ratio 20:1 Time (min)—20 Revolution speed (RPM)—800 | Fucosterol, 24-methylenecholesterol, phytol | - | [153] |
| Porphyridium cruentum | UAE | Solvent: 2mL of ethanol, 10mg ascorbic acid, 3mL of n-hexane, Time (min)—20 | Tocopherol | - | [154] |
| Nannochlorops sp. | PEF | The electric field (kV/cm)—20 Consecutive pulses 1–400 | Carotenoids | - | [155] |
| Waste | Active Compounds | Application | Ref. |
|---|---|---|---|
| Applications of antioxidant compounds recovered from edible oil industry wastes | |||
| Palm pressed fiber | β-Sitosterol, α-tocopherol, squalene | Cosmetic formulation with high sun protection factor | [81] |
| Sunflower leaves | Diterpenoids, flavonoids | Natural herbicide | [92] |
| Sunflower seed | Phenolic compounds | Antioxidant additive for sunflower oil | [192] |
| Soy bean waste | Proteins | Biopackaging | [193] |
| Olive waste extract | Phenolic compounds | Food industry (increasing shelf life of meat) | [194] |
| Olive mill wastes | Phenolic compounds | Food antioxidants | [195] |
| Applications of antioxidant compounds from fruits wastes | |||
| Apple seeds | Phenolic compounds | Food industry | [12] |
| Berries | Phenolic compounds | Pharmaceutical formulations | [191] |
| Mango peels | Carotene | Antioxidant additive for edible oil | [115] |
| Banana peels | Caffeic acid | Cosmetic formulations | [196] |
| Citrus peels | Phenolic compounds, essential oils and flavonoids | Pharmaceutical formulations | [197] |
| Citrus wastes | Phenolics and flavonoids | Cosmetic formulations | [198] |
| Citrus peels | Terpinene, cymene | Pharmaceutical formulations | [199] |
| Cocoa | Total extract | Larvicidal nanoparticles | [200] |
| Grape pomace | Phenolic compounds | Food industry | [201] |
| Applications of antioxidant from vegetable wastes | |||
| Tomato wastes | Lycopene | Health related applications | [202] |
| Beetroot pomace | Betalains | Medicinal and food applications | [203] |
| Carrot pomace | Carotenoids | Pharmaceutical formulations | [204] |
| Garlic waste | Ethanolic extract | Food additive to increase products shelf life | [205] |
| Onion waste | Phenolic compounds | Food industry | [206] |
| Cauliflower by-products | Isothiocyanates | Food industry | [207] |
| Applications of antioxidant compounds from other industries | |||
| Meat industry wastes | Gelatin Heparin | Pharmaceutical formulations (antioxidant and antihypertensive) | [1] |
| Algal biomass | Sulfated polysaccharides | Pharmaceutical formulations | [208] |
| Algal biomass | α-Carnitine | Nutraceutical products | [209] |
| Squid waste | Astaxanthin | Pharmaceutical industry | [210] |
| Shrimps shells | Astaxanthin | Food packaging material | [211] |
| Shrimps shells | Carotenoprotein | Supplementary nutritive feed | [212] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fierascu, R.C.; Fierascu, I.; Avramescu, S.M.; Sieniawska, E. Recovery of Natural Antioxidants from Agro-Industrial Side Streams through Advanced Extraction Techniques. Molecules 2019, 24, 4212. https://doi.org/10.3390/molecules24234212
Fierascu RC, Fierascu I, Avramescu SM, Sieniawska E. Recovery of Natural Antioxidants from Agro-Industrial Side Streams through Advanced Extraction Techniques. Molecules. 2019; 24(23):4212. https://doi.org/10.3390/molecules24234212
Chicago/Turabian StyleFierascu, Radu Claudiu, Irina Fierascu, Sorin Marius Avramescu, and Elwira Sieniawska. 2019. "Recovery of Natural Antioxidants from Agro-Industrial Side Streams through Advanced Extraction Techniques" Molecules 24, no. 23: 4212. https://doi.org/10.3390/molecules24234212