Personalize, participate, predict, and prevent: 4Ps in inflammatory bowel disease
- 1Department of Internal Medicine and Medical Therapeutics, University of Pavia, Pavia, Italy
- 2Department of Internal Medicine, San Matteo Hospital Foundation, Pavia, Italy
- 3Villa Stuart, Multi-Speciality Clinic, Rome, Italy
- 4Unit of Gastroenterology, Department of Systems Medicine, University of Rome "Tor Vergata", Rome, Italy
- 5Gastroenterology Unit, Department of Medicine, A.O.U.I. Policlinico G.B. Rossi and University of Verona, Verona, Italy
- 6CEMAD Digestive Disease Center, Fondazione Policlinico Universitario "A. Gemelli" IRCCS, Università Cattolica del Sacro Cuore, Rome, Italy
- 7Liver and Gastroenterology Unit, ASST Santi Paolo e Carlo, Milan, Italy
- 8Department of Health Sciences, University of Milan, Milan, Italy
- 9IBD Unit, Ospedale San Camillo-Forlanini, Rome, Italy
- 10Department of Gastroenterology, San Raffaele Hospital and Vita-Salute San Raffaele University, Milan, Italy
- 11Gastroenterology Unit, Department of Surgery, Oncology and Gastroenterology, University of Padua, Padua, Italy
- 12Gastroenterology and Endoscopy Unit, Fondazione Istituto di Ricovero e Cura a Carattere Scientifico Cà Granda, Ospedale Maggiore Policlinico and Università degli Studi di Milano, Milan, Italy
- 13Gastroenterology and Digestive Endoscopy Unit, ASST Fatebenefratelli Sacco, Milan, Italy
- 14Department of Medical Science and Public Health, University of Cagliari, Cagliari, Italy
- 15Gastroenterology Unit, Azienda Ospedaliero-Universitaria (AOU) di Cagliari, Cagliari, Italy
- 16Department of Pathophysiology and Transplantation, Fondazione IRCCS Ca' Granda Ospedale Maggiore Policlinico, University of Milan, Milano, Italy
- 17Inflammatory Bowel Disease Unit, Azienda Ospedaliera Ospedali Riuniti "Villa Sofia-Cervello" Palermo, Palermo, Italy
- 18Division of General and HBP Surgery, Rho Memorial Hospital, ASST Rhodense, Rho, Milano, Italy
- 19Gastroenterology Operative Unit, Azienda Ospedaliera San Camillo-Forlanini FR, Rome, Italy
- 20Division of Gastroenterology, Ospedale Ordine Mauriziano di Torino, Turin, Italy
Inflammatory bowel disease (IBD), which includes Crohn’s disease (CD) and ulcerative colitis (UC), is a complex, immune-mediated, disorder which leads to several gastrointestinal and systemic manifestations determining a poor quality of life, disability, and other negative health outcomes. Our knowledge of this condition has greatly improved over the last few decades, and a comprehensive management should take into account both biological (i.e., disease-related, patient-related) and non-biological (i.e., socioeconomic, cultural, environmental, behavioral) factors which contribute to the disease phenotype. From this point of view, the so called 4P medicine framework, including personalization, prediction, prevention, and participation could be useful for tailoring ad hoc interventions in IBD patients. In this review, we discuss the cutting-edge issues regarding personalization in special settings (i.e., pregnancy, oncology, infectious diseases), patient participation (i.e., how to communicate, disability, tackling stigma and resilience, quality of care), disease prediction (i.e., faecal markers, response to treatments), and prevention (i.e., dysplasia through endoscopy, infections through vaccinations, and post-surgical recurrence). Finally, we provide an outlook discussing the unmet needs for implementing this conceptual framework in clinical practice.
Introduction
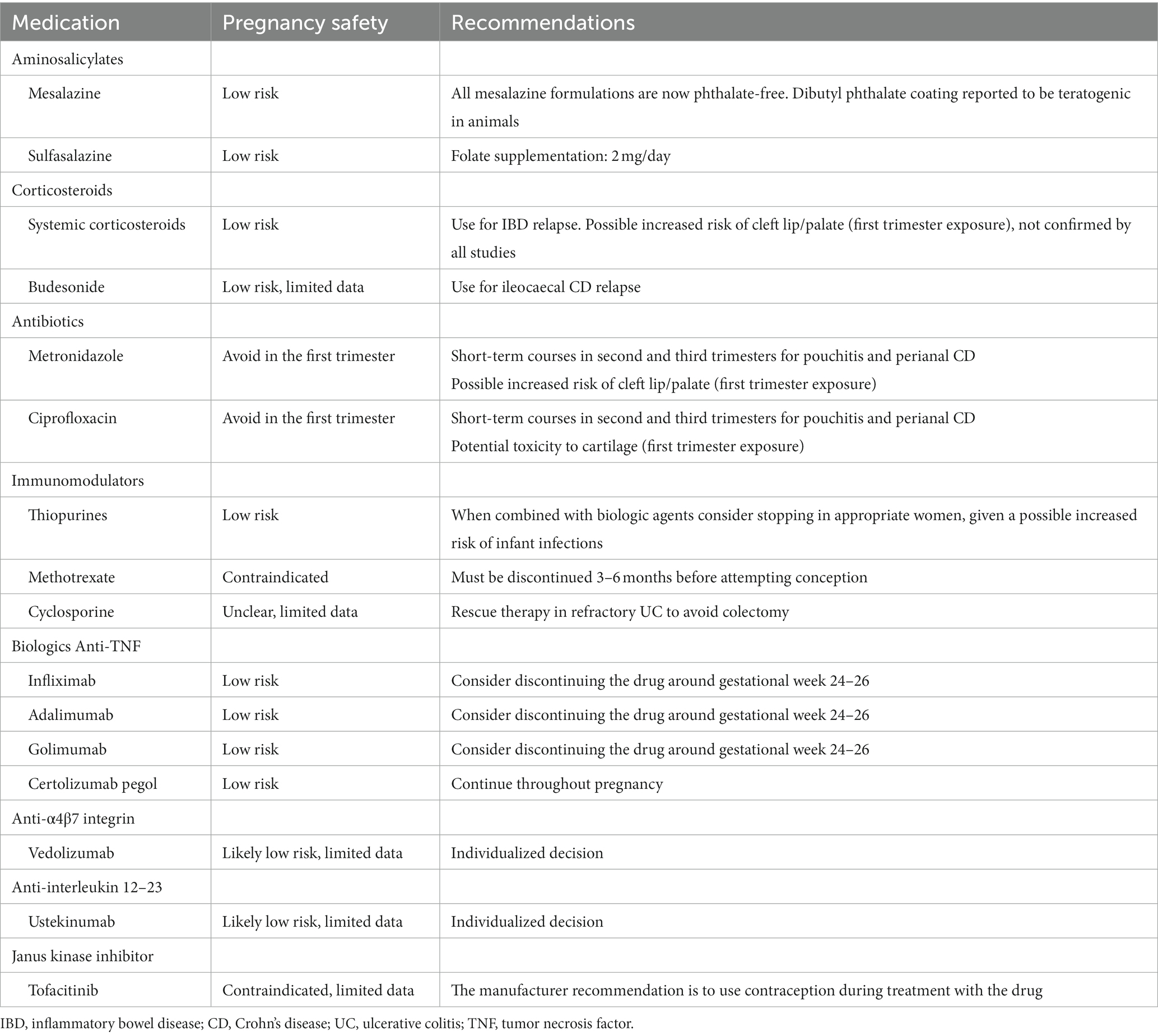
Inflammatory bowel disease (IBD) is an immune-mediated, lifelong, chronic, and disabling condition (1–4), which is constantly increasing worldwide (5), affecting individuals of any age and gender. IBD includes two main entities, namely ulcerative colitis (UC) and Crohn’s disease (CD), both causing intestinal (e.g., abdominal pain, diarrhoea and/or constipation, rectal bleeding, perianal symptoms) and extraintestinal (e.g., fatigue, anaemia, micronutrient deficiencies, arthritis, fertility issues) manifestations, deeply affecting patients’ quality of life, and social and sexual relationships (1, 2, 4). Over the last three decades, our knowledge about this condition has greatly improved, and the treatment paradigm has shifted from the use of unselective anti-inflammatory and immunosuppressive agents (e.g., mesalamine, corticosteroids, methotrexate, azathioprine, 6-mercaptopurine) to the use of more selective, pathogenesis-oriented, therapies (e.g., biological agents, small molecules) (6). Given all the proteiform and multifaceted aspects that should be taken into account, namely patient-, disease-, and drug-related factors (Figure 1), IBD can be considered as a clear example of a clinically complex disease (7), in which both biological and non-biological factors play a role in determining patients’ outcomes.
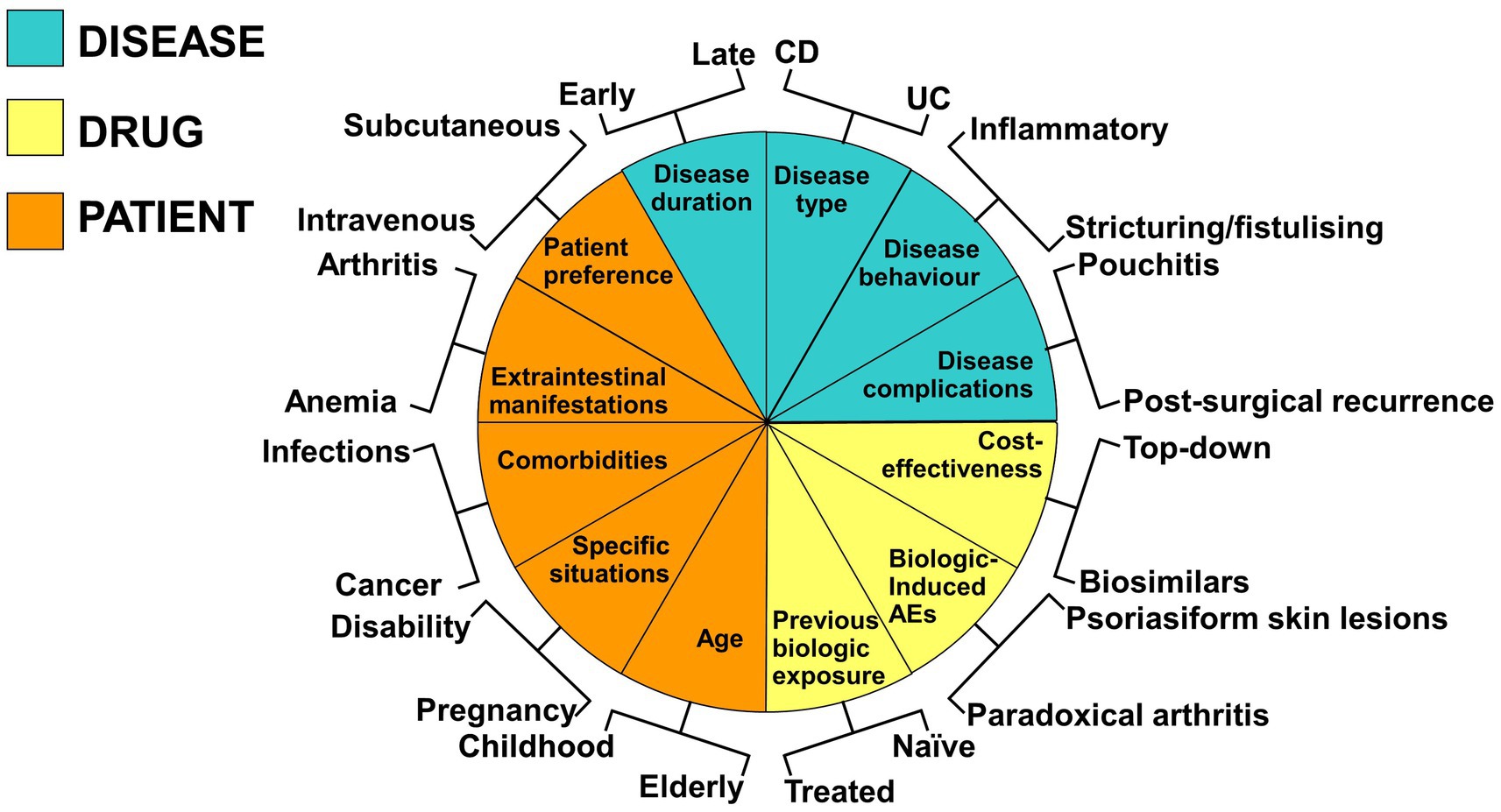
Figure 1. Schematic representation of disease-, drug-, and patient-related factors to be considered in the overall management of inflammatory bowel disease (IBD). The factors depicted in the Figure encompasses the four main domains of the 4P medicine, including prediction, prevention, participation, and personalization. Current guidelines do not comprehensively cover these aspects, that all pertain to an internal medicine setting. Indeed, many other variables not depicted in the Figure should also be considered and discussed with patients.
For these reasons, IBD should be considered as a systemic disorder, rather than as a specialty-oriented condition. In fact, although authoritative guidelines are available (8–11), they do not comprehensively consider all the possible aspects that may influence decision-making and patients’ outcomes, such as specific age groups (e.g., childhood, age of transition, elderly), specific situations (e.g., pregnancy and lactation, poor medication adherence, socioeconomic deprivation), and comorbidities (e.g., cancer, autoimmunity, infectious diseases). From this point of view, the so called 4P medicine (12), which stands for predictive, preventive, personalized, and participatory, might be considered as a proper framework for tailoring interventions for IBD at a patient—rather than at a disease—level, depending on their specific needs. Three main drivers have been implicated in the emergence of the 4P medicine, including the expansion of systems biology and systems medicine, the digital revolution, and the consumer-driver healthcare and social networks (12). All of these have indeed fuelled much interest in more precise and personalized medicine.
In this narrative review, within the framework of the 4P medicine, we have chosen a series of the most controversial and the currently debated issues in IBD, with an expert-based approach. Specifically, we have chosen three main topics per each “P” included in the framework. Additionally, we have also commented on the unmet needs in the field of IBD, which could soon be implemented.
No new data were generated or analyzed in support of this paper.
Personalize
Tailoring treatment of IBD in pregnant women
IBD commonly affects most patients in their reproductive age. Women with IBD experience significant concerns when considering pregnancy, especially related to the potential risks of drugs on pregnancy and foetal development, which may lead to a poor adherence to the therapy (13). It is of paramount importance to achieve a stable disease remission prior to conception and maintain a quiescent disease throughout pregnancy (14). Therefore, patients must be aware that if the treatment is discontinued, the risk of maternal and foetal complications related to disease relapse outweighs the potential adverse effects of medications. Preconception counseling is recommended, as it can improve drug adherence and pregnancy outcomes (15).
Many studies have evaluated the impact of therapy on the course and outcome of pregnancy in IBD women. However, determining the safety of drugs in this context is challenging as several factors such as disease activity, concomitant therapies, comorbidities and other maternal aspects can confound the study results. Most drugs have a favorable safety profile for use in pregnant patients (Table 1). Aminosalicylates are considered safe, therefore guidelines recommend their continuation throughout pregnancy (16, 17). In women who have an IBD flare during pregnancy, corticosteroids may be employed (16–18). The PIANO registry, including 1,490 pregnant women with IBD, showed an association between corticosteroid use and preterm birth, intrauterine growth restriction and low birthweight (19). However, the risk might be associated with IBD activity rather than drug use. Budesonide may be preferred for the treatment of mild-to-moderate CD relapse (17). The antibiotics metronidazole and ciprofloxacin should be avoided during pregnancy, especially in the first trimester (16). An increased rate of preterm birth has been observed with use of thiopurines (4–6). However, more recent data reported no heightened risk of adverse pregnancy outcomes associated with these medications (20, 21). Therefore, therapy with thiopurines should be maintained throughout pregnancy because the benefits outweigh potential risks (17). Methotrexate is absolutely contraindicated in pregnancy, due to its teratogenic and abortifacient action (16–18). Evidence on the use of cyclosporine in IBD patients is limited to pregnant women with a severe UC refractory to treatment (22). An increased rate of prematurity and low birthweight has been observed.
Results of several studies suggest low risk for use of anti-tumor necrosis factor (TNF) agents during pregnancy (23). In the PIANO and TREAT registers no increase in congenital abnormalities or adverse pregnancy outcomes was found among women who received anti-TNFs compared to the unexposed IBD control group (21, 24). The anti-TNF drugs, aside from certolizumab pegol, cross the placenta in the second and, especially, in the third trimester. Discontinuing the therapy around gestational week 24–26 is recommended by European guidelines to reduce anti-TNF exposure of the foetus (16). Indeed, this is recommended for patients who are in stable and complete remission, otherwise this therapy can be continued. Some studies, including the PIANO registry and the European TEDDY study, showed that anti-TNF use throughout pregnancy was not associated with an increased rate of infantile infections or serious infections (21, 25). Stopping anti-TNF therapy before the third trimester is an individualized decision based on both IBD activity and the woman’s risk profile (18).
Available data on the use of vedolizumab in pregnancy is limited. A retrospective study of 24 pregnancies exposed to vedolizumab reported some maternal and neonatal complications (26). Different results arise from a prospective comparison study on 24 pregnancies in 21 women treated with vedolizumab, whose use appeared low risk (27). Likewise, no concerns were observed in the case–control European CONCEIVE study, or in the PREGNANCY-GETAID study (28, 29). Therefore, the use of vedolizumab during pregnancy seems to convey a low risk, however no firm conclusion can be drawn, and a personalized decision should be made.
The effects of ustekinumab in pregnant women with IBD were evaluated in 29 pregnancies in the PREGNACY-GETAID study, without negative signals on maternal or neonatal outcomes (30). This data was confirmed by the preliminary results from the DUMBO prospective registry (31). Thus, ustekinumab appears to be safe during pregnancy, however the experience is limited, and its use should be individualized.
Tofacitinib, at doses exceeding human dose, is teratogenic in animals (18). Limited reported outcomes of pregnancy and new-borns in women on tofacitinib appeared to be similar to those observed in the general population (31, 32). Nevertheless, at present, the use of tofacitinib during pregnancy is contraindicated.
Tailoring treatment of IBD in cancer patients
Cancer risk is increased in IBD patients with a history of cancer, regardless of IBD-related treatments, as in the general population. The risk of new or recurrent cancer in patients with a present or past history of cancer using immunomodulators is under investigation. Most of these studies include IBD patients with less invasive cancer types, associated with a low risk of recurrence and limited follow up, thus limiting this analysis. The frequent combined conventional immunomodulators and biologics also does not allow an appropriate assessment of the risk of new/recurrent cancer using each specific treatment in IBD. Moreover, the time interval between diagnosis or remission of cancer and immunomodulators use differ among studies. Finally, patients with less severe cancer or with a lower risk of recurrence has mostly been considered. Overall, a joint management with oncologists shared with patients and follow up tailored on a patient’s basis is required when considering immunomodulators in IBD patients with a history of cancer.
Thiopurines use in IBD has been associated with a higher risk of skin cancers, particularly non melanoma skin cancers and lymphoma, further increased by combined TNFα-antagonists (33, 34). A slightly increased risk of cervical abnormalities has also been suggested (33, 34). The few available evidences assessing the risk of new/recurrent cancer under thiopurines in IBD patients with previous cancer are encouraging (35), including a large meta-analysis in patients with a history of malignancy treated or untreated with thiopurines or other immunomodulators (36).Nevertheless, a careful evaluation of IBD patients with a history of cancer is required before using thiopurines and drug discontinuation at diagnosis of cancer currently appears the initial option. Cancer type, time interval from diagnosis of cancer, age and IBD severity should be considered, with decision made on a patients’ basis shared with oncologists and patients.
Evidence regarding the risk of new/recurrent cancer in IBD patients with present/prior cancer using methotrexate monotherapy is lacking and further studies are required. Not conclusive, but reassuring data derive using methotrexate in rheumatoid arthritis (36, 37).
TNF-α is a cytokine provided of several activities, including necrotic activity on cancer cells “in vitro.” Several large independent studies support that TNF-inhibitors do not increase the overall cancer risk in IBD (33, 38–42). However, a slight excess risk of skin cancers, particularly melanoma, has been reported together with an increased risk of lymphoma particularly using combined thiopurines (43, 44). Whether TNFα-antagonists increase the risk of new/recurrent cancer in IBD patients with a diagnosis of cancer is being investigated. This analysis is currently limited by the small samples size of patients with specific cancer types. Additional limitations include differences among studies in terms of cancer stage, concomitant treatments, length of follow up and time interval from diagnosis of cancer to TNF-antagonists use. Despite these limitations, available preliminary evidence (mostly referring to overall cancer or melanoma) does not support that blocking TNFα increases the risk of new/recurrent cancer in IBD patients with prior cancer (36, 45, 46). As for other cancers, indication for TNF-antagonists in IBD patients with a history of cancer requires a careful selection of patients in a multidisciplinary approach with oncologists.
Vedolizumab (anti-α4β7 integrin) and ustekinumab (anti-IL-21/IL-23) have recently been used in IBD. Although a longer follow up is required, evidences suggest that their use does not increase the overall cancer risk in IBD (47, 48).In patients with a present or prior history of cancer, one retrospective study reported that vedolizumab does not increase the risk of new/recurrent cancer (49). Comparable findings were observed using vedolizumab or ustekinumab in IBD patients with prior malignancy (50). Additional data and a longer follow up is required for assessing the risk of new/recurrent cancer using these more immunomodulators, and particularly tofacitinib (pan-JAK inhibitor) (51) in IBD patients with a history of cancer.
Tailoring treatment of IBD in patients with superimposed viral infections
The main culprits of superimposed viral infection in IBD are human cytomegalovirus (HCMV, a β-herpesvirus) and Epstein–Barr virus (EBV, a ɣ-herpesvirus) (52, 53) They display the unique ability of establishing a lifelong latency (54–56) and reactivating in cases of reduced host immunity giving rise to systemic or end-organ disease (57, 58). Latent infection is the condition where viral genome is integrated with the host genome, viral gene expression is limited, and no virus is produced. The shift from latency to replication largely depends on T-cell immune control and/or changes in the differentiation/activation state of cells harboring viral genome (57, 58). While isolated reactivation events occur uneventfully in the immunocompetent host, critical stress or immunosuppression may trigger the lytic phase of the viral life cycle (57, 59). In this case, the involvement of colonic mucosa in IBD patients (60, 61) makes challenging to distinguish between a relapse of the underlying disease and a superimposed viral colitis. Indeed, both the clinical picture and endoscopic features are similar and viral DNA in peripheral blood is detectable only in a minority of patients, while serology is unreliable. The measurement of mucosal viral load by quantitative real-time polymerase chain reaction allows not only to diagnose a superimposed viral colitis, but also to distinguish this condition from infection by applying the cut-off value of 1,000 copies/105 cells, as follows:
viral colitis for values >103 copies/105 cells,
reactivation of latent infection for values between 102 and 103 copies/105 cells,
latent infection for values <102 copies/105 cells,
systemic disease when detectable copies are found in peripheral blood together with viral colitis.
Indeed, both immunohistochemistry and in situ hybridization are poorly sensitive and even in case of positivity, they do not give any information about the state of virus replication (62). Moreover, a correct and timely therapeutic management depends on the stratification of patients accordingly to these value ranges (Figure 2) since in the case of viral colitis/systemic disease a quick tapering and discontinuation of steroid therapy is mandatory, since it favors viral replication (63), while it is only preferable in case of reactivation to avoid an overt viral colitis, and no modification is required in case of latent infection. As far as the underlying therapy with azathioprine or anti-tumor necrosis factor-α agents is regarded, their long-lasting therapeutic activity makes unnecessary their discontinuation, rather the ability of infliximab to reinduce T-cell apoptosis (64) may limit the activation of the viral lytic phase. By contrast, the effects of inhibitors of T-cell homing (such as anti-integrin monoclonal antibodies, modulators of sphingosine -phosphate receptor) are unpredictable since their use may hamper the number of effector memory T-cells that are largely responsible for the containment of virus-infected cells and virus replication (65). No data are available on the effects, if any, of small molecules (Janus kinase inhibitors) on EBV and HCMV reactivation in IBD patients. In addition, antiviral therapy with ganciclovir (5 mg/kg bid iv) for 4 weeks is recommended in those with HCMV disease (66), with monitoring of the mucosal viral load at the end of treatment, while no effective treatment is available for EBV disease. The use of the anti-CD20 monoclonal antibody rituximab (375 mg/m2 body surface iv weekly) (67) was tentatively applied, but the poorness of information does not allow to draw firm conclusion. The use of adoptive transfer of virus-specific cytotoxic T-cell clones (68) has great therapeutic potential, but it is limited to those centers with good manufacturing practice facility and approval by regulatory body is required. This strategy is even more important if considering that an impairment of EBV-specific T-cell immunity was shown in ulcerative colitis patients, mostly in those refractory to current therapy (69). Whether passive transfer of virus-specific antibodies or immunoglobulin preparations may help the immune system of IBD patients to recover the ability to silence viral replication deserves investigation. Finally, an increased risk of lymphoma has been found especially in young IBD males under thiopurines, where a role for primary EBV infection has been proposed (70). Therefore, it is mandatory to start this therapy only after positivity of EBV serology has been verified. In parallel, all those factors favoring viral reactivation, such as malnourishment, should be corrected.
Figure 2. Schematic representation of the management of patients with inflammatory bowel disease treated with immunosuppressants or biologics and developing a viral colitis sustained by human cytomegalovirus (HCMV) and/or Epstein–Barr virus (EBV). Depending on colitis refractoriness and the viral load, different scenarios are depicted. The assessment of the viral load is crucial, as HCMV and EBV may be normally found in very low concentrations in the gut mucosa and are considered innocent bystanders in this case.
Participate
How to communicate with IBD patients
An effective and adequate communication between patients and healthcare providers (HCP) constitutes a critical step in the management of IBD throughout the whole course of disease, indeed representing the foundation for a successful engagement in the decision-making process. As a matter of fact, playing an active role in the therapeutic strategies is ranked as “very important” by most of patients (71) and it is associated with a higher rate of satisfaction and adherence to therapy, as well as with improved clinical outcomes (72, 73). Education concerning the disease itself, its course, management, and prognosis starts at the time of diagnosis and requires a careful assessment of patients’ needs, concerns, and level of awareness by HCP (74). At this phase, several barriers could influence the quality of the communication, such as the diagnostic delay responsible for patients’ distrust in medicine and HCP, the difficult acceptance of a chronic disease and its potential impact on quality of life (75). A useful tool to assess patients’ concern and to plan specific interventions accordingly, is represented by the rating form of IBD patient concerns (RFIPC), a 25-item questionnaire, requiring an individual score for the level of concern about several topics (e.g., cancer, specific symptoms, drug safety) (76). A survey from University of Manitoba, enrolling newly diagnosed IBD patients, showed that about 25% of them were dissatisfied with the amount of information received at the time of diagnosis, especially on specific issues such as long-term prognosis, infertility risk and inheritance, therapeutic management of clinical symptoms and impact on daily living activities (77). Similar findings also emerged from the UC Narrative Global Surveys, including both 2,100 UC patients and 1,254 physicians, showing an overall satisfying doctor-patient relationship; nevertheless, it is worth pointing out that up to 72% of patients wished they had received more information and support at the time of the diagnosis. Moreover, specific themes, in particular those ones related to emotional domains, seem to represent a taboo for almost half of patients. Of note, sexual health and sexual dysfunction are among the most neglected topics during medical visits, for several generical barriers such as “lack of time” or “more urgent issue to discuss,” but also embarrassment perceived by both actors and the inadequate physicians’ competence on how to manage potential emerging problems (78, 79).
Among HCP, a key role for the establishment of an efficacious communication with patients is played by the IBD-nurses. In a study promoted by the Crohn’s and Colitis UK organization, 1,081 young adults IBD patients (mean age 23.3 years) were asked to respond to an online questionnaire about several issues related to their disease, including the quality of communication with HCP. Gastroenterologists were shown to be the main source of information, especially at the time of diagnosis. However, IBD-nurses usually represented the first contact during a flare, ranked highest for the frequency of contacts and for the mean level of comfort in communication, compared to gastroenterologists (p < 0.0001) (80).
Moreover, IBD-nurses often represent the gatekeeper and filter on lots of information derived from the patients’ online health information seeking (81).
A specific mention should be made in regard to the specific language adopted by physicians while communicating with patients about their health status and when addressing their specific concerns. In particular, the Food and Drug Administration presented some evidence-based practical advice for the communication of risk and benefits, including, among others: (1) avoiding excessive use of technical terms, which would increase the risk of misunderstanding and ineffective communication, (2) employing visual tools to ease the comprehension of particularly challenging issues, and (3) providing absolute risks – rather than relative risks – when discussing drug safety (82).
Disability in IBD
In IBD patients, the altered bowel functions and non-bowel related symptoms (83, 84) heavily impact on patients’ work, social and home life activities, leading to various degrees of disability.
According to the World Health Organization (WHO) disability is defined as: “…any restriction or lack of ability to perform an activity in the manner or within the range considered normal for a human being” (85).
Recent data demonstrate that up to 46.6% of IBD patients complain some form of disability (86); given its relevance, researchers and clinicians felt the need to standardize the definition and the tools for assessing disability. The IBD-disability index was developed in 2011 and includes different aspects of life, namely General health, Body functions, Body structures, Activities and participation, Environmental factors (87). Although, this index appeared a solid and reliable research tool, it is not administered with ease in daily practice. As such, in 2017, the most important items from this questionnaire were selected and entered in a new visual instrument, called IBD-Disk, a self-administered tool (88).
Indeed, disease clinical activity in both CD and UC significantly correlates with disability, CD having a greater impact than UC (86, 89). Disability also strongly correlates to surgeries, hospitalizations, corticosteroid or anti-TNF usage. The presence of extraintestinal manifestations, as well as anaemia, has a profound impact on patients’ quality of life and disability as well (89). Patients with ileal pouch anal anastomosis had greater disability compared to UC patients with medically managed disease; female sex and having a public, rather than private, health insurance were further detrimental factors (90, 91). Also, overweight/obesity, being a social worker or having the perception of the need for a psychotherapist were identified as markers for a higher disability (86).
IBD-related disability also has consequences on working life and productivity (92). A Spanish community-based study revealed that 32% of patients had an officially recognized disability degree, while 4.1% had a work-disability pension; again, markers of a severe and progressive disease were major drivers of disability (93). Eventually, disability causes work loss and absenteeism, but also presenteeism (94) and affects patients’ perception of their working life (95). Indeed, work-related disability does have a detrimental impact on patients’ productivity, careers and earnings, which is specifically relevant for the female sex (96).
To conclude, preventing disability should be one of our pivotal aims when managing IBD patients. Disease control and avoiding disease progression may be a proper starting point; still, disability is multifaceted and not fully reflected by disease activity parameters. A new challenge for the years to come is to incorporate disability scores in clinical practice and trials (97).
Stigmatization and resilience in IBD
Given the symptoms experienced by IBD patients and IBD-related treatments, it appears clear that stigmatization may constitute a major issue in this context (98–101). Stigmatization is defined as the identification of negative characteristics that distinguish a person as different and worthy of separation from their family, social, or working environments, leading to losing social status, discrimination, and isolation (102). The sigma “perceived” by patients over time may be “internalized,” thus leading to the belief that stereotypes about their condition are true and adapt their life to this new situation (4, 103). While stigmatization has been extensively investigated in patients living with chronic conditions having a high social impact, as in the case of mental illnesses (102), HIV infection (103), and cancer (104), only over the last decade has stigmatization drawn attention in the IBD field. Stigmatization can be quantified through different scales. Taft et al. (105) proposed the perceived stigma scale (PSS) which was, however, adapted from the original PSS in irritable bowel syndrome (106). The PSS-IBD is a self-administered scale comprising 10 statements which refer to either significant others or health care professionals. According to a recent review (4), roughly 30 papers have looked at stigmatization in IBD so far, highlighting some key areas of intervention, including the work/school environment, sexual life, social relationships, depression and anxiety. As stigmatization does not seem to depend on age, sex, and IBD clinical activity (98), other factors should be investigated for tackling this issue.
The concept of stigmatization may be related to that of resilience (4), which is the ability to adapt well in the face of adversity, tragedy, threats or significant sources of stress (107). Hence, resilience is the ability to cope positively and to “bounce back” despite an adverse event, and this characteristic has been studied in patients with different chronic illnesses (108–110), a higher resilience being generally correlated with better outcomes. Several scales have been proposed, the most used being the 25-item Connor-Davidson resilience scale (CD-RISC) (111), validated into more than 70 different languages. Despite the availability of effective interventions (112), resilience has been poorly addressed in IBD (4), and some results are contrasting. For example, in a recent Italian study (98), resilience was shown to be inversely related to disease activity in UC, but not in CD, while in a previous study from Korea (112), the clinical characteristics of UC did not affect resilience. Other factors that have been assessed in other studies are age, sex, disease characteristics, and social context, although with some limitations (4). The only study specifically looking at their mutual relationship found a significant—though modest—and inverse correlation between the two (98).
To conclude, stigmatization and resilience should be considered as important features in IBD patients. Future studies should investigate both their determinants and possible ad-hoc interventions for improving resilience and fighting stigmatization.
Quality of care standards in IBD
IBD is a spectrum of inflammatory chronic intestinal conditions which need a complex approach in regards of diagnosis, management and follow-up (113). Despite several guidelines (8, 11, 114, 115), the approach to IBD patients remains heterogeneous across countries and even in the same country, as it depends on several factors, such as access to healthcare system, reimbursement policies, expertise of healthcare professionals and knowledge of IBD by the general population. Therefore, a clear definition of standards of care (SoC), and objective outcome measures in IBD are needed.
The most recent version of a general definition of SoC “that which a minimally competent physician in the same field would do under similar circumstances” (116). Based on this definition, the European Crohn’s Colitis Organisation (ECCO) decided to approach and define quality SoC by a Consensus process among expert healthcare professionals who are involved in the IBD management. The first step was to make a systematic review of the literature to identify the main domains of SoC in IBD. The panel identified three main domains: structure, processes, and outcomes (117). These results served as a basis to set up a list of statements which should identify the essential and desirable characteristics of the structure of an IBD unit, processes for diagnosis, treatment, and follow-up of IBD patients, and outcome to measure the quality of care. The final list of statements was voted during a Consensus meeting held in Copenhagen in 2019 by a multidisciplinary panel of experts and by the ECCO National Representatives, and published as an ECCO position paper (118). Because of the wide heterogeneity across countries and local situations, this position paper was intended to be a starting point for further definition of SoC in IBD that could be adopted, adapted or revised at national and local level. The ECCO is now planning a further step to harmonize SoC in Europe through the E-Quality project, which should identify gaps between the desired SoC and local reality in order to improve knowledge and remove barriers for a high-quality service for IBD patients (119).
Predict
Predicting relapse through faecal markers in IBD
IBD is characterized by dysregulated activity of gut-associated lymphoid tissue, leading to overly active intestinal inflammation. This, in turn, leads to chronic inflammatory symptoms, including diarrhoea and abdominal pain, and possible progression toward bowel wall fibrosis and penetrating complications, i.e., fistula and abscesses (120).
Similarly, to other autoimmune conditions, despite maintenance treatment, IBD follow a relapsing–remitting course, as inflammatory flares are interspersed with periods of symptomatic remission. The possibility to predict in advance disease exacerbations would greatly help in the management of IBD patients, as anti-inflammatory therapy escalation before symptomatic flares could prevent their occurrence and potentially positively impact the course of the disease (120). Traditionally, endoscopy has been considered the technique of choice to achieve this goal, as endoscopic activity has been associated with both short and long-term clinical outcomes in UC and CD patients (121–123). Endoscopy, however, has several limitations, including costs, availability, and acceptability by patients, which prevent a more widespread use of this technique for the purpose of relapse prediction. Hence, several efforts have been made over the last few years to discover and validate biomarkers able to predict inflammatory flares in asymptomatic patients, both in UC and CD patients. In the latter clinical condition, a significant proportion of studies focused on the identification of biomarkers predicting post-operative endoscopic and clinical relapse, in order to replace the predominant role of endoscopy in this clinical setting. Even if initial studies suggested a potential role for circulating inflammatory markers, including C reactive protein (CRP) levels, in predicting relapse in asymptomatic patients with CD (124), the current focus has been put on faecal biomarkers, owning to their high specificity and reduced invasiveness.
Faecal biomarkers used in IBD are direct products of inflammatory cells which infiltrate the intestinal mucosa during disease activity. Calprotectin, a protein which constitute 60% of the cytosolic protein in human neutrophils, is a calcium binding heterocomplex of S100A8 and S100A9, whose stool levels are proportional to the influx of neutrophils into the gut lumen, and therefore closely reflects mucosal inflammation (125). Other neutrophil-derived proteins, i.e., faecal lactoferrin and S100A12, are currently being studied as a potential alternative to faecal calprotectin in IBD patients, even if additional studies will be needed to determine their comparative values. Nevertheless, a large number of studies have been carried out demonstrating that levels of these biomarkers, and particularly those of faecal calprotectin, significantly correlate with the endoscopic and histological activity in both UC and CD patients (126).
More than 30 studies have so far evaluated the role of faecal calprotectin in predicting clinical relapse in patients with quiescent IBD (127–136). These studies widely differ in terms of population enrolled, faecal calprotectin cut-off value and method used to assess its levels, follow-up duration and definition of relapse. These limitations explain the extreme heterogeneity of results obtained, as sensitivity and specificity values for predicting relapse range from 28 to 100% and 43 to 52% in CD, and from 31 to 100% and 63 to 100% in UC patients. By considering data from six studies, a meta-analysis by Mao et al. calculated a pooled sensitivity and specificity of faecal calprotectin in predicting relapse in quiescent IBD to be 78 and 73%, respectively, in 672 adult IBD patients (318 UC and 354 CD) (137). Similar figures were observed regarding the role of faecal calprotectin in predicting post-operative recurrence. In the POCER study, applying a faecal calprotectin threshold of 100 μg/g, sensitivity and specificity of this test for detecting a meaningful endoscopic relapse (i.e., endoscopic Rutgeerts’ score ≥ 2) was 89 and 58%, respectively. Authors of the study calculated that applying an algorithm based upon calprotectin values at 6 months following intervention could allow 41% of patients to avoid colonoscopy (138). In a meta-analysis performed by Tham et al., when a faecal calprotectin threshold of 150 μg/g was used, a pooled sensitivity and specificity for of 70 and 69%, respectively, was calculated in the post-operative setting (139). Indeed, the calprotectin cut-off to be used must consider several variables. First, there are differences between CD and UC. For CD, a cut-off of 250 μg/g discriminates disease activity and predicts disease flare (140). For UC, different cut-offs are able to discriminate remission (<150 μg/g), mild (150–200 μg/g), or moderate/severe (>200 μg/g) disease. Finally, according to a recent expert opinion, a cut-off of <40 μg/g indicates deep IBD remission (140).
Predicting response and toxicity to thiopurines in IBD
Thiopurines, azathioprine, 6-mercaptopurine and thioguanine, have long been used for the medical treatment of IBD (141–143). They have been shown to be effective in maintaining remission in patients with IBD, effectively helping to reduce the rate of surgery in both CD and UC patients, the post-operative relapse rate in CD and the risk of colorectal cancer associated with IBD. Furthermore, when used in combined therapy, they improve the pharmacokinetics of anti-TNF monoclonal antibodies (144). However, with the continuous advent of new biological therapies with different mechanisms of action, the role of thiopurines is being questioned (145).
Up to 20% of patients will respond inadequately or develop toxicity to thiopurine (146), requiring treatment interruption or modification. The availability of predictors for thiopurine response and toxicity allows us to customize this treatment for patients with IBD.
The ability to predict who will respond to thiopurine therapy and to maximize the likelihood of responding earlier in the course of the disease would allow clinicians to tailor therapy earlier, with the goal of altering the natural history of the disease.
Obviously, pre-treatment predictors would be the ideal way to personalize thiopurine therapy in IBD patients.
Several clinical predictors [intestinal site, body mass index, disease activity, disease duration, early or late introduction of thiopurines, ethnicity, sex, etc.] were explored with conflicting results (147). Thiopurine metabolites, thiopurine-S-methyltransferase [TPMT] activity, TPMT polymorphisms and deletion of GSTM1, a polymorphism of the gene encoding glutathione-S-transferase, the enzyme responsible for converting azathioprine in 6-mercaptopurine, they have been studied as predictors of response to thiopurine (147). The use of thiopurine is undoubtedly hindered by the high incidence of adverse drug reactions affecting up to 25% of people who take them, leading to discontinuation of the drug in 17% of patients (148). Side effects often occur in the first few months. Consequently, the ability to predict which patients might develop these potentially serious side effects would be of great use in clinical practice. Toxicities addressed will include myelotoxicity, hepatotoxicity, pancreatitis, alopecia, gastrointestinal and flu-like symptoms and complications associated with Epstein–Barr virus. Thus, while monitoring of thiopurine metabolites shows utility in those already initiated with thiopurine, pharmacogenetic tests, which already play a significant role in preventing toxicity, show some promise in predicting response.
Predicting response to biologics in IBD
Since the introduction of TNF-alpha inhibitors in the IBD therapeutic armamentarium, other monoclonal antibodies targeting different inflammatory mechanisms have been developed. Namely, vedolizumab targeting alpha4-beta7 integrin involved in T cell intestinal homing and ustekinumab blocking p40, a subunit shared by both by interleukin (IL) 12 and 23 are now available for patients.
Despite the increasing number of therapies available, a high rate of primary failure is observed. Overall, about 30–40% of patients do not respond to TNF inhibitors and those in which an initial clinical benefit is reached tend to lose response and experience clinical relapse over time (149, 150). Similar results have been observed with vedolizumab (151, 152) and ustekinumab (153, 154). In this situation patients are at risk of multiple therapeutic failures with an increased risk to develop complications and permanent organ damage due to uncontrolled inflammation. For these reasons, the identification of predictive factors describing the most appropriate patient for a specific mode of action is currently concentrating large part of research efforts in the IBD field.
Moving from the concept “one drug fits all” to a more personalized approach to patients, different putative predictive factors have been considered that can be divided in different subgroups: patient-related factors, disease-related factors, biomarkers, immune-system-related factors, and microbiota composition. Most of the association studies aiming at identifying predictive factors have been conducted in patients treated with TNF inhibitors and only few have been explored to predict response to vedolizumab and ustekinumab.
Age, gender, and body mass index as predictive factors of response to infliximab reported contrasting results with some studies reporting an association while other failed to do so (reviewed in (155)). Though well defined as risk factor for disabling disease in Crohn’s disease (CD), the role of smoking in predicting response to anti-TNF agents is uncertain (156, 157).
As for disease characteristics, post-hoc analysis from anti-TNF agents registrative trials have found longer disease duration, fibro-stenosing behavior and ileal localization to be associated to worse outcome in anti-TNF-treated CD patients. Overall, the effect of these factors, although not systematically confirmed, might be reconducted to the progressive development of fibrotic ileal stenosis in long-standing CD, a condition that is unlikely to respond to any biologic therapy. Accordingly, the predictive role of these factors in UC is less defined (155).
Finally, biomarkers of inflammation such as CRP and faecal calprotectin have been associated in some studies to higher response rate to anti-TNF agents in CD (158–162) and UC (163–165). Whether more severe inflammation is required for anti-TNF agents to better exert their effect or CRP and faecal calprotectin merely support the presence of intestinal inflammation in patients otherwise affected by symptoms caused by other conditions (e.g., IBS) remains unclear.
While GWAS identified several gene variants associated with the increased risk to develop IBD and some of them were shown to be associated with specific disease phenotype and disease course severity, few have been linked to response to anti-TNF agents. Variants of genes involved in cell apoptosis have been associated to infliximab response while the low affinity immunoglobulin gamma Fc region receptor III-A (FCGR3A) and HLA-DQA1*05 were shown to be associated to antidrug antibody formation and secondary loss of response in CD (166, 167).
A few studies have also assessed the response to therapies in relation to anti-drug antibodies and trough levels, with conflicting results (168). In a previous study, the presence of anti-drug antibodies and low trough levels was predictive of early failure with biosimilar infliximab (169). In another, retrospective study, the baseline infliximab trough levels were not predictive of IBD relapse over the following 2 years (170). Hence, therapeutic drug monitoring for this purpose is uncertain.
In addition to the limited results obtained to identify patient predisposition to respond to biologics, genetic analysis does not consider the dynamic nature of the inflammatory process characterizing IBD. To identify factors that may influence response to therapy in a specific phase of IBD course, analysis of gene expression at mRNA and protein level in the peripheral blood and the intestinal mucosa have been performed. For instance, low expression levels of the triggering receptor expressed on myeloid cells (TREM-1) in the peripheral blood of IBD patients was predictive of primary non-response to anti-TNF therapy (171). However, in another independent CD cohort, downregulation of TREM-1 was predictive of mucosal healing (172). Similarly, serum and tissue expression of oncostatin M was also associated with primary non-response to infliximab and golimumab (173, 174). More recently the expression of membrane -bound TNF and alpha4-beta7 at mucosal level detected by fluorescent-adalimumab and vedolizumab in conjunction with confocal endomicroscopy was shown to predict response in small cohorts of patients treated with adalimumab and vedolizumab, respectively, (175, 176). Additionally, in a very recent study, prediction of response to biologics was assessed through a computerized image analysis of probe confocal laser endomicroscopy (pCLE) in vivo and by assessing the binding of fluorescent-labeled biologics ex vivo (177). The authors found an 85% and an 80% accuracy in predicting response to therapy in UC and CD, respectively. Additionally, a good prediction to response to anti-TNFs was noticed through a panel of expressed genes (AUC > 0.7).
Microbiota composition has been also explored as predictive factor of response to biologic therapy. Metagenomic analysis of stool samples from patients commencing vedolizumab therapy showed higher alpha-diversity and greater abundance of Roseburia inulinivorans and a Burkholderiales species in CD who achieved remission at week 14 (178). Interestingly, in this study the model incorporating metagenomic and clinical data performed better than the model considering metagenomic data alone.
Most of the aforementioned studies exploring potential predictive factors are biased by the limited number of patients analyzed and in most of the cases by the lack of a validation in a separate independent cohort of patients. The future will see the integration of datasets from different “omic” approaches, as in the case of the Endo-omic study (177), in addition to clinical characteristics in the attempt to define a reliable patient profile to predict response to biologics.
Prevent
Preventing flare-up and dysplasia through advanced endoscopy in IBD
Endoscopy is the gold standard for diagnosis, surveillance and assessment of luminal disease activity and extent in IBD patients (115). In recent years, several advanced endoscopic imaging and artificial intelligence techniques were introduced, allowing a detailed analysis of the endoscopic images for precise and reproducible quantification of early inflammatory and dysplastic changes at a microscopic and ultrastructural level. Currently, the main goal of advanced endoscopy in IBD is the assessment of deep mucosal healing looking beyond the standard definition of endoscopic remission for early prevention of disease flare-up and tight drug monitoring (179, 180).
In 2020, the Picasso study, a large, international, multicenter, prospective trial, demonstrated that virtual chromoendoscopy can predict valuable clinical outcomes with the similar accuracy provided by histology in UC patients (181, 182). The same study group is now developing two different operator-independent AI systems to detect deep endoscopic remission on virtual chromoendoscopy video-images (183) and histologic remission on digitalized histological slides (184) to predict the risk of a flare-up at 12 months, hereby enabling clinicians of novel diagnostic tools and strategies for precise patient-tailored management.
In 2021, Bossuyt et al. have described two different endoscopic prototypes for computer-aided assessment of mucosal inflammation in UC introducing the firsts operator-independent scoring systems in the field of IBD. Both prototypes can be activated in real time, do not require contrast agents and are supported by computer-aided algorithms (machine learning algorithm) that as opposed to other types of artificial intelligence (likewise convolutional neural network) must be fed structured data and are not able to cope with unforeseen circumstances. The red density score (185) provides a quantitative assessment of disease activity based on the redness map and vascular pattern recognition, which correlates well with both endoscopic and histopathological disease activity. The second artificial intelligence-powered endoscope provides a single short wave-length monochromatic LED light illumination enabling a magnified visualization of inflammatory changes on the pericryptal vessels (186). This artificial intelligence system predicts histologic remission with higher accuracy (86%) as compared with standard endoscopic scoring systems (74% with Mayo endoscopic sub score, 79% with UC endoscopic index of severity). Larger studies and validation in independent cohorts are on the way to confirm the preliminary results obtained with these colonoscopy prototypes.
In 2022, Maeda et al. have described a novel artificial intelligence system working on endocytoscopy images able to predict clinical relapse during 12 months of follow-up in UC patients in clinical remission from a single referral Japanese center. The endocytoscope was integrated into the distal tip of a standard colonoscope to collect ultra-magnified images (×520) in real time. Notably, irrespective of the operator expertise, this AI system predicts clinical relapse better than expert assessment of endoscopic remission (Mayo endoscopic score ≤ 1) and comparably than expert assessment of endoscopic deep remission (Mayo endoscopic score = 0) or histologic remission. Other studies either based on recorded colonoscopy videos (187–189) or prospectively collected fixed images (190) have produced confirmatory evidence on the promising role of AI in refining mucosa activity assessment (187–189) to predict prognosis in UC patients (190).
The diagnosis of colorectal dysplastic changes in patients with longstanding IBD colitis or primary sclerosing cholangitis is still an unmet issue (191). Small but clinically relevant non pedunculated colorectal lesions are often overlooked during surveillance colonoscopy due to their subtle or inflammatory-like appearance (192). In addition, the optical characterization of neoplasia is still challenging in IBD colitis, even using advanced endoscopic technology in expert hands (193). Within this context, the future goal of artificial intelligence is the detection and characterization of colitis-associated dysplasia at an early stage, revealing non advanced colorectal lesions when still suitable for curative endoscopic resection or for timely prophylactic surgery based on a patient-tailored multidisciplinary approach (194). The first artificial intelligence-assisted detection of colitis-associated neoplasia has been recently video-documented (195). However, the artificial intelligence systems successfully adopted for sporadic colorectal adenoma detection and characterization were not trained in active and post-inflammatory changes, and therefore need further developing in the setting of IBD.
Preventing infections in IBD
The treatment of IBD has been revolutionized in recent years by the increasing use of immunosuppressants, biologics and small molecules. Although these powerful drugs have proved effective in controlling disease activity, they can lead to a condition of immunosuppression, consequently increasing the risk of new infections or reactivation of latent infections (196). In this context, the evaluation of the immunological and serological status of patients with IBD is extremely useful in proposing adequate preventive measures against such infections, first of all vaccines (197).
In this sense, the guidelines of the Infectious Diseases Society of America distinguish patients with low- and high-level immunosuppression (198). This distinction is crucial in evaluating the advisability of immunizing patients using live vaccines.
According to various evidences, the main risk factors for opportunistic infections are advanced age (OR 3.0 for patients>50 years) (199) and the simultaneous use of multiple immunosuppressive drugs, since the transition from a single immunomodulator to combination therapy increases the relative risk of infections from 2.9 to 14.5 (200) Considering TNF-α inhibitors alone – which are in themselves sufficient to define high-level immunosuppression, as previously mentioned – the TREAT registry showed an increased risk of serious infections with these drugs (HR 1.47) (201). Regarding the risk of infections with new biological therapies approved in IBD, vedolizumab – a monoclonal gut-selective antibody that targets integrin α4β7 and ustekinumab a monoclonal antibody directed against the p40 subunit of IL-12 and IL-23 do not appear to be associated with important infectious complications (202, 203). Tofacitinib – an orally administered non-selective JAK inhibitor – is associated with an increased risk of herpes zoster infection in patients with IBD, with an observed incidence of 4 herpes zoster infections per 100 patient-years (204). In this regard, the availability of Shingrix®, a new recombinant non-live vaccine already approved for adults aged 50 and over against virus reactivation, could be of great help in patients who will be treated with Tofacitinib and future drugs of the same class (205).
All patients with IBD must be tested for the hepatitis C and B virus (HBsAg, anti-Hbs, anti-HBc) to assess the infectious status (current, past, inactive carrier) or vaccination status. In patients with evidence of active HBV infection, HBeAg, anti-HBe and HBV DNA must also be evaluated (206). Obviously, the HBV vaccine is recommended in all seronegative patients.
In patients who will be treated with TNF-α inhibitors, a careful preliminary assessment of the presence of latent tuberculosis is mandatory, since there is a high risk of reactivation of the infection with these drugs (207). If treatment with thiopurines is planned, screening should be performed to ascertain the serological status against EBV (208, 209).
Studies conducted in women with IBD demonstrated a high frequency of Human Papilloma Virus (HPV) associated Pap-smears abnormalities (210). In this setting, immunosuppressive drugs could increase the risk of persistent HPV infection and ultimately cervical cancer.
Infections caused by Neisseria meningitidis can evolve into a disease with high mortality, if not recognized and promptly treated. Meningococcal infections are endemic in Western countries, and the annual incidence of invasive meningococcal disease varies in multiyear cycles (211, 212).
The ongoing pandemic of COVID-19 caused by infection with SARS-CoV-2 is worthy of a brief comment in the context of vaccinations for patients with IBD. COVID-19 has raised substantial concerns for patients with IBD who are receiving immunosuppressive agents. Patients with IBD do not appear to be at greater risk of infection by SARS-CoV-2 compared to the general population, and current treatments are not associated with worse prognosis. However, clinicians should be cautious about the use of systemic steroids for treatment of COVID-19 (213).
Recently, a consensus on be-half of the International Organization for the Study of Inflammatory Bowel Disease provided several statements supporting SARS-CoV-2 vaccination in patients with IBD regardless the immune-modifying therapies (214). Generally, all IBD patients should be vaccinated for the following diseases: tetanus, diphtheria and polio, measles, mumps and rubella, HBV, influenza, varicella, HPV, pneumococcus, and meningococcus (215).
Physicians, and in particular gastroenterologists, should be encouraged to conduct a structured interview of all IBD patients at diagnosis, about the immunization status and the definition of a vaccination plan before the start of immunosuppressive treatment (215).
Preventing post-surgical recurrence in IBD
Only 10 years ago, the idea that a surgeon should be involved in the prevention of post-surgical recurrence of IBD would have been at least considered imaginative, but nowadays surgery has great an impact on the patients’ clinical history, including the disease recurrence.
In UC surgery is curative, even if in very selected cases, a segmental colectomy could be considered to avoid total proctocolectomy (216). However, the patient could experience post-surgical consequences such as incisional hernias, stenosing or fistulizing complications of the ileo-pouch-anal anastomosis, and adhesions potentially causing obstructions and reduced fertility in women. The standardization of restorative proctocolectomy procedure, taking into account all the problems emerged during the past years of surgical practice, and the new technologies available, have significantly reduced all the major drawbacks, in particular, the long-term pouch disfunction and failure, giving the patients the possibility to avoid definite ileostomy (217–219). The extended use of laparoscopy in referral centers has not only drastically reduced perioperative complications, length of hospitalization, and incisional hernias, but it left the women’s fertility preserved through a notable reduction in adhesions (219–222). It is worth pointing out that laparoscopy can be used to complete a restorative proctocolectomy even in case of staged procedures, with very low conversion rate (220).
Looking at the other side of the IBD-moon, in the “traditional” view of CD clinical history, surgeons were only involved when any pharmacological option had been failed and perforating, or stenotic complications had been occurred. Due to this gastroenterologists’ “last-resort” approach, the surgeons, for their part, have tried to focus on identifying all the risk factors for post-operative complications that they could have acted upon. Thanks to the evidence from surgical trials, performed at the turn of the 21st century, today we know that steroids and anti-TNF agents should be withdrawn before surgery, nutritional status checked and amended, deep venous thrombosis prevented, intra-abdominal abscesses radiologically drained, and acute obstruction managed conservatively, exploiting a “postpone and optimize” strategy (223). Furthermore, some risk factor for post-operative recurrence has been identified such as a genetic predisposition, a paediatric diagnosis, the need for surgery within 1 year of diagnosis, a smoking habit, a penetrating disease behavior in the terminal ileum and colon, a stricturing behavior associated with upper gastrointestinal location, the presence of perianal disease, and the bowel wall thickening and the endoscopic recurrence within 1 year after surgery (224–228). However, focusing on anastomotic post-surgical recurrence using the score proposed by Rutgeerts and colleagues, it became evident that different anastomotic configurations presented different long-term recurrence, with side-to-side anastomosis (either isoperistaltic or anisoperistaltic – also known as functional end-to-end) having the better results over all the other anastomotic configurations (end-to-side, end-to-end, and side-to-side with blind loops) (223, 229). Recently, a particular kind of functional end-to-end anastomosis, proposed by Kono et al., has been claimed to drastically reduce both post-operative complications and long term endoscopic and surgical recurrence, but a direct comparison with a stapled similar anastomosis, considering also postoperative adjuvant treatment, is still lacking (230, 231). Another argument of actual debate is the treatment of the mesentery during ileo-colic retention, since a group from Limerick supported a role of mesentery removal in preventing post-surgical recurrence. On the one hand, the well-known low recurrence rate observed on the site of a strictureplasty, a technique where both the diseased bowel and mesentery are preserved, is in stark contrast with this hypothesis (222, 224). On the other hand, a large retrospective study and a retrospective comparative analysis, both performed on prospective databases, failed to find any evidence and strengthened the importance of post-operative prophylaxis with biological treatment (231–234).
Unmet needs in IBD
Despite the recent developments in the diagnosis and monitoring of IBD (235–237), together with the new biologics and small molecules which expanded our therapeutic armamentarium for treating these diseases (238–240), several patients still suffer from disabling bowel symptoms and relevant disease complications (241, 242). Moreover, patients with IBD experience impairment of health-related quality of life, fatigue, depression, and anxiety (243, 244). Thus, many questions remain to improve the management of IBD.
Both CD and UC are heterogeneous diseases, with significant differences in terms of disease location, activity, onset and course in each patient. However, current treatment strategies suggest a standard approach to all patients, because of the limited evidence regarding the existence of useful and easy-to-use predictors of response to therapy (245). In the last decade, with the novel drugs available, various efforts have been made to fill this gap, by evaluating the individual and molecular specificities of the disease, identifying new biomarkers and stratifying individual patients’ risk factors in its early stages (246). Nevertheless, the incorporation of these predictors in routine clinical practice is not yet established.
IBDs are progressive disorders in which chronic inflammation may lead to disease progression, disability and complications (247–250). Various studies have shown that early use of biologic drugs is associated with improved clinical outcomes and, therefore, the accurate identification of patients who may benefit of early intervention seems to be crucial. Although several studies identified and suggested biochemical markers (e.g., faecal calprotectin, C-reactive protein, oncostatin M), clinical indices (i.e., early disease onset, smoking, use of steroids, extended disease, perianal involvement), genetic tests, proteomic, metabolomic or metagenomic and radiomics or imaging-based biomarkers, to identify higher-risk patients, prospective outcome data corroborating these variables are lacking. Thus, more objective and prospectively validated biomarkers are needed in order to implement a precision-medicine approach in IBD (246).
To date, registration trials demonstrated that current available treatments for both UC and CD do not achieve remission rates over 50%, underlining a therapeutic ceiling in the management of both diseases and potential challenges that need to be addressed (251). For this reason, a treat-to-target approach with tight monitoring of the disease during maintenance treatment has been investigated, emphasizing the potential to improve drug efficacy, but also highlighting the fact that patients still may present with complications in at least 30% of the cases in the mid-term (252). Moreover, treatment goals are challenging to achieve in clinical practice since they depend on many variables, including the disease duration, timing of treatment initiation, drugs available, compliance to treatment, and duration of therapy. Therefore, new and more effective therapies for IBD are needed. On the other hand, other questions arise from the use of current available treatments: which drug should be used as first-line treatment, which drug should be used after a failure, when to start or stop a drug, how to best adjust timing and dosing (i.e., the role of therapeutic drug monitoring). Moreover, should we combine biologics and small molecules to improve the efficacy without taking into consideration safety issues? Unfortunately, to date most of the comparative efficacy data can be retrieved from systematic reviews and network meta-analyses because head-to-head trials are in their early development (253, 254), whereas evidence regarding the combined use of drugs with different mechanisms of action is very limited and the risk-safety and cost–benefit analyses are still unsatisfactory (255).
There are other different clinical scenarios where there is a lack of evidence regarding the correct management of IBD. For instance, for the treatment of perianal fistulizing CD, infliximab represents the best-studied drug showing a long-term healing rate lower than 50%, while vedolizumab and ustekinumab have been only evaluated in retrospective studies and post-hoc analyses (256). Limited evidence is also available for the use of anti-JAK, mesenchymal stem cells, and combination of biological therapies in this setting. Likewise, the treatment of acute severe UC suffers from the same limitations mentioned above for perianal fistulizing CD, with a few treatment modalities assessed to date (257). Ulcerative proctitis (UP) represents another example of difficult-to-treat disease, since approximately half of these patients are refractory to conventional medical treatment and the efficacy data from registration biologic trials, where UP patients are systematically excluded, is limited (258). Prevention of post-operative recurrence is still a controversial field, although a recent systematic review and meta-analysis showed that anti-TNF therapies, alone or in combination, seem to be the best way for preventing endoscopic recurrence (259). However, which patients should be treated, the best timing to initiate biologic therapy in the post-operative setting, as well as the role of new biologic therapies, are all features that need to be investigated. Further, there is an urgent need to identify new therapeutic targets and new effective molecules for tackling intestinal fibrosis in CD. The uncertainties regarding the potential detrimental effects of such drugs for CD (i.e., perforation, worsening of intestinal inflammation) (3). In fact, there are no available therapies for this purpose, and, for this reason, the rate of surgery in CD is still high.
Also the frail patient, usually having an age over 65 years, with various comorbidities including the cardiovascular ones, represents a challenging clinical situation, since the high risk of developing significant adverse events (i.e., infections) leads frequently to adopt sub-optimal IBD therapy, as shown by chronic corticosteroid use and elevated surgical rates (260). In this type of patient, the use of gut-selective immunosuppressants seems to have the potential to reduce the risk of adverse events, but further studies are needed. Finally, other settings requiring further data and advancements are relative to the use of novel non-invasive markers for monitoring the disease, with a better correlation with mucosal inflammation and disease progression, and the improved control of fatigue together with anxiety and depression which are able to affect greatly the disease course and therefore should be considered in its management (261).
Conclusions and outlook
Due to its multifactorial pathogenesis, its clinical manifestations, and its long-life clinical impact, IBD represents a typical example of a complex disease (7), making its clinical management challenging for physicians. As we have discussed, age- and gender-specific issues, specific comorbidities, availability of different biologics and small molecules with different route of administrations, and socioeconomic and cultural factors, are just some of the variables that affect decision-making in IBD. Additionally, despite the availability of novel drugs, clinical and endoscopic remission rates on a long-term are still poor and proper drug positioning is uncertain. Hence, an integrated effort is required to meet the key objectives needed to pursue targeted treatment. The 4P framework seems to be the best currently available tool for clinicians that could be applied for tailoring interventions in day-by-day clinical practice in the IBD field. Table 2 summarises the main variables to be considered in the 4P medicine framework that could prove useful in the overall IBD management, along with a possible future trend per each area.
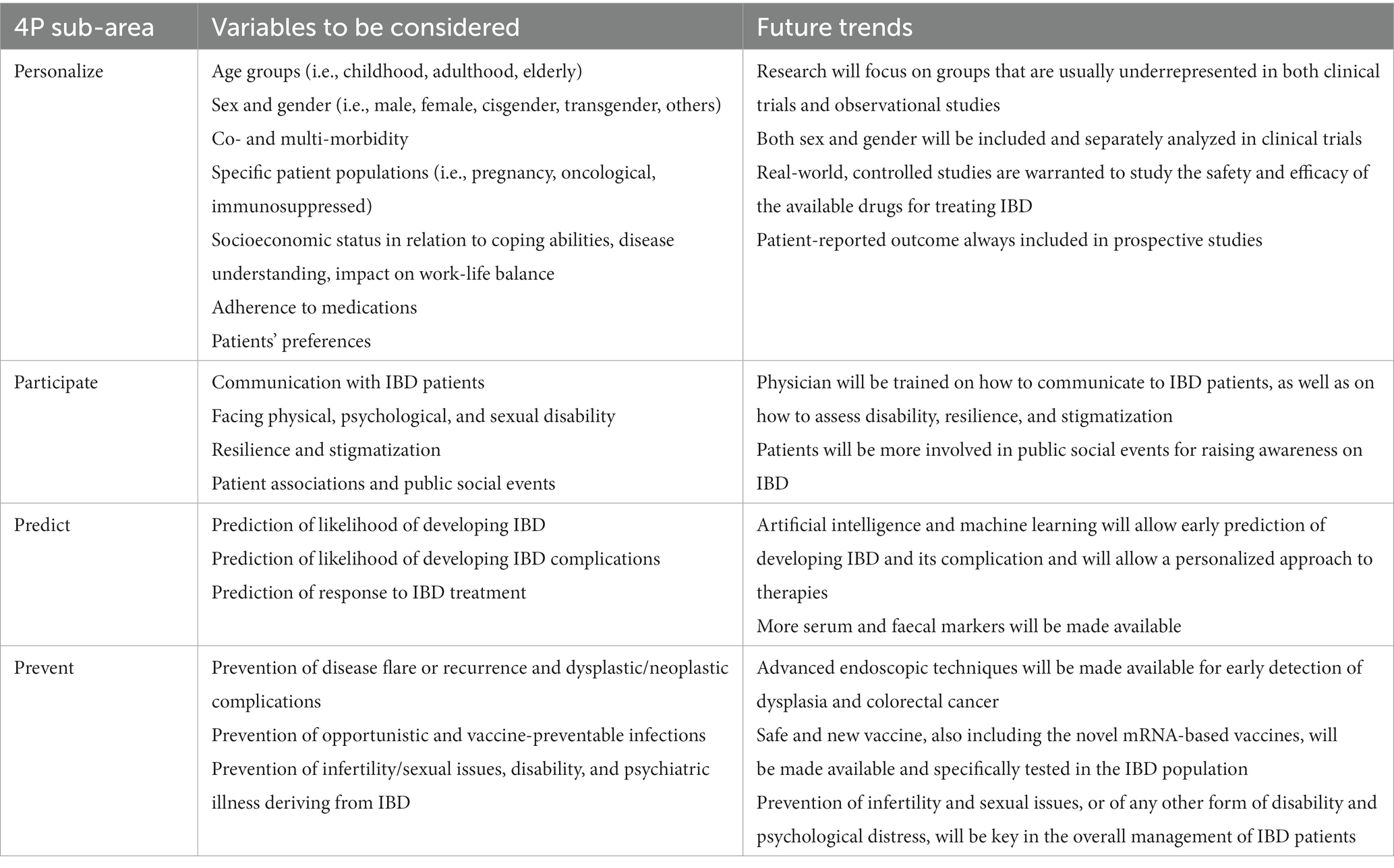
Table 2. Main variables to be considered in the 4P medicine framework that could prove useful in the overall management of inflammatory bowel disease (IBD).
In the future, new tools for predicting important disease outcomes may become available and are eagerly awaited. Particularly, classical statistics, such as linear regression or advanced regression analyses, may fail to capture a meaningful sense of all the variables called into question in IBD. In fact, the increase in data complexity makes classical statistical inference less tractable (262). Recent advancements in the research field of machine learning have fostered the development and deployment of healthcare-focused algorithms. In this context, historical patient data are used to draw correlations and build machine learning models able to predict, with a known confidence, what is likely to happen to a patient, given their current medical condition. The use of artificial intelligence algorithms (263) may be able to capture and decipher complex, nonlinear relationships, that otherwise would be left unconsidered, and could be consider as an upgrade of the 4P medicine. So far, the only application of artificial intelligence in the field of IBD is that of endoscopy, field in which it proved useful in detecting preneoplastic alterations (264). A broader clinical application of artificial intelligence should be sought in future studies.
Author contributions
MVL and ADS introduction, conclusions, and outlook. MLS tailoring treatment of IBD in pregnant women. LB tailoring treatment of IBD in cancer patients. RC tailoring treatment of IBD in patients with superimposed viral infections. DP how to communicate with IBD patients. LP disability in IBD. MVL stigmatization and resilience in IBD. GF quality of care standards in IBD. FC predicting relapse through faecal markers in IBD. SA predicting response and toxicity to thiopurines in IBD. MF predicting response to biologics in IBD. GT preventing flare-up and dysplasia through advanced endoscopy in IBD. AO preventing infections in IBD. GMS preventing post-surgical recurrence in IBD. ES unmet needs in IBD. GC, MVL, ADS, RD’I, MD, MV, GM, AK, and GCS overview and revision for important intellectual contents. All authors contributed to the article and approved the submitted version.
Funding
The publication costs are supported by the network “Rete Aging” – Fondazione IRCCS Policlinico San Matteo.
Acknowledgments
MVL is grateful to the University of Pavia for supporting his research projects. We thank Giacomo Broglio for technical support.
Conflict of interest
ES has served as speaker for Abbvie, AGPharma, Alfasigma, EG Stada Group, Fresenius Kabi, Grifols, Janssen, Innovamedica, Malesci, Pfizer, Reckitt Benckiser, Sandoz, SILA, Sofar, Takeda, Unifarco; has served as consultant for Alfasigma, Amgen, Biogen, Bristol-Myers Squibb, Celltrion, Diadema Farmaceutici, Falk, Fresenius Kabi, Janssen, Merck & Co, Reckitt Benckiser, Regeneron, Sanofi, Shire, SILA, Sofar, Synformulas GmbH, Takeda, Unifarco; he received research support from Reckitt Benckiser, SILA, Sofar, Unifarco. DP has received speaker’s fee from Takeda, Pfizer, MSD, Janssen, MSD, and a grant from Amgen and Pfizer.
The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Publisher’s note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
References
1. Roda, G, Chien Ng, S, Kotze, PG, Argollo, M, Panaccione, R, Spinelli, A, et al. Crohn's disease. Nat Rev Dis Primers. (2020) 6:22. doi: 10.1038/s41572-020-0156-2
2. Kobayashi, T, Siegmund, B, Le Berre, C, Wei, SC, Ferrante, M, Shen, B, et al. Ulcerative colitis. Nat Rev Dis Primers. (2020) 6:74. doi: 10.1038/s41572-020-0205-x
3. Lenti, MV, and Di Sabatino, A. Intestinal fibrosis. Mol Asp Med. (2019) 65:100–9. doi: 10.1016/j.mam.2018.10.003
4. Lenti, MV, Cococcia, S, Ghorayeb, J, Di Sabatino, A, and Selinger, CP. Stigmatisation and resilience in inflammatory bowel disease. Intern Emerg Med. (2020) 15:211–23. doi: 10.1007/s11739-019-02268-0
5. GBD 2017 Inflammatory Bowel Disease Collaborators. The global, regional, and national burden of inflammatory bowel disease in 195 countries and territories, 1990-2017: a systematic analysis for the global burden of disease study 2017. Lancet Gastroenterol Hepatol. (2020) 5:17–30. doi: 10.1016/S2468-1253(19)30333-4
6. Al-Bawardy, B, Shivashankar, R, and Proctor, DD. Novel and emerging therapies for inflammatory bowel disease. Front Pharmacol. (2021) 12:651415. doi: 10.3389/fphar.2021.651415
7. Corazza, GR, Formagnana, P, and Lenti, MV. Bringing complexity into clinical practice: an internistic approach. Eur J Intern Med. (2019) 61:9–14. doi: 10.1016/j.ejim.2018.11.009
8. Raine, T, Bonovas, S, Burisch, J, Kucharzik, T, Adamina, M, Annese, V, et al. ECCO guidelines on therapeutics in ulcerative colitis: medical treatment. J Crohns Colitis. (2022) 16:2–17. doi: 10.1093/ecco-jcc/jjab178
9. Spinelli, A, Bonovas, S, Burisch, J, Kucharzik, T, Adamina, M, Annese, V, et al. ECCO guidelines on therapeutics in ulcerative colitis: surgical treatment. J Crohns Colitis. (2022) 16:179–89. doi: 10.1093/ecco-jcc/jjab177
10. Adamina, M, Bonovas, S, Raine, T, Spinelli, A, Warusavitarne, J, Armuzzi, A, et al. ECCO guidelines on therapeutics in Crohn's disease: surgical treatment. J Crohns Colitis. (2020) 14:155–68. doi: 10.1093/ecco-jcc/jjz187
11. Torres, J, Bonovas, S, Doherty, G, Kucharzik, T, Gisbert, JP, Raine, T, et al. ECCO guidelines on therapeutics in Crohn's disease: medical treatment. J Crohns Colitis. (2020) 14:4–22. doi: 10.1093/ecco-jcc/jjz180
12. Flores, M, Glusman, G, Brogaard, K, Price, ND, and Hood, L. P4 medicine: how systems medicine will transform the healthcare sector and society. Per Med. (2013) 10:565–76. doi: 10.2217/pme.13.57
13. Gallinger, ZR, Rumman, A, and Nguyen, GC. Perceptions and attitudes towards medication adherence during pregnancy in inflammatory bowel disease. J Crohns Colitis. (2016) 10:892–7. doi: 10.1093/ecco-jcc/jjw052
14. Bortoli, A, Pedersen, N, Duricova, D, Dinca, R, Gionchetti, P, Panelli, MR, et al. European Crohn-colitis organisation (ECCO) study Group of Epidemiologic Committee (EpiCom). Pregnancy outcome in inflammatory bowel disease: prospective European case-control ECCO-EpiCom study, 2003-2006. Aliment Pharmacol Ther. (2011) 34:724–34. doi: 10.1111/j.1365-2036.2011.04794.x
15. Mahadevan, U, Robinson, C, Bernasko, N, Boland, B, Chambers, C, Dubinsky, M, et al. Inflammatory bowel disease in pregnancy clinical care pathway: a report from the American Gastroenterological Association IBD parenthood project working group. Gastroenterology. (2019) 156:1508–24. doi: 10.1053/j.gastro.2018.12.022
16. van der Woude, CJ, Ardizzone, S, Bengtson, MB, Fiorino, G, Fraser, G, Katsanos, K, et al. European Crohn’s and colitis organization. The second European evidenced-based consensus on reproduction and pregnancy in inflammatory bowel disease. J Crohns Colitis. (2015) 9:107–24. doi: 10.1093/ecco-jcc/jju006
17. Nguyen, GC, Seow, CH, Maxwell, C, Huang, V, Leung, Y, Jones, J, et al. The Toronto consensus statements for the Management of Inflammatory Bowel Disease in pregnancy. Gastroenterology. (2016) 150:734–757.e1. doi: 10.1053/j.gastro.2015.12.003
18. Italian Group for the Study of Inflammatory Bowel Disease Working GroupArmuzzi, A, Bortoli, A, Castiglione, F, Contaldo, A, Daperno, M, et al. Female reproductive health and inflammatory bowel disease: a practice-based review. Dig Liver Dis. (2022) 54:19–29. doi: 10.1016/j.dld.2021.05.020
19. Odufalu, FD, Long, M, Lin, K, and Mahadevan, U, PIANO investigators from the Crohn’s and colitis foundation (CCF) clinical research Alliance recruited patients for their respective centers for participant enrollment. Exposure to corticosteroids in pregnancy is associated with adverse perinatal outcomes among infants of mothers with inflammatory bowel disease: results from the PIANO registry. Gut. (2021) 71:1766–72. doi: 10.1136/gutjnl-2021-325317
20. Kanis, SL, de Lima-Karagiannis, A, de Boer, NKH, and van der Woude, CJ. Use of Thiopurines during conception and pregnancy is not associated with adverse pregnancy outcomes or health of infants at one year in a prospective study. Clin Gastroenterol Hepatol. (2017) 15:1232–1241.e1. doi: 10.1016/j.cgh.2017.02.041
21. Mahadevan, U, Long, MD, Kane, SV, Roy, A, Dubinsky, MC, Sands, BE, et al. Pregnancy and neonatal outcomes after fetal exposure to biologics and Thiopurines among women with inflammatory bowel disease. Gastroenterology. (2021) 160:1131–9. doi: 10.1053/j.gastro.2020.11.038
22. Branche, J, Cortot, A, Bourreille, A, Coffin, B, de Vos, M, de Saussure, P, et al. Cyclosporine treatment of steroid-refractory ulcerative colitis during pregnancy. Inflamm Bowel Dis. (2009) 15:1044–8. doi: 10.1002/ibd.20858
23. Nielsen, OH, Gubatan, JM, Juhl, CB, Streett, SE, and Maxwell, C. Biologics for inflammatory bowel disease and their safety in pregnancy: a systematic review and meta-analysis. Clin Gastroenterol Hepatol. (2022) 20:74–87.e3. doi: 10.1016/j.cgh.2020.09.021
24. Lichtenstein, GR, Feagan, BG, Mahadevan, U, Salzberg, BA, Langholff, W, Morgan, JG, et al. Pregnancy outcomes reported during the 13-year TREAT registry: a descriptive report. Am J Gastroenterol. (2018) 113:1678–88. doi: 10.1038/s41395-018-0202-9
25. Chaparro, M, Verreth, A, Lobaton, T, Gravito-Soares, E, Julsgaard, M, Savarino, E, et al. Long-term safety of in utero exposure to anti-TNFα drugs for the treatment of inflammatory bowel disease: results from the multicenter European TEDDY study. Am J Gastroenterol. (2018) 113:396–403. doi: 10.1038/ajg.2017.501
26. Moens, A, van Hoeve, K, Humblet, E, Rahier, JF, Bossuyt, P, Dewit, S, et al. Outcome of pregnancies in female patients with inflammatory bowel diseases treated with Vedolizumab. J Crohns Colitis. (2019) 13:12–8. doi: 10.1093/ecco-jcc/jjy142
27. Bar-Gil Shitrit, A, Ben Yaʼacov, A, Livovsky, DM, Cuker, T, Farkash, R, Hoyda, A, et al. Exposure to Vedolizumab in IBD pregnant women appears of low risk for mother and neonate: a first prospective comparison study. Am J Gastroenterol. (2019) 114:1172–5. doi: 10.14309/ajg.0000000000000186
28. Moens, A, van der Woude, CJ, Julsgaard, M, Humblet, E, Sheridan, J, Baumgart, DC, et al. Pregnancy outcomes in inflammatory bowel disease patients treated with vedolizumab, anti-TNF or conventional therapy: results of the European CONCEIVE study. Aliment Pharmacol Ther. (2020) 51:129–38. doi: 10.1111/apt.15539
29. Wils, P, Seksik, P, Stefanescu, C, Nancey, S, Allez, M, Pineton de Chambrun, G, et al. Safety of ustekinumab or vedolizumab in pregnant inflammatory bowel disease patients: a multicentre cohort study. Aliment Pharmacol Ther. (2021) 53:460–70. doi: 10.1111/apt.16192
30. Chaparro, M, Gutiérrez, A, Calviño-Suárez, C, Huguet, JM, Calvo, M, Aguas, M, et al. Safety of ustekinumab in pregnant patients with inflammatory bowel disease and in their offspring: results from the DUMBO registry of GETECCU. J Crohns Colitis. (2022) 16:i491–3. doi: 10.1093/ecco-jcc/jjab232.672
31. Clowse, ME, Feldman, SR, Isaacs, JD, Kimball, AB, Strand, V, and Warren, RB. Pregnancy outcomes in the Tofacitinib safety databases for rheumatoid arthritis and psoriasis. Drug Saf. (2016) 39:755–62. doi: 10.1007/s40264-016-0431-z
32. Mahadevan, U, Dubinsky, MC, Su, C, Lawendy, N, Jones, TV, Marren, A, et al. Outcomes of pregnancies with maternal/paternal exposure in the Tofacitinib safety databases for ulcerative colitis. Inflamm Bowel Dis. (2018) 24:2494–500. doi: 10.1093/ibd/izy160
33. Beaugerie, L, and Itzkowitz, SH. Cancers complicating inflammatory bowel disease. N Engl J Med. (2015) 372:1441–52. doi: 10.1056/NEJMra1403718
34. Annese, V, Beaugerie, L, Egan, L, Biancone, L, Bolling, C, Brandts, C, et al. European evidence-based consensus: inflammatory bowel disease and malignancies. J Crohns Colitis. (2015) 9:945–65. doi: 10.1093/ecco-jcc/jjv141
35. Beaugerie, L, Carrat, F, Colombel, JF, Bouvier, AM, Sokol, H, Babouri, A, et al. Risk of new or recurrent cancer under immunosuppressive therapy in patients with IBD and previous cancer. Gut. (2014) 63:1416–23. doi: 10.1136/gutjnl-2013-305763
36. Shelton, E, Laharie, D, Scott, FI, Mamtani, R, Lewis, JD, Colombel, JF, et al. Cancer recurrence following immune-suppressive therapies in patients with immune-mediated diseases: a systematic review and meta-analysis. Gastroenterology. (2016) 151:97–109.e4. doi: 10.1053/j.gastro.2016.03.037
37. Salliot, C, and van der Heijde, D. Long-term safety of methotrexate monotherapy in patients with rheumatoid arthritis: a systematic literature research. Ann Rheum Dis. (2009) 68:1100–4. doi: 10.1136/ard.2008.093690
38. Biancone, L, Orlando, A, Kohn, A, Colombo, E, Sostegni, R, Angelucci, E, et al. Infliximab and newly diagnosed neoplasia in Crohn's disease: a multicentre matched pair study. Gut. (2006) 55:228–33. doi: 10.1136/gut.2005.075937
39. Lichtenstein, GR, Feagan, BG, Cohen, RD, Salzberg, BA, Diamond, RH, Langholff, W, et al. Drug therapies and the risk of malignancy in Crohn's disease: results from the TREAT™ registry. Am J Gastroenterol. (2014) 109:212–23. doi: 10.1038/ajg.2013.441
40. Bonovas, S, Fiorino, G, Allocca, M, Lytras, T, Nikolopoulos, GK, Peyrin-Biroulet, L, et al. Biologic therapies and risk of infection and malignancy in patients with inflammatory bowel disease: a systematic review and network meta-analysis. Clin Gastroenterol Hepatol. (2016) 14:1385–1397.e10. doi: 10.1016/j.cgh.2016.04.039
41. Nyboe Andersen, N, Pasternak, B, Basit, S, Andersson, M, Svanström, H, Caspersen, S, et al. Association between tumor necrosis factor-α antagonists and risk of cancer in patients with inflammatory bowel disease. JAMA. (2014) 311:2406–13. doi: 10.1001/jama.2014.5613
42. Williams, CJ, Peyrin-Biroulet, L, and Ford, AC. Systematic review with meta-analysis: malignancies with anti-tumour necrosis factor-α therapy in inflammatory bowel disease. Aliment Pharmacol Ther. (2014) 39:447–58. doi: 10.1111/apt.12624
43. Singh, S, Nagpal, SJ, Murad, MH, Yadav, S, Kane, SV, Pardi, DS, et al. Inflammatory bowel disease is associated with an increased risk of melanoma: a systematic review and meta-analysis. Clin Gastroenterol Hepatol. (2014) 12:210–8. doi: 10.1016/j.cgh.2013.04.033
44. Kotlyar, DS, Osterman, MT, Diamond, RH, Porter, D, Blonski, WC, and Wasik, M. A systematic review of factors that contribute to hepatosplenic T-cell lymphoma in patients with inflammatory bowel disease. Clin Gastroenterol Hepatol. (2011) 9:36–41.e1. doi: 10.1016/j.cgh.2010.09.016
45. Axelrad, J, Bernheim, O, Colombel, JF, Malerba, S, Ananthakrishnan, A, Yajnik, V, et al. Risk of new or recurrent cancer in patients with inflammatory bowel disease and previous cancer exposed to immunosuppressive and anti-tumor necrosis factor agents. Clin Gastroenterol Hepatol. (2016) 14:58–64. doi: 10.1016/j.cgh.2015.07.037
46. Poullenot, F, Seksik, P, Beaugerie, L, Amiot, A, Nachury, M, Abitbol, V, et al. Risk of incident cancer in inflammatory bowel disease patients starting anti-TNF therapy while having recent malignancy. Inflamm Bowel Dis. (2016) 22:1362–9. doi: 10.1097/MIB.0000000000000741
47. Card, T, Ungaro, R, Bhayat, F, Blake, A, Hantsbarger, G, and Travis, S. Vedolizumab use is not associated with increased malignancy incidence: GEMINI LTS study results and post-marketing data. Aliment Pharmacol Ther. (2020) 51:149–57. doi: 10.1111/apt.15538
48. Sandborn, WJ, Feagan, BG, Danese, S, O'Brien, CD, Ott, E, Marano, C, et al. Safety of Ustekinumab in inflammatory bowel disease: pooled safety analysis of results from phase 2/3 studies. Inflamm Bowel Dis. (2021) 27:994–1007. doi: 10.1093/ibd/izaa236
49. Vedamurthy, A, Gangasani, N, and Ananthakrishnan, AN. Vedolizumab or tumor necrosis factor antagonist use and risk of new or recurrent cancer in patients with inflammatory bowel disease with prior malignancy: a retrospective cohort study. Clin Gastroenterol Hepatol. (2022) 20:88–95. doi: 10.1016/j.cgh.2020.10.007
50. Hong, SJ, Zenger, C, Pecoriello, J, Pang, A, Vallely, M, Hudesman, DP, et al. Ustekinumab and Vedolizumab are not associated with subsequent cancer in IBD patients with prior malignancy. Inflamm Bowel Dis. (2022) 28:1826–32. doi: 10.1093/ibd/izac035
51. Sandborn, WJ, Lawendy, N, Danese, S, Su, C, Loftus, EVJr, Hart, A, et al. Safety and efficacy of tofacitinib for treatment of ulcerative colitis: final analysis of OCTAVE open, an open-label, long-term extension study with up to 7.0 years of treatment. Aliment Pharmacol Ther. (2022) 55:464–78. doi: 10.1111/apt.16712
52. Vega, R, Bertrán, X, Menacho, M, Domènech, E, Moreno de Vega, V, Hombrados, M, et al. Cytomegalovirus infection in patients with inflammatory bowel disease. Am J Gastroenterol. (1999) 94:1053–6. doi: 10.1111/j.1572-0241.1999.01013.x
53. Yanai, H, Shimizu, N, Nagasaki, S, Mitani, N, and Okita, K. Epstein-Barr virus infection of the colon with inflammatory bowel disease. Am J Gastroenterol. (1999) 94:1582–6. doi: 10.1111/j.1572-0241.1999.01148.x
54. Jarvis, MA, and Nelson, JA. Human cytomegalovirus persistence and latency in endothelial cells and macrophages. Curr Opin Microbiol. (2002) 5:403–7. doi: 10.1016/s1369-5274(02)00334-x
55. Sinclair, J. Human cytomegalovirus: latency and reactivation in the myeloid lineage. J Clin Virol. (2008) 41:180–5. doi: 10.1016/j.jcv.2007.11.014
56. Thorley-Lawson, DA, Hawkins, JB, Tracy, SI, and Shapiro, M. The pathogenesis of Epstein-Barr virus persistent infection. Curr Opin Virol. (2013) 3:227–32. doi: 10.1016/j.coviro.2013.04.005
57. Loewendorf, A, and Benedict, CA. Modulation of host innate and adaptive immune defenses by cytomegalovirus: timing is everything. J Intern Med. (2010) 267:483–501. doi: 10.1111/j.1365-2796.2010.02220.x
58. Murata, T, and Tsurumi, T. Switching of EBV cycles between latent and lytic states. Rev Med Virol. (2014) 24:142–53. doi: 10.1002/rmv.1780
59. Vogl, BA, Fagin, U, Nerbas, L, Schlenke, P, Lamprecht, P, and Jabs, WJ. Longitudinal analysis of frequency and reactivity of Epstein-Barr virus-specific T lymphocytes and their association with intermittent viral reactivation. J Med Virol. (2012) 84:119–31. doi: 10.1002/jmv.22258
60. Sager, K, Alam, S, Bond, A, Chinnappan, L, and Probert, CS. Review article: cytomegalovirus and inflammatory bowel disease. Aliment Pharmacol Ther. (2015) 41:725–33. doi: 10.1111/apt.13124
61. Ryan, JL, Shen, YJ, Morgan, DR, Thorne, LB, Kenney, SC, Dominguez, RL, et al. Epstein-Barr virus infection is common in inflamed gastrointestinal mucosa. Dig Dis Sci. (2012) 57:1887–98. doi: 10.1007/s10620-012-2116-5
62. Ciccocioppo, R, Racca, F, Paolucci, S, Campanini, G, Pozzi, L, Betti, E, et al. Human cytomegalovirus and Epstein-Barr virus infection in inflammatory bowel disease: need for mucosal viral load measurement. World J Gastroenterol. (2015) 21:1915–26. doi: 10.3748/wjg.v21.i6.1915
63. Forbes, BA, Bonville, CA, and Dock, NL. The effects of a promoter of cell differentiation and selected hormones on human cytomegalovirus infection using an in vitro cell system. J Infect Dis. (1990) 162:39–45. doi: 10.1093/infdis/162.1.39
64. Kirman, I, Whelan, RL, and Nielsen, OH. Infliximab: mechanism of action beyond TNF-alpha neutralization in inflammatory bowel disease. Eur J Gastroenterol Hepatol. (2004) 16:639–41. doi: 10.1097/01.meg.0000108345.41221.c2
65. Sylwester, AW, Mitchell, BL, Edgar, JB, Taormina, C, Pelte, C, Ruchti, F, et al. Broadly targeted human cytomegalovirus-specific CD4+ and CD8+ T cells dominate the memory compartments of exposed subjects. J Exp Med. (2005) 202:673–85. doi: 10.1084/jem.20050882
66. Kotton, CN, Kumar, D, Caliendo, AM, Huprikar, S, Chou, S, Danziger-Isakov, L, et al. The third international consensus guidelines on the Management of Cytomegalovirus in solid-organ transplantation. Transplantation. (2018) 102:900–31. doi: 10.1097/TP.0000000000002191
67. Green, M, and Michaels, MG. Epstein-Barr virus infection and posttransplant lymphoproliferative disorder. Am J Transplant. (2013) 13:41–54. doi: 10.1111/ajt.12004
68. Keller, MD, and Bollard, CM. Virus-specific T-cell therapies for patients with primary immune deficiency. Blood. (2020) 135:620–8. doi: 10.1182/blood.2019000924
69. Ciccocioppo, R, Mengoli, C, Betti, E, Comolli, G, Cassaniti, I, Piralla, A, et al. Human cytomegalovirus and Epstein-Barr virus specific immunity in patients with ulcerative colitis. Clin Exp Med. (2021) 21:379–88. doi: 10.1007/s10238-021-00702-2
70. Dayharsh, GA, Loftus, EVJr, Sandborn, WJ, Tremaine, WJ, Zinsmeister, AR, Witzig, TE, et al. Epstein-Barr virus-positive lymphoma in patients with inflammatory bowel disease treated with azathioprine or 6-mercaptopurine. Gastroenterology. (2002) 122:72–7. doi: 10.1053/gast.2002.30328
71. Baars, JE, Markus, T, Kuipers, EJ, and van der Woude, CJ. Patients' preferences regarding shared decision-making in the treatment of inflammatory bowel disease: results from a patient-empowerment study. Digestion. (2010) 81:113–9. doi: 10.1159/000253862
72. Glass, KE, Wills, CE, Holloman, C, Olson, J, Hechmer, C, Miller, CK, et al. Shared decision making and other variables as correlates of satisfaction with health care decisions in a United States national survey. Patient Educ Couns. (2012) 88:100–5. doi: 10.1016/j.pec.2012.02.010
73. Zolnierek, KB, and Dimatteo, MR. Physician communication and patient adherence to treatment: a meta-analysis. Med Care. (2009) 47:826–34. doi: 10.1097/MLR.0b013e31819a5acc
74. Burisch, J, Vegh, Z, Pedersen, N, Cuković-Čavka, S, Turk, N, Kaimakliotis, I, et al. Health care and patients' education in a European inflammatory bowel disease inception cohort: an ECCO-EpiCom study. J Crohns Colitis. (2014) 8:811–8. doi: 10.1016/j.crohns.2013.12.023
75. Chew, D, Zhiqin, W, Ibrahim, N, and Ali, RAR. Optimizing the multidimensional aspects of the patient-physician relationship in the management of inflammatory bowel disease. Intest Res. (2018) 16:509–21. doi: 10.5217/ir.2018.00074
76. Drossman, DA, Leserman, J, Li, ZM, Mitchell, CM, Zagami, EA, and Patrick, DL. The rating form of IBD patient concerns: a new measure of health status. Psychosom Med. (1991) 53:701–12. doi: 10.1097/00006842-199111000-00010
77. Bernstein, KI, Promislow, S, Carr, R, Rawsthorne, P, Walker, JR, and Bernstein, CN. Information needs and preferences of recently diagnosed patients with inflammatory bowel disease. Inflamm Bowel Dis. (2011) 17:590–8. doi: 10.1002/ibd.21363
78. Rubin, DT, Hart, A, Panaccione, R, Armuzzi, A, Suvanto, U, Deuring, JJ, et al. Ulcerative colitis narrative global survey findings: communication gaps and agreements between patients and physicians. Inflamm Bowel Dis. (2021) 27:1096–106. doi: 10.1093/ibd/izaa257
79. Christensen, B, Prentice, RE, Al-Ani, AH, Zhang, E, Bedell, A, and Rubin, DT. Self-reported failure to address sexual function in patients with inflammatory bowel disease by gastroenterologists: barriers and areas for improvement. Inflamm Bowel Dis. (2022) 28:1465–8. doi: 10.1093/ibd/izac025
80. Alexakis, C, Davies, G, Stephens, J, Clark, S, Rogers, S, and Poullis, A. Perspectives and attitudes of young patients with inflammatory bowel disease: symptoms, burden of disease and communication with their healthcare professionals. Frontline Gastroenterol. (2014) 5:197–202. doi: 10.1136/flgastro-2013-100400
81. Linn, AJ, Schouten, BC, Sanders, R, van Weert, JCM, and Bylund, CL. Talking about Dr. Google: communication strategies used by nurse practitioners and patients with inflammatory bowel disease in the Netherlands to discuss online health information. Patient Educ Couns. (2020) 103:1216–22. doi: 10.1016/j.pec.2020.01.011
82. Fischhoff, B, et al. Communicating Risks and Benefits: An Evidence-Based User's Guide. (2011). Available at: http://www.fda.gov/AboutFDA/ReportsManualsForms/Reports/ucm268078.html
83. Kurt, S, Caron, B, Gouynou, C, Netter, P, Vaizey, CJ, Wexner, SD, et al. Faecal incontinence in inflammatory bowel disease: the Nancy experience. Dig Liver Dis. (2022) 54:1195–201. doi: 10.1016/j.dld.2022.01.125
84. Marinelli, C, Savarino, EV, Marsilio, I, Lorenzon, G, Gavaruzzi, T, D'Incà, R, et al. Sleep disturbance in inflammatory bowel disease: prevalence and risk factors – a cross-sectional study. Sci Rep. (2020) 10:507. doi: 10.1038/s41598-020-57460-6
85. World Health Organization. International classification of impairments, disabilities and handicaps of the World Health Organisation. (1976). Available at: https://apps.who.int/iris/bitstream/handle/10665/41003/9241541261_eng.pdf;sequence=1
86. Tannoury, J, Nachury, M, Martins, C, Serrero, M, Filippi, J, Roblin, X, et al. Determinants of IBD-related disability: a cross-sectional survey from the GETAID. Aliment Pharmacol Ther. (2021) 53:1098–107. doi: 10.1111/apt.16353
87. Peyrin-Biroulet, L, Cieza, A, Sandborn, WJ, Coenen, M, Chowers, Y, Hibi, T, et al. Development of the first disability index for inflammatory bowel disease based on the international classification of functioning, disability and health. Gut. (2012) 61:241–7. doi: 10.1136/gutjnl-2011-300049
88. Ghosh, S, Louis, E, Beaugerie, L, Bossuyt, P, Bouguen, G, Bourreille, A, et al. Development of the IBD disk: a visual self-administered tool for assessing disability in inflammatory bowel diseases. Inflamm Bowel Dis. (2017) 23:333–40. doi: 10.1097/MIB.0000000000001033
89. Marinelli, C, Savarino, E, Inferrera, M, Lorenzon, G, Rigo, A, Ghisa, M, et al. Influencing disability and quality of life during treatment: a cross-sectional study on IBD patients. Gastroenterol Res Pract. (2019) 2019:5354320. doi: 10.1155/2019/5354320
90. Shafer, LA, Walker, JR, Chhibba, T, Targownik, LE, Singh, H, Ivekovic, M, et al. Health care indicators of moderate to severe IBD and subsequent IBD-related disability: a longitudinal study. Inflamm Bowel Dis. (2019) 25:1996–2005. doi: 10.1093/ibd/izz102
91. Kayal, M, Ungaro, RC, Riggs, A, Kamal, K, Agrawal, M, Cohen-Mekelburg, S, et al. Ileal pouch anal anastomosis for the Management of Ulcerative Colitis is Associated with Significant Disability. Clin Gastroenterol Hepatol. (2022) 20:e761–9. doi: 10.1016/j.cgh.2021.05.033
92. Paulides, E, Daker, C, Frampton, C, Gearry, RB, Eglinton, T, de Boer, NKH, et al. Overcoming workplace disability in IBD patients: an observational study. Inflamm Intest Dis. (2020) 5:84–92. doi: 10.1159/000506702
93. Ramos, A, Calvet, X, Sicilia, B, Vergara, M, Figuerola, A, Motos, J, et al. IBD-related work disability in the community: prevalence, severity and predictive factors. A cross-sectional study. United Eur Gastroenterol J. (2015) 3:335–42. doi: 10.1177/2050640615577532
94. Leso, V, Gervetti, P, Macrini, MC, Russo, F, and Iavicoli, I. Inflammatory bowel diseases and work disability: a systematic review of predictive factors. Eur Rev Med Pharmacol Sci. (2021) 25:165–81. doi: 10.26355/eurrev_202101_24382
95. van Gennep, S, de Boer, NKH, Gielen, ME, Rietdijk, ST, Gecse, KB, Ponsioen, CY, et al. Impaired quality of working life in inflammatory bowel disease patients. Dig Dis Sci. (2021) 66:2916–24. doi: 10.1007/s10620-020-06647-y
96. Everhov, ÅH, Bruze, G, Söderling, J, Askling, J, Halfvarson, J, Westberg, K, et al. Women's earnings are more affected by inflammatory bowel disease than Men's: a register-based Swedish cohort study. J Crohns Colitis. (2021) 15:980–7. doi: 10.1093/ecco-jcc/jjaa238
97. Allen, PB, Gower-Rousseau, C, Danese, S, and Peyrin-Biroulet, L. Preventing disability in inflammatory bowel disease. Ther Adv Gastroenterol. (2017) 10:865–76. doi: 10.1177/1756283X17732720
98. Cococcia, S, Lenti, MV, Mengoli, C, Klersy, C, Borrelli de Andreis, F, Borrelli de Andreis, F, et al. Validation of the Italian translation of the perceived stigma scale and resilience assessment in inflammatory bowel disease patients. World J Gastroenterol. (2021) 27:6647–58. doi: 10.3748/wjg.v27.i39.6647
99. Taft, TH, and Keefer, L. A systematic review of disease-related stigmatization in patients living with inflammatory bowel disease. Clin Exp Gastroenterol. (2016) 9:49–58. doi: 10.2147/CEG.S83533
100. Taft, TH, Keefer, L, Leonhard, C, and Nealon-Woods, M. Impact of perceived stigma on inflammatory bowel disease patient outcomes. Inflamm Bowel Dis. (2009) 15:1224–32. doi: 10.1002/ibd.20864
101. Link, BG, and Phelan, JC. Conceptualizing stigma. Annu Rev Sociol. (2001) 27:363–85. doi: 10.1146/annurev.soc.27.1.363
102. Corrigan, PW, and Watson, AC. Understanding the impact of stigma on people with mental illness. World Psychiatry. (2002) 1:16–20.
103. Mahajan, AP, Sayles, JN, Patel, VA, Remien, RH, Sawires, SR, Ortiz, DJ, et al. Stigma in the HIV/AIDS epidemic: a review of the literature and recommendations for the way forward. AIDS. (2008) 22:S67–79. doi: 10.1097/01.aids.0000327438.13291.62
104. Lebel, S, and Devins, GM. Stigma in cancer patients whose behavior may have contributed to their disease. Future Oncol. (2008) 4:717–33. doi: 10.2217/14796694.4.5.717
105. Jones, MP, Keefer, L, Bratten, J, Taft, TH, Crowell, MD, Levy, R, et al. Development and initial validation of a measure of perceived stigma in irritable bowel syndrome. Psychol Health Med. (2009) 14:367–74. doi: 10.1080/13548500902865956
106. The Road to Resilience – American Psychological Association; Available at: https://www.apa.org/ (Accessed November 10, 2022).
107. Cal, SF, Sá, LR, Glustak, ME, and Santiago, MB. Resilience in chronic diseases: a systematic review. Cogent Psychol. (2015) 2:1024928. doi: 10.4172/2324-005.1000176
108. Quiceno, JM, and Venaccio, S. Resilience: a perspective from the chronic disease in the adult population. Pensam Psicol. (2011) 9:69–82. doi: 10.1007/978-3-030-32811-5_117-1
109. Ghanei Gheshlagh, R, Sayehmiri, K, Ebadi, A, Dalvandi, A, Dalvand, S, and Nourozi, TK. Resilience of patients with chronic physical diseases: a systematic review and meta-analysis. Iran Red Crescent Med J. (2016) 18:e38562. doi: 10.5812/ircmj.38562
110. Connor, KM, and Davidson, JR. Development of a new resilience scale: the Connor-Davidson resilience scale (CD-RISC). Depress Anxiety. (2003) 18:76–82. doi: 10.1002/da.10113
111. Macedo, T, Wilheim, L, Gonçalves, R, Coutinho, ES, Vilete, L, Figueira, I, et al. Building resilience for future adversity: a systematic review of interventions in non-clinical samples of adults. BMC Psychiatry. (2014) 14:227. doi: 10.1186/s12888-014-0227-6
112. Hwang, JH, and Yu, CS. Depression and resilience in ulcerative colitis and Crohn's disease patients with ostomy. Int Wound J. (2019) 16:62–70. doi: 10.1111/iwj.13076
113. Fiorino, G, and Danese, S. The new frontier: certifying quality standards in the inflammatory bowel disease care. United European Gastroenterol J. (2021) 9:745–7. doi: 10.1002/ueg2.12122
114. Kucharzik, T, Ellul, P, Greuter, T, Rahier, JF, Verstockt, B, Abreu, C, et al. ECCO guidelines on the prevention, diagnosis, and Management of Infections in inflammatory bowel disease. J Crohns Colitis. (2021) 15:879–913. doi: 10.1093/ecco-jcc/jjab052
115. Maaser, C, Sturm, A, Vavricka, SR, Kucharzik, T, Fiorino, G, Annese, V, et al. ECCO-ESGAR guideline for diagnostic assessment in IBD part 1: initial diagnosis, monitoring of known IBD, detection of complications. J Crohns Colitis. (2019) 13:144–64. doi: 10.1093/ecco-jcc/jjy113
116. Moffett, P, and Moore, G. The standard of care: legal history and definitions: the bad and good news. West J Emerg Med. (2011) 12:109–12.
117. Fiorino, G, Allocca, M, Chaparro, M, Coenen, S, Fidalgo, C, Younge, L, et al. 'Quality of Care' standards in inflammatory bowel disease: a systematic review. J Crohns Colitis. (2019) 13:127–37. doi: 10.1093/ecco-jcc/jjy140
118. Fiorino, G, Lytras, T, Younge, L, Fidalgo, C, Coenen, S, Chaparro, M, et al. Quality of care standards in inflammatory bowel diseases: a European Crohn's and colitis organisation [ECCO] position paper. J Crohns Colitis. (2020) 14:1037–48. doi: 10.1093/ecco-jcc/jjaa023
119. E.C.C.O. (ECCO), E-QUALITY: ECCO QUALITY OF CARE PROJECT, (2021). Available at: https://www.ecco-ibd.eu/education/e-quality.html (Accessed April 27, 2022).
120. Abraham, C, and Cho, JH. Inflammatory bowel disease. N Engl J Med. (2009) 361:2066–78. doi: 10.1056/NEJMra0804647
121. Rutgeerts, P, Van Assche, G, Sandborn, WJ, Wolf, DC, Geboes, K, Colombel, JF, et al. Adalimumab induces and maintains mucosal healing in patients with Crohn's disease: data from the EXTEND trial. Gastroenterology. (2012) 142:1102–1111.e2. doi: 10.1053/j.gastro.2012.01.035
122. Rutgeerts, P, Diamond, RH, Bala, M, Olson, A, Lichtenstein, GR, Bao, W, et al. Scheduled maintenance treatment with infliximab is superior to episodic treatment for the healing of mucosal ulceration associated with Crohn's disease. Gastrointest Endosc. (2006) 63:433–42. doi: 10.1016/j.gie.2005.08.011
123. Shah, SC, Colombel, JF, Sands, BE, and Narula, N. Systematic review with meta-analysis: mucosal healing is associated with improved long-term outcomes in Crohn's disease. Aliment Pharmacol Ther. (2016) 43:317–33. doi: 10.1111/apt.13475
124. André, C, Descos, L, Vignal, J, and Gillon, J. C-reactive protein as a predictor of relapse in asymptomatic patients with Crohn's disease. Scott Med J. (1983) 28:26–9. doi: 10.1177/003693308302800106
125. Abraham, BP, and Kane, S. Fecal markers: calprotectin and lactoferrin. Gastroenterol Clin N Am. (2012) 41:483–95. doi: 10.1016/j.gtc.2012.01.007
126. Lin, JF, Chen, JM, Zuo, JH, Yu, A, Xiao, ZJ, Deng, FH, et al. Meta-analysis: fecal calprotectin for assessment of inflammatory bowel disease activity. Inflamm Bowel Dis. (2014) 20:1407–15. doi: 10.1097/MIB.0000000000000057
127. Buisson, A, Mak, WY, Andersen, MJ, Lei, D, Kahn, SA, Pekow, J, et al. Faecal calprotectin is a very reliable tool to predict and monitor the risk of relapse after therapeutic De-escalation in patients with inflammatory bowel diseases. J Crohns Colitis. (2019) 13:1012–24. doi: 10.1093/ecco-jcc/jjz023
128. Ferreiro-Iglesias, R, Barreiro-de Acosta, M, Lorenzo-Gonzalez, A, and Dominguez-Muñoz, JE. Accuracy of consecutive fecal calprotectin measurements to predict relapse in inflammatory bowel disease patients under maintenance with anti-TNF therapy: a prospective longitudinal cohort study. J Clin Gastroenterol. (2018) 52:229–34. doi: 10.1097/MCG.0000000000000774
129. Kittanakom, S, Shajib, MS, Garvie, K, Turner, J, Brooks, D, Odeh, S, et al. Comparison of fecal calprotectin methods for predicting relapse of pediatric inflammatory bowel disease. Can J Gastroenterol Hepatol. (2017) 2017:1450970. doi: 10.1155/2017/1450970
130. Diederen, K, Hoekman, DR, Leek, A, Wolters, VM, Hummel, TZ, de Meij, TG, et al. Raised faecal calprotectin is associated with subsequent symptomatic relapse, in children and adolescents with inflammatory bowel disease in clinical remission. Aliment Pharmacol Ther. (2017) 45:951–60. doi: 10.1111/apt.13950
131. Roblin, X, Duru, G, Williet, N, Del Tedesco, E, Cuilleron, M, Jarlot, C, et al. Development and internal validation of a model using fecal calprotectin in combination with infliximab trough levels to predict clinical relapse in Crohn's disease. Inflamm Bowel Dis. (2017) 23:126–32. doi: 10.1097/MIB.0000000000000986
132. Naismith, GD, Smith, LA, Barry, SJ, Munro, JI, Laird, S, Rankin, K, et al. A prospective evaluation of the predictive value of faecal calprotectin in quiescent Crohn's disease. J Crohns Colitis. (2014) 8:1022–9. doi: 10.1016/j.crohns.2014.01.029
133. Laharie, D, Mesli, S, El Hajbi, F, Chabrun, E, Chanteloup, E, Capdepont, M, et al. Prediction of Crohn's disease relapse with faecal calprotectin in infliximab responders: a prospective study. Aliment Pharmacol Ther. (2011) 34:462–9. doi: 10.1111/j.1365-2036.2011.04743.x
134. D'Incà, R, Dal Pont, E, Di Leo, V, Benazzato, L, Martinato, M, Lamboglia, F, et al. Can calprotectin predict relapse risk in inflammatory bowel disease? Am J Gastroenterol. (2008) 103:2007–14. doi: 10.1111/j.1572-0241.2008.01870.x
135. Costa, F, Mumolo, MG, Ceccarelli, L, Bellini, M, Romano, MR, Sterpi, C, et al. Calprotectin is a stronger predictive marker of relapse in ulcerative colitis than in Crohn's disease. Gut. (2005) 54:364–8. doi: 10.1136/gut.2004.043406
136. Tibble, JA, Sigthorsson, G, Bridger, S, Fagerhol, MK, and Bjarnason, I. Surrogate markers of intestinal inflammation are predictive of relapse in patients with inflammatory bowel disease. Gastroenterology. (2000) 119:15–22. doi: 10.1053/gast.2000.8523
137. Mao, R, Xiao, YL, Gao, X, Chen, BL, He, Y, Yang, L, et al. Faecal calprotectin in predicting relapse of inflammatory bowel diseases: a meta-analysis of prospective studies. Inflamm Bowel Dis. (2012) 18:1894–9. doi: 10.1002/ibd.22861
138. Wright, EK, Kamm, MA, De Cruz, P, Hamilton, AL, Ritchie, KJ, Krejany, EO, et al. Measurement of fecal calprotectin improves monitoring and detection of recurrence of Crohn's disease after surgery. Gastroenterology. (2015) 148:938–947.e1. doi: 10.1053/j.gastro.2015.01.026
139. Tham, YS, Yung, DE, Fay, S, Yamamoto, T, Ben-Horin, S, Eliakim, R, et al. Fecal calprotectin for detection of postoperative endoscopic recurrence in Crohn's disease: systematic review and meta-analysis. Ther Adv Gastroenterol. (2018) 11:1756284818785571. doi: 10.1177/1756284818785571
140. Agrawal, M, Spencer, EA, Colombel, JF, and Ungaro, RC. Approach to the management of recently diagnosed inflammatory bowel disease patients: a user's guide for adult and pediatric gastroenterologists. Gastroenterology. (2021) 161:47–65. doi: 10.1053/j.gastro.2021.04.063
141. Chande, N, Townsend, CM, Parker, CE, and MacDonald, JK. Azathioprine or 6-mercaptopurine for induction of remission in Crohn's disease. Cochrane Database Syst Rev. (2016) 10:CD000545. doi: 10.1002/14651858.CD000545.pub5
142. Chande, N, Patton, PH, Tsoulis, DJ, Thomas, BS, and MacDonald, JK. Azathioprine or 6-mercaptopurine for maintenance of remission in Crohn's disease. Cochrane Database Syst Rev. (2015) 2016:CD000067. doi: 10.1002/14651858.CD000067.pub3
143. Ardizzone, S, Maconi, G, Russo, A, Imbesi, V, Colombo, E, and Bianchi, PG. Randomised controlled trial of azathioprine and 5-aminosalicylic acid for treatment of steroid dependent ulcerative colitis. Gut. (2006) 55:47–53. doi: 10.1136/gut.2005.068809
144. Ferretti, F, Cannatelli, R, Monico, MC, Maconi, G, and Ardizzone, S. An update on current Pharmacotherapeutic options for the treatment of ulcerative colitis. J Clin Med. (2022) 11:2302. doi: 10.3390/jcm11092302
145. Hanauer, SB, Sandborn, WJ, and Lichtenstein, GR. Evolving considerations for Thiopurine therapy for inflammatory bowel diseases-A clinical practice update: commentary. Gastroenterology. (2019) 156:36–42. doi: 10.1053/j.gastro.2018.08.043
146. Jharap, B, Seinen, ML, de Boer, NK, van Ginkel, JR, Linskens, RK, and Kneppelhout, JC. Thiopurine therapy in inflammatory bowel disease patients: analyses of two 8-year intercept cohorts. Inflamm Bowel Dis. (2010) 16:1541–9. doi: 10.1002/ibd.21221
147. Luber, RP, Honap, S, Cunningham, G, and Irving, PM. Can we predict the toxicity and response to thiopurines in iunfalmmatory bowel disease? Front Med. (2019) 6:279. doi: 10.3389/fmed.2019.00279
148. Chaparro, M, Ordás, I, Cabré, E, Garcia-Sanchez, V, Bastida, G, Peñalva, M, et al. Safety of thiopurine therapy in inflammatory bowel disease: long-term follow-up study of 3931 patients. Inflamm Bowel Dis. (2013) 19:1404–10. doi: 10.1097/MIB.0b013e318281f28f
149. Billioud, V, Sandborn, WJ, and Peyrin-Biroulet, L. Loss of response and need for adalimumab dose intensification in Crohn's disease: a systematic review. Am J Gastroenterol. (2011) 106:674–84. doi: 10.1038/ajg.2011.60
150. Papamichael, K, Gils, A, Rutgeerts, P, Levesque, BG, Vermeire, S, Sandborn, WJ, et al. Role for therapeutic drug monitoring during induction therapy with TNF antagonists in IBD: evolution in the definition and management of primary nonresponse. Inflamm Bowel Dis. (2015) 21:182–97. doi: 10.1097/MIB.0000000000000202
151. Feagan, BG, Rutgeerts, P, Sands, BE, Hanauer, S, Colombel, JF, Sandborn, WJ, et al. Vedolizumab as induction and maintenance therapy for ulcerative colitis. N Engl J Med. (2013) 369:699–710. doi: 10.1056/NEJMoa1215734
152. Sandborn, WJ, Feagan, BG, Rutgeerts, P, Hanauer, S, Colombel, JF, Sands, BE, et al. Vedolizumab as induction and maintenance therapy for Crohn's disease. N Engl J Med. (2013) 369:711–21. doi: 10.1056/NEJMoa1215739
153. Feagan, BG, Sandborn, WJ, Gasink, C, Jacobstein, D, Lang, Y, Friedman, JRP, et al. Ustekinumab as induction and maintenance therapy for Crohn's disease. N Engl J Med. (2016) 375:1946–60. doi: 10.1056/NEJMoa1602773
154. Sands, BE, Sandborn, WJ, Panaccione, R, O'Brien, CD, Zhang, H, Johanns, J, et al. Ustekinumab as induction and maintenance therapy for ulcerative colitis. N Engl J Med. (2019) 381:1201–14. doi: 10.1056/NEJMoa1900750
155. Gisbert, JP, and Chaparro, M. Predictors of primary response to biologic treatment [anti-TNF, Vedolizumab, and Ustekinumab] in patients with inflammatory bowel disease: from basic science to clinical practice. J Crohns Colitis. (2020) 14:694–14709. doi: 10.1093/ecco-jcc/jjz195
156. Inamdar, S, Volfson, A, Rosen, L, Sunday, S, Katz, S, and Sultan, K. Smoking and early infliximab response in Crohn’s disease: a meta-analysis. J Crohns Colitis. (2015) 9:140–9146. doi: 10.1093/ecco-jcc/jju018
157. Narula, N, and Fedorak, RN. Does smoking reduce infliximab's effectiveness against Crohn's disease? Can J Gastroenterol. (2009) 23:121–5. doi: 10.1155/2009/431349
158. Beltrán, B, Iborra, M, Sáez-González, E, Marqués-Miñana, MR, Moret, I, Cerrillo, E, et al. Fecal calprotectin pretreatment and induction infliximab levels for prediction of primary nonresponse to infliximab therapy in Crohn's disease. Dig Dis. (2019) 37:108–15. doi: 10.1159/000492626
159. Colombel, JF, Sandborn, WJ, Reinisch, W, Mantzaris, GJ, Kornbluth, A, Rachmilewitz, D, et al. Infliximab, azathioprine, or combination therapy for Crohn's disease. N Engl J Med. (2010) 362:1383–95. doi: 10.1056/NEJMoa0904492
160. Ho, GT, Lee, HM, Brydon, G, Ting, T, Hare, N, Drummond, H, et al. Fecal calprotectin predicts the clinical course of acute severe ulcerative colitis. Am J Gastroenterol. (2009) 104:673–8. doi: 10.1038/ajg.2008.119
161. Jürgens, M, Mahachie John, JM, Cleynen, I, Schnitzler, F, Fidder, H, van Moerkercke, W, et al. Levels of C-reactive protein are associated with response to infliximab therapy in patients with Crohn's disease. Clin Gastroenterol Hepatol. (2011) 9:421–7.e1. doi: 10.1016/j.cgh.2011.02.008
162. Louis, E, Vermeire, S, Rutgeerts, P, De Vos, M, Van Gossum, A, Pescatore, P, et al. A positive response to infliximab in Crohn disease: association with a higher systemic inflammation before treatment but not with −308 TNF gene polymorphism. Scand J Gastroenterol. (2002) 37:818–24. doi: 10.1080/gas.37.7.818.824
163. Angelison, L, Almer, S, Eriksson, A, Karling, P, Fagerberg, U, Halfvarson, J, et al. Swedish Organization for the Study of inflammatory bowel diseases (SOIBD). Long-term outcome of infliximab treatment in chronic active ulcerative colitis: a Swedish multicentre study of 250 patients. Aliment Pharmacol Ther. (2017) 45:519–32. doi: 10.1111/apt.13893
164. Beswick, L, Rosella, O, Rosella, G, Headon, B, Sparrow, MP, Gibson, PR, et al. Exploration of predictive biomarkers of early infliximab response in acute severe colitis: a prospective pilot study. J Crohns Colitis. (2018) 12:289–97. doi: 10.1093/ecco-jcc/jjx146
165. Dahlén, R, Magnusson, MK, Bajor, A, Lasson, A, Ung, KA, Strid, H, et al. Global mucosal and serum cytokine profile in patients with ulcerative colitis undergoing anti-TNF therapy. Scand J Gastroenterol. (2015) 50:1118–26. doi: 10.3109/00365521.2015.1031167
166. Louis, E, El Ghoul, Z, Vermeire, S, Dall'Ozzo, S, Rutgeerts, P, Paintaud, G, et al. Association between polymorphism in IgG fc receptor IIIa coding gene and biological response to infliximab in Crohn's disease. Aliment Pharmacol Ther. (2004) 19:511–9. doi: 10.1111/j.1365-2036.2004.01871.x
167. Sazonovs, A, Kennedy, NA, Moutsianas, L, Heap, GA, Rice, DL, Reppell, M, et al. PANTS consortium. HLA-DQA1*05 carriage associated with development of anti-drug antibodies to infliximab and adalimumab in patients with Crohn's disease. Gastroenterology. (2020) 158:189–99. doi: 10.1053/j.gastro.2019.09.041
168. A SPECIAL MEETING REVIEW EDITION: Highlights in anti-tumor necrosis factor monitoring and antibody monitoring from the 2014 DDW meeting: digestive disease week 2014 may 3-6, 2014 • Chicago, Illinois. Gastroenterol Hepatol (N Y). (2014) 10:1–20.
169. Gonczi, L, Vegh, Z, Golovics, PA, Rutka, M, Gecse, KB, Bor, R, et al. Prediction of short- and medium-term efficacy of biosimilar infliximab therapy. Do trough levels and antidrug antibody levels or clinical and biochemical markers play the more important role? J Crohns Colitis. (2017) 11:jjw203–705. doi: 10.1093/ecco-jcc/jjw203
170. Borren, NZ, Paulides, E, Frinack, JL, Olson, RN, Willrich, MAV, van der Woude, CJ, et al. Infliximab trough levels are not predictive of relapse in patients with IBD in endoscopic remission: a multicenter cohort study. Dig Dis Sci. (2021) 66:3548–54. doi: 10.1007/s10620-020-06645-0
171. Gaujoux, R, Starosvetsky, E, Maimon, N, Vallania, F, Bar-Yoseph, H, Pressman, S, et al. Cell-centred meta-analysis reveals baseline predictors of anti-TNFα non-response in biopsy and blood of patients with IBD. Gut. (2019) 68:604–14. doi: 10.1136/gutjnl-2017-315494
172. Verstockt, B, Verstockt, S, Dehairs, J, Ballet, V, Blevi, H, Wollants, WJ, et al. Low TREM1 expression in whole blood predicts anti-TNF response in inflammatory bowel disease. EBioMedicine. (2019) 40:733–42. doi: 10.1016/j.ebiom.2019.01.027
173. Bertani, L, Fornai, M, Fornili, M, Antonioli, L, Benvenuti, L, Tapete, G, et al. Serum oncostatin M at baseline predicts mucosal healing in Crohn's disease patients treated with infliximab. Aliment Pharmacol Ther. (2020) 52:284–91. doi: 10.1111/apt.15870
174. Minar, P, Lehn, C, Tsai, YT, Jackson, K, Rosen, MJ, and Denson, LA. Elevated pretreatment plasma Oncostatin M is associated with poor biochemical response to infliximab. Crohns Colitis. (2019) 1:otz026. doi: 10.1093/crocol/otz026
175. Atreya, R, Neumann, H, Neufert, C, Waldner, MJ, Billmeier, U, Zopf, Y, et al. In vivo imaging using fluorescent antibodies to tumor necrosis factor predicts therapeutic response in Crohn's disease. Nat Med. (2014) 20:313–8. doi: 10.1038/nm.3462
176. Rath, T, Bojarski, C, Neurath, MF, and Atreya, R. Molecular imaging of mucosal alpha4beta7 integrin expression with the fluorescent anti-adhesion antibody vedolizumab in Crohn's disease. Gastrointest Endosc. (2017) 86:406–8. doi: 10.1016/j.gie.2017.01.012
177. Iacucci, M, Jeffery, L, Acharjee, A, Grisan, E, Buda, A, Nardone, OM, et al. Computer-aided imaging analysis of probe-based confocal laser Endomicroscopy with molecular labeling and gene expression identifies markers of response to biological therapy in IBD patients: the Endo-omics study. Inflamm Bowel Dis. (2022):izac233. doi: 10.1093/ibd/izac233
178. Ananthakrishnan, AN, Luo, C, Yajnik, V, Khalili, H, Garber, JJ, Stevens, BW, et al. Gut microbiome function predicts response to anti-integrin biologic therapy in inflammatory bowel diseases. Cell Host Microbe. (2017) 21:603–610.e3. doi: 10.1016/j.chom.2017.04.010
179. Limdi, JK, and Farraye, FA. Real-time use of artificial intelligence at colonoscopy predicts relapse in ulcerative colitis: predicting with "intelligence". Gastrointest Endosc. (2022) 95:757–9. doi: 10.1016/j.gie.2021.12.048
180. Tontini, GE, Rimondi, A, Vernero, M, Neumann, H, Vecchi, M, Bezzio, C, et al. Artificial intelligence in gastrointestinal endoscopy for inflammatory bowel disease: a systematic review and new horizons. Ther Adv Gastroenterol. (2021) 14:17562848211017730. doi: 10.1177/17562848211017730
181. Iacucci, M, Smith, SCL, Bazarova, A, Shivaji, UN, Bhandari, P, Cannatelli, R, et al. An international multicenter real-life prospective study of electronic Chromoendoscopy score PICaSSO in ulcerative colitis. Gastroenterology. (2021) 160:1558–1569.e8. doi: 10.1053/j.gastro.2020.12.024
182. Nardone, OM, Bazarova, A, Bhandari, P, Cannatelli, R, Daperno, M, Ferraz, J, et al. PICaSSO virtual electronic chromendoscopy accurately reflects combined endoscopic and histological assessment for prediction of clinical outcomes in ulcerative colitis. United European. Gastroenterol J. (2022) 10:147–59. doi: 10.1002/ueg2.12185
183. Iacucci, M, Cannatelli, R, Parigi, TL, Buda, A, Labarile, N, and Nardone, OM. OP16 the first virtual chromoendoscopy artificial intelligence system to detect endoscopic and histologic remission in ulcerative colitis. J Crohn's Colitis. (2022) 16:i017–8. doi: 10.1093/ecco-jcc/jjab232.015
184. Villanacci, V, Parigi, TL, Del Amor, R, Mesguer Esbrì, P, Gui, X, Bazarova, A, et al. OP15 A new simplified histology artificial intelligence system for accurate assessment of remission in ulcerative colitis. J Crohn's Colitis. (2022) 16:i015–7. doi: 10.1093/ecco-jcc/jjab232.014
185. Bossuyt, P, Nakase, H, Vermeire, S, de Hertogh, G, Eelbode, T, Ferrante, M, et al. Automatic, computer-aided determination of endoscopic and histological inflammation in patients with mild to moderate ulcerative colitis based on red density. Gut. (2020) 69:1778–86. doi: 10.1136/gutjnl-2019-320056
186. Bossuyt, P, De Hertogh, G, Eelbode, T, Vermeire, S, and Bisschops, R. Computer-aided diagnosis with monochromatic light endoscopy for scoring histologic remission in ulcerative colitis. Gastroenterology. (2021) 160:23–5. doi: 10.1053/j.gastro.2020.09.053
187. Stidham, RW, Liu, W, Bishu, S, Rice, MD, Higgins, PDR, Zhu, J, et al. Performance of a deep learning model vs human reviewers in grading endoscopic disease severity of patients with ulcerative colitis. JAMA Netw Open. (2019) 2:e193963. doi: 10.1001/jamanetworkopen.2019.3963
188. Gottlieb, K, Requa, J, Karnes, W, Chandra Gudivada, R, Shen, J, Rael, E, et al. Central Reading of ulcerative colitis clinical trial videos using neural networks. Gastroenterology. (2021) 160:710–719.e2. doi: 10.1053/j.gastro.2020.10.024
189. Yao, H, Stidham, RW, Soroushmehr, R, Gryak, J, and Najarian, K. Automated detection of non-informative frames for colonoscopy through a combination of deep learning and feature extraction. Annu Int Conf IEEE Eng Med Biol Soc. (2019) 2019:2402–6. doi: 10.1109/EMBC.2019.8856625
190. Takenaka, K, Ohtsuka, K, Fujii, T, Oshima, S, Okamoto, R, and Watanabe, M. Deep neural network accurately predicts prognosis of ulcerative colitis using endoscopic images. Gastroenterology. (2021) 160:2175–2177.e3. doi: 10.1053/j.gastro.2021.01.210
191. Kabir, M, Fofaria, R, Arebi, N, Bassett, P, Tozer, PJ, Hart, AL, et al. Systematic review with meta-analysis: IBD-associated colonic dysplasia prognosis in the videoendoscopic era (1990 to present). Aliment Pharmacol Ther. (2020) 52:5–19. doi: 10.1111/apt.15778
192. Iacucci, M, Cannatelli, R, Tontini, GE, Panaccione, R, Danese, S, et al. Improving the quality of surveillance colonoscopy in inflammatory bowel disease. Lancet Gastroenterol Hepatol. (2019) 4:971–83. doi: 10.1016/S2468-1253(19)30194-3
193. Maeda, Y, Kudo, SE, Ogata, N, Kuroki, T, Takashina, Y, Takishima, K, et al. Use of advanced endoscopic technology for optical characterization of neoplasia in patients with ulcerative colitis: a systematic review. Dig Endosc. (2022) 34:1297–310. doi: 10.1111/den.14335
194. Murthy, SK, Feuerstein, JD, Nguyen, GC, and Velayos, FS. AGA clinical practice update on endoscopic surveillance and Management of Colorectal Dysplasia in inflammatory bowel diseases: expert review. Gastroenterology. (2021) 161:1043–1051.e4. doi: 10.1053/j.gastro.2021.05.063
195. Maeda, Y, Kudo, SE, Ogata, N, Misawa, M, Mori, Y, Mori, K, et al. Can artificial intelligence help to detect dysplasia in patients with ulcerative colitis? Endoscopy. (2021) 53:E273–4. doi: 10.1055/a-1261-2944
196. Rahier, JF, Magro, F, Abreu, C, Armuzzi, A, Ben-Horin, S, Chowers, Y, et al. Second European evidence-based consensus on the prevention, diagnosis and management of opportunistic infections in inflammatory bowel disease. J Crohns Colitis. (2014) 8:443–68. doi: 10.1016/j.crohns.2013.12.013
197. Mazzola, G, Macaluso, FS, Adamoli, L, Renna, S, Cascio, A, and Orlando, A. Diagnostic and vaccine strategies to prevent infections in patients with inflammatory bowel disease. J Infect. (2017) 74:433–41. doi: 10.1016/j.jinf.2017.02.009
198. Rubin, LG, Levin, MJ, Ljungman, P, Davies, EG, Avery, R, Tomblyn, M, et al. IDSA clinical practice guideline for vaccination of the immunocompromised host. Clin Infect Dis. (2013) 58:e44–e100. doi: 10.1093/cid/cit684
199. Cottone, M, Kohn, A, Daperno, M, Armuzzi, A, Guidi, L, D'Inca, R, et al. Advanced age is an independent risk factor for severe infections and mortality in patients given anti-tumor necrosis factor therapy for inflammatory bowel disease. Clin Gastroenterol Hepatol. (2011) 9:30–5. doi: 10.1016/j.cgh.2010.09.026
200. Toruner, M, Loftus, EVJr, Harmsen, WS, Zinsmeister, AR, Orenstein, R, Sandborn, WJ, et al. Risk factors for opportunistic infections in patients with inflammatory bowel disease. Gastroenterology. (2008) 134:929–36. doi: 10.1053/j.gastro.2008.01.012
201. Lichtenstein, GR, Feagan, BG, Cohen, RD, Salzberg, BA, Diamond, RH, Price, S, et al. Serious infection and mortality in patients with Crohn's disease: more than 5 years of follow-up in the TREAT™ registry. Am J Gastroenterol. (2012) 107:1409–22. doi: 10.1038/ajg.2012.218
202. Colombel, JF, Sands, BE, Rutgeerts, P, Sandborn, W, Danese, S, and D'Haens, G. The safety of vedolizumab for ulcerative colitis and Crohn's disease. Gut. (2017) 66:839–51. doi: 10.1136/gutjnl-2015-311079
203. MacDonald, JK, Nguyen, TM, Khanna, R, and Timmer, A. Anti-IL-12/23p40 antibodies for induction of remission in Crohn's disease. Cochrane Database Syst Rev. (2016) 2016:CD007572. doi: 10.1002/14651858.CD007572.pub3
204. Winthrop, KL, Melmed, GY, Vermeire, S, Long, MD, Chan, G, Pedersen, RD, et al. Herpes zoster infection in patients with ulcerative colitis receiving Tofacitinib. Inflamm Bowel Dis. (2018) 24:2258–65. doi: 10.1093/ibd/izy131
205. Guillo, L, Rabaud, C, Choy, EH, D'Amico, F, Danese, S, Ng, SC, et al. Herpes zoster and vaccination strategies in inflammatory bowel diseases: a practical guide. Clin Gastroenterol Hepatol. (2022) 20:481–90. doi: 10.1016/j.cgh.2020.10.027
206. European Association For The Study Of The Liver. EASL clinical practice guidelines: management of chronic hepatitis B virus infection. J Hepatol. (2012) 57:167–85. doi: 10.1016/j.jhep.2012.02.010
207. Aberra, FN, Stettler, N, Brensinger, C, Lichtenstein, GR, and Lewis, JD. Risk for active tuberculosis in inflammatory bowel disease patients. Clin Gastroenterol Hepatol. (2007) 5:1070–1075.e1. doi: 10.1016/j.cgh.2007.04.007
208. Beaugerie, L, Brousse, N, Bouvier, AM, Colombel, JF, Lémann, M, Cosnes, J, et al. Lymphoproliferative disorders in patients receiving thiopurines for inflammatory bowel disease: a prospective observational cohort study. Lancet. (2009) 374:1617–25. doi: 10.1016/S0140-6736(09)61302-7
209. Sokol, H, Beaugerie, L, Maynadié, M, Laharie, D, Dupas, JL, Flourié, B, et al. Excess primary intestinal lymphoproliferative disorders in patients with inflammatory bowel disease. Inflamm Bowel Dis. (2012) 18:2063–71. doi: 10.1002/ibd.22889
210. Kane, S, Khatibi, B, and Reddy, D. Higher incidence of abnormal pap smears in women with inflammatory bowel disease. Am J Gastroenterol. (2008) 103:631–6. doi: 10.1111/j.1572-0241.2007.01582.x
211. Harrison, LH. Epidemiological profile of meningococcal disease in the United States. Clin Infect Dis. (2010) 50:S37–44. doi: 10.1086/648963
212. Cohn, AC, MacNeil, JR, Clark, TA, Ortega-Sanchez, IR, Briere, EZ, Meissner, HC, et al. Prevention and control of meningococcal disease: recommendations of the advisory committee on immunization practices (ACIP). MMWR Recomm Rep. (2013) 22:1–28.
213. De León-Rendón, JL, Hurtado-Salazar, C, and Yamamoto-Furusho, JK. Aspects of inflammatory bowel disease during the COVID-19 pandemic and general considerations. Rev Gastroenterol Mex (Engl Ed). (2020) 85:295–302. doi: 10.1016/j.rgmx.2020.05.001
214. Siegel, CA, Melmed, GY, McGovern, DP, Rai, V, Krammer, F, Rubin, DT, et al. SARS-CoV-2 vaccination for patients with inflammatory bowel diseases: recommendations from an international consensus meeting. Gut. (2021) 70:635–40. doi: 10.1136/gutjnl-2020-324000
215. Macaluso, FS, Liguori, G, and Galli, M. Vaccinations in patients with inflammatory bowel disease. Dig Liver Dis. (2021) 53:1539–45. doi: 10.1016/j.dld.2021.05.015
216. Frontali, A, Cohen, L, Bridoux, V, Myrelid, P, Sica, G, Poggioli, G, et al. Segmental colectomy for ulcerative colitis: is there a place in selected patients without active colitis? An international multicentric retrospective study in 72 patients. J Crohns Colitis. (2020) 14:1687–92. doi: 10.1093/ecco-jcc/jjaa107
217. Øresland, T, Bemelman, WA, Sampietro, GM, Spinelli, A, Windsor, A, Ferrante, M, et al. European evidence based consensus on surgery for ulcerative colitis. J Crohns Colitis. (2015) 9:4–25. doi: 10.1016/j.crohns.2014.08.012
218. Pellino, G, Keller, DS, Sampietro, GM, Carvello, M, Celentano, V, Coco, C, et al. Inflammatory bowel disease position statement of the Italian Society of Colorectal Surgery (SICCR): ulcerative colitis. Tech Coloproctol. (2020) 24:397–419. doi: 10.1007/s10151-020-02175-z
219. Colombo, F, Sahami, S, De Buck Van Overstraeten, A, Tulchinsky, H, Mege, D, Dotan, I, et al. Restorative Proctocolectomy in elderly IBD patients: a multicentre comparative study on safety and efficacy. J Crohns Colitis. (2017) 11:671–9. doi: 10.1093/ecco-jcc/jjw209
220. Sampietro, GM, Colombo, F, and Corsi, F. Sequential approach for a critical-view COlectomy (SACCO): a laparoscopic technique to reduce operative time and complications in IBD acute severe colitis. J Clin Med. (2020) 9:3382. doi: 10.3390/jcm9103382
221. Frontali, A, Colombo, F, and Sampietro, GM. Acute severe ulcerative colitis: how to reduce operating time under 150 min for emergency laparoscopic colectomy – a video vignette. Color Dis. (2021) 23:3280–1. doi: 10.1111/codi.15928
222. Bartels, SA, Dhoore, A, Cuesta, MA, Bensdorp, AJ, Lucas, C, and Bemelman, WA. Significantly increased pregnancy rates after laparoscopic restorative proctocolectomy: a cross-sectional study. Ann Surg. (2012) 256:1045–8. doi: 10.1097/SLA.0b013e318250caa9
223. Bemelman, WA, Warusavitarne, J, Sampietro, GM, Serclova, Z, Zmora, O, Luglio, G, et al. ECCO-ESCP consensus on surgery for Crohn's disease. J Crohns Colitis. (2018) 12:1–16. doi: 10.1093/ecco-jcc/jjx061
224. Cristaldi, M, Sampietro, GM, Danelli, PG, Bollani, S, Bianchi Porro, G, and Taschieri, AM. Long-term results and multivariate analysis of prognostic factors in 138 consecutive patients operated on for Crohn's disease using "bowel-sparing" techniques. Am J Surg. (2000) 179:266–70. doi: 10.1016/s0002-9610(00)00334-2
225. Parente, F, Sampietro, GM, Molteni, M, Greco, S, Anderloni, A, Sposito, C, et al. Behaviour of the bowel wall during the first year after surgery is a strong predictor of symptomatic recurrence of Crohn's disease: a prospective study. Aliment Pharmacol Ther. (2004) 20:959–68. doi: 10.1111/j.1365-2036.2004.02245.x
226. Sampietro, GM, Cristaldi, M, Maconi, G, Parente, F, Sartani, A, Ardizzone, S, et al. A prospective, longitudinal study of nonconventional strictureplasty in Crohn's disease. J Am Coll Surg. (2004) 199:8–20. doi: 10.1016/j.jamcollsurg.2004.01.039
227. Maconi, G, Colombo, E, Sampietro, GM, Lamboglia, F, D'Incà, R, and Daperno, M. CARD15 gene variants and risk of reoperation in Crohn's disease patients. Am J Gastroenterol. (2009) 104:2483–91. doi: 10.1038/ajg.2009.413
228. Sampietro, GM, Corsi, F, Maconi, G, Ardizzone, S, Frontali, A, Corona, A, et al. Prospective study of long-term results and prognostic factors after conservative surgery for small bowel Crohn's disease. Clin Gastroenterol Hepatol. (2009) 7:183–91. doi: 10.1016/j.cgh.2008.10.008
229. Rutgeerts, P, Geboes, K, Vantrappen, G, Beyls, J, Kerremans, R, and Hiele, M. Predictability of the postoperative course of Crohn's disease. Gastroenterology. (1990) 99:956–63. doi: 10.1016/0016-5085(90)90613-6
230. Kono, T, Nomura, M, Kasai, S, and Kohgo, Y. Effect of ecabet sodium enema on mildly to moderately active ulcerative proctosigmoiditis: an open-label study. Am J Gastroenterol. (2001) 96:793–7. doi: 10.1111/j.1572-0241.2001.03624.x
231. Luglio, G, Rispo, A, Imperatore, N, Giglio, MC, Amendola, A, Tropeano, FP, et al. Surgical prevention of anastomotic recurrence by excluding mesentery in Crohn's disease: the SuPREMe-CD study – A randomized clinical trial. Ann Surg. (2020) 272:210–7. doi: 10.1097/SLA.0000000000003821
232. Sampietro, GM, Maconi, G, Colombo, F, Dilillo, D, Fiorina, P, D'Addio, F, et al. Prevalence and significance of mesentery thickening and lymph nodes enlargement in Crohn's disease. Dig Liver Dis. (2022) 54:490–9. doi: 10.1016/j.dld.2021.06.030
233. Mineccia, M, Maconi, G, Daperno, M, Cigognini, M, Cherubini, V, Colombo, F, et al. Has the removing of the mesentery during Ileo-colic resection an impact on post-operative complications and recurrence in Crohn's disease? Results from the resection of the mesentery study (remedy). J Clin Med. (2022) 11:1961. doi: 10.3390/jcm11071961
234. Coffey, CJ, Kiernan, MG, Sahebally, SM, Jarrar, A, Burke, JP, Kiely, PA, et al. Inclusion of the mesentery in ileocolic resection for Crohn's disease is associated with reduced surgical recurrence. J Crohns Colitis. (2018) 12:1139–50. doi: 10.1093/ecco-jcc/jjx187
235. Plevris, N, and Lees, CW. Disease monitoring in inflammatory bowel disease: evolving principles and possibilities. Gastroenterology. (2022) 162:1456–1475.e1. doi: 10.1053/j.gastro.2022.01.024
236. D'Haens, G, Kelly, O, Battat, R, Silverberg, MS, Laharie, D, Louis, E, et al. Development and validation of a test to monitor endoscopic activity in patients with Crohn's disease based on serum levels of proteins. Gastroenterology. (2020) 158:515–526.e10. doi: 10.1053/j.gastro.2019.10.034
237. Facchin, S, Buda, A, Cardin, R, Agbariah, N, Zingone, F, de Bona, M, et al. Rapid point-of-care anti-infliximab antibodies detection in clinical practice: comparison with ELISA and potential for improving therapeutic drug monitoring in IBD patients. Ther Adv Gastroenterol. (2021) 14:1756284821999902. doi: 10.1177/1756284821999902
238. Lasa, JS, Olivera, PA, Danese, S, and Peyrin-Biroulet, L. Efficacy and safety of biologics and small molecule drugs for patients with moderate-to-severe ulcerative colitis: a systematic review and network meta-analysis. Lancet Gastroenterol Hepatol. (2022) 7:161–70. doi: 10.1016/S2468-1253(21)00377-0
239. Singh, S, Murad, MH, Fumery, M, Sedano, R, Jairath, V, Panaccione, R, et al. Comparative efficacy and safety of biologic therapies for moderate-to-severe Crohn's disease: a systematic review and network meta-analysis. Lancet Gastroenterol Hepatol. (2021) 6:1002–14. doi: 10.1016/S2468-1253(21)00312-5
240. Ben-Horin, S, Novack, L, Mao, R, Guo, J, Zhao, Y, Sergienko, R, et al. Efficacy of biologic drugs in short-duration versus Long-duration inflammatory bowel disease: a systematic review and an individual-patient data meta-analysis of randomized controlled trials. Gastroenterology. (2022) 162:482–94. doi: 10.1053/j.gastro.2021.10.037
241. Torres, J, Caprioli, F, Katsanos, KH, Lobatón, T, Micic, D, Zerôncio, M, et al. Predicting outcomes to optimize disease Management in Inflammatory Bowel Diseases. J Crohns Colitis. (2016) 10:1385–94. doi: 10.1093/ecco-jcc/jjw116
242. Mao, EJ, Hazlewood, GS, Kaplan, GG, Peyrin-Biroulet, L, and Ananthakrishnan, AN. Systematic review with meta-analysis: comparative efficacy of immunosuppressants and biologics for reducing hospitalisation and surgery in Crohn's disease and ulcerative colitis. Aliment Pharmacol Ther. (2017) 45:3–13. doi: 10.1111/apt.13847
243. Barberio, B, Zamani, M, Black, CJ, Savarino, EV, and Ford, AC. Prevalence of symptoms of anxiety and depression in patients with inflammatory bowel disease: a systematic review and meta-analysis. Lancet Gastroenterol Hepatol. (2021) 6:359–70. doi: 10.1016/S2468-1253(21)00014-5
244. Marinelli, C, Zingone, F, Inferrera, M, Lorenzon, G, Rigo, A, Facchin, S, et al. Factors associated with disability in patients with ulcerative colitis: a cross-sectional study. J Dig Dis. (2020) 21:81–7. doi: 10.1111/1751-2980.12837
245. Verstockt, B, Parkes, M, and Lee, JC. How do we predict a Patient's disease course and whether they will respond to specific treatments? Gastroenterology. (2022) 162:1383–95. doi: 10.1053/j.gastro.2021.12.245
246. Verstockt, B, Noor, NM, Marigorta, UM, Pavlidis, P, Deepak, P, and Ungaro, RC. Scientific workshop steering committee. Results of the seventh scientific workshop of ECCO: precision medicine in IBD-disease outcome and response to therapy. J Crohns Colitis. (2021) 15:1431–42. doi: 10.1093/ecco-jcc/jjab050
247. D'Haens, G, Baert, F, van Assche, G, Caenepeel, P, Vergauwe, P, Tuynman, H, et al. Belgian inflammatory bowel disease research group; North-Holland gut Club. Early combined immunosuppression or conventional management in patients with newly diagnosed Crohn's disease: an open randomised trial. Lancet. (2008) 371:660–7. doi: 10.1016/S0140-6736(08)60304-9
248. Hamdeh, S, Aziz, M, Altayar, O, Olyaee, M, Murad, MH, and Hanauer, SB. Early vs late use of anti-TNFa therapy in adult patients with Crohn disease: a systematic review and meta-analysis. Inflamm Bowel Dis. (2020) 26:1808–18. doi: 10.1093/ibd/izaa031
249. Bodini, G, Giannini, EG, De Maria, C, Dulbecco, P, Furnari, M, Marabotto, E, et al. Anti-TNF therapy is able to stabilize bowel damage progression in patients with Crohn's disease. A study performed using the Lémann index. Dig Liver Dis. (2017) 49:175–80. doi: 10.1016/j.dld.2016.10.014
250. Singh, S, Stitt, LW, Zou, G, Khanna, R, Dulai, PS, Sandborn, WJ, et al. Early combined immunosuppression may be effective and safe in older patients with Crohn's disease: post hoc analysis of REACT. Aliment Pharmacol Ther. (2019) 49:1188–94. doi: 10.1111/apt.15214
251. Raine, T, and Danese, S. Breaking through the therapeutic ceiling: what will it take? Gastroenterology. (2022) 162:1507–11. doi: 10.1053/j.gastro.2021.09.078
252. Colombel, JF, Panaccione, R, Bossuyt, P, Lukas, M, Baert, F, Vaňásek, T, et al. Effect of tight control management on Crohn's disease (CALM): a multicentre, randomised, controlled phase 3 trial. Lancet. (2017) 390:2779–89. doi: 10.1016/S0140-6736(17)32641-7
253. Irving, PM, Sands, BE, Hoops, T, Izanec, JL, Gao, LL, Gasink, C, et al. OP02 Ustekinumab versus adalimumab for induction and maintenance therapy in moderate-to-severe Crohn’s disease: the SEAVUE study. J Crohn's Colitis. (2021) 15:S001–2. doi: 10.1093/ecco-jcc/jjab075.001
254. Sands, BE, Peyrin-Biroulet, L, Loftus, EVJr, Danese, S, Colombel, JF, Törüner, M, et al. Vedolizumab versus Adalimumab for Moderate-to-Severe Ulcerative Colitis. N Engl J Med. (2019) 381:1215–26. doi: 10.1056/NEJMoa1905725
255. Ahmed, W, Galati, J, Kumar, A, Christos, PJ, Longman, R, Lukin, DJ, et al. Dual biologic or small molecule therapy for treatment of inflammatory bowel disease: a systematic review and meta-analysis. Clin Gastroenterol Hepatol. (2022) 20:e361–79. doi: 10.1016/j.cgh.2021.03.034
256. Molendijk, I, Nuij, VJ, van der Meulen-de Jong, AE, and van der Woude, CJ. Disappointing durable remission rates in complex Crohn's disease fistula. Inflamm Bowel Dis. (2014) 20:2022–8. doi: 10.1097/MIB.0000000000000148
257. Barberio, B, Black, CJ, Savarino, EV, and Ford, AC. Ciclosporin or infliximab as rescue therapy in acute Glucorticosteroid-refractory ulcerative colitis: systematic review and network meta-analysis. J Crohns Colitis. (2021) 15:733–41. doi: 10.1093/ecco-jcc/jjaa226
258. Pineton de Chambrun, G, Amiot, A, Bouguen, G, Viennot, S, Altwegg, R, Louis, E, et al. PROTECT-GETAID study group. Efficacy of tumor necrosis factor antagonist treatment in patients with refractory ulcerative Proctitis. Clin Gastroenterol Hepatol. (2020) 18:620–627.e1. doi: 10.1016/j.cgh.2019.05.060
259. Beelen, EMJ, Nieboer, D, Arkenbosch, JHC, Regueiro, MD, Satsangi, J, Ardizzone, S, et al. Risk prediction and comparative efficacy of anti-TNF vs Thiopurines, for preventing postoperative recurrence in Crohn's disease: a pooled analysis of 6 trials. Clin Gastroenterol Hepatol. (2021) 20:2741–2752.e6. doi: 10.1016/j.cgh.2021.10.021
260. Ananthakrishnan, AN, Donaldson, T, Lasch, K, and Yajnik, V. Management of Inflammatory Bowel Disease in the elderly patient: challenges and opportunities. Inflamm Bowel Dis. (2017) 23:882–93. doi: 10.1097/MIB.0000000000001099
261. Revés, J, Ungaro, RC, and Torres, J. Unmet needs in inflammatory bowel disease. Curr Res Pharmacol Drug Discov. (2021) 2:100070. doi: 10.1016/j.crphar.2021.100070
262. Bzdok, D, Altman, N, and Krzywinski, M. Statistics versus machine learning. Nat Methods. (2018) 15:233–4. doi: 10.1038/nmeth.4642
263. Le Berre, C, Sandborn, WJ, Aridhi, S, Devignes, MD, Fournier, L, Smaïl-Tabbone, M, et al. Application of artificial intelligence to gastroenterology and hepatology. Gastroenterology. (2020) 158:76–94.e2. doi: 10.1053/j.gastro.2019.08.058
Keywords: clinical complexity, Crohn’s disease, internal medicine, precision medicine, ulcerative colitis
Citation: Lenti MV, Scribano ML, Biancone L, Ciccocioppo R, Pugliese D, Pastorelli L, Fiorino G, Savarino E, Caprioli FA, Ardizzone S, Fantini MC, Tontini GE, Orlando A, Sampietro GM, Sturniolo GC, Monteleone G, Vecchi M, Kohn A, Daperno M, D’Incà R, Corazza GR and Di Sabatino A (2023) Personalize, participate, predict, and prevent: 4Ps in inflammatory bowel disease. Front. Med. 10:1031998. doi: 10.3389/fmed.2023.1031998
Edited by:
Azita Hekmatdoost, National Nutrition and Food Technology Research Institute, IranReviewed by:
Sudabeh Alatab, Tehran University of Medical Sciences, IranMarietta Iacucci, University College Cork, Ireland
Giulia Roda, Humanitas University, Italy
Copyright © 2023 Lenti, Scribano, Biancone, Ciccocioppo, Pugliese, Pastorelli, Fiorino, Savarino, Caprioli, Ardizzone, Fantini, Tontini, Orlando, Sampietro, Sturniolo, Monteleone, Vecchi, Kohn, Daperno, D’Incà, Corazza and Di Sabatino. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Antonio Di Sabatino, a.disabatino@smatteo.pv.it
 Marco Vincenzo Lenti
Marco Vincenzo Lenti Maria Lia Scribano
Maria Lia Scribano Livia Biancone
Livia Biancone Rachele Ciccocioppo
Rachele Ciccocioppo Daniela Pugliese6
Daniela Pugliese6  Gionata Fiorino
Gionata Fiorino Edoardo Savarino
Edoardo Savarino Flavio Andrea Caprioli
Flavio Andrea Caprioli Ambrogio Orlando
Ambrogio Orlando Giovanni Monteleone
Giovanni Monteleone Maurizio Vecchi
Maurizio Vecchi Marco Daperno
Marco Daperno Renata D’Incà
Renata D’Incà Gino Roberto Corazza
Gino Roberto Corazza Antonio Di Sabatino
Antonio Di Sabatino