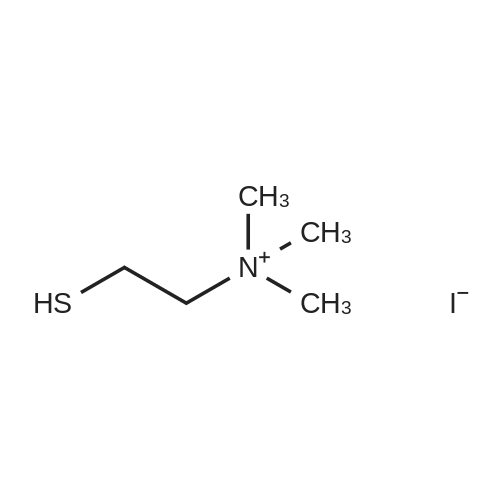
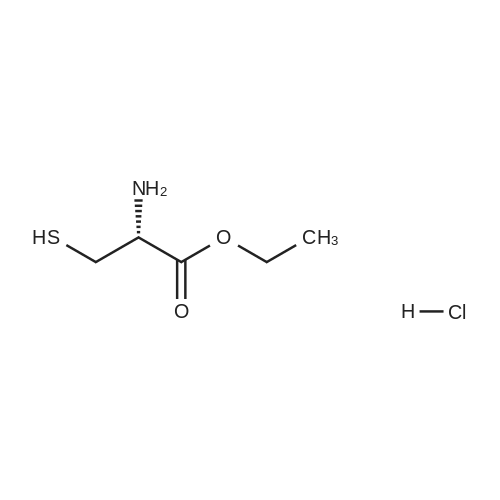
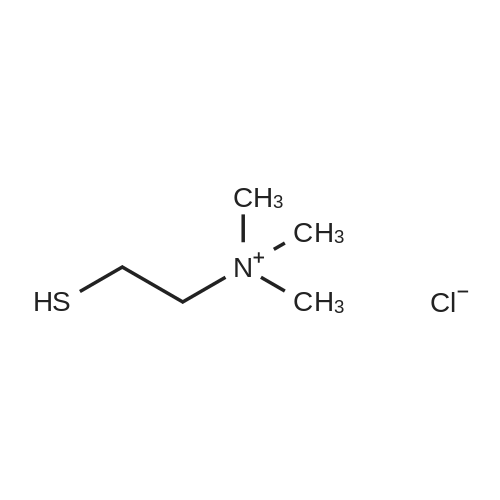
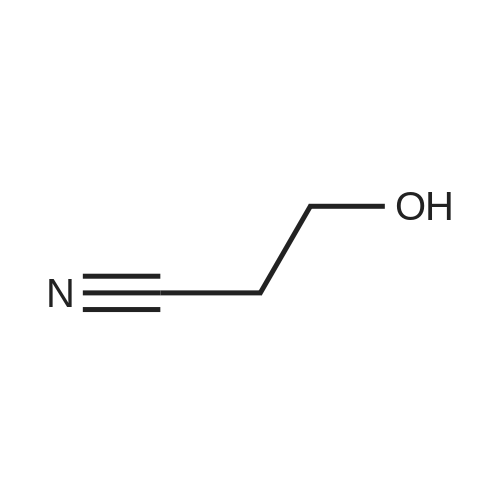
|
In 1,4-dioxane; methanol; ethanol; chloroform; |
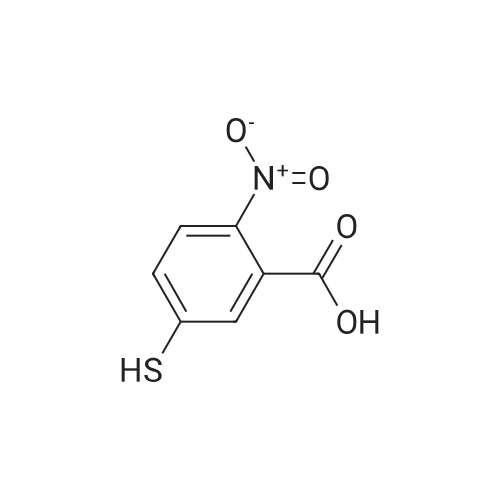
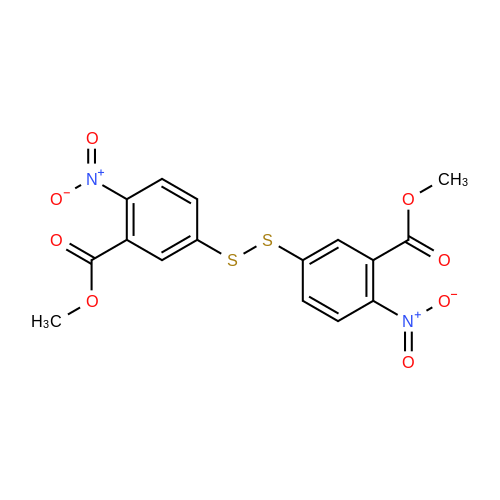
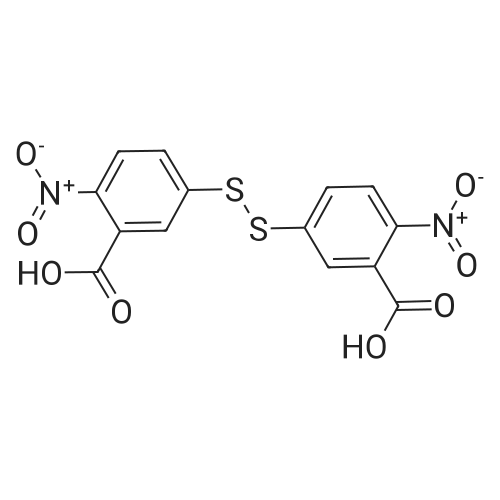
Synthesis of Activated Disulfide Containing Co-monomers Synthesis of 5,5'-dithiobis(2-nitrobenzoate)propionitrile: <strong>[69-78-3]5,5'-dithiobis(2-nitrobenzoic acid)</strong> [Ellman's reagent] (500 mg, 1.26 mmol) was dissolved in 4.0 ml dioxane. Dicylohexylcarbodiimide (540 mg, 2.6 mmol) and 3-hydroxypropionitrile (240 muL, 188 mg, 2.60 mmol) were added. The reaction mixture was stirred overnight at room temperature. The urea precipitate was removed by centrifugation. The dioxane was removed on rotary evaporator. The residue was washed with saturated bicarbonate, water, and brine; and dried over magnesium sulfate. Solvent removal yielded 696 mg yellow/orange foam. The residue was purified using normal phase HPLC (Alltech econosil, 250*22 nm), flow rate=9.0 ml/min, mobile phase=1% ethanol in chloroform, retention time=13 min. Removal of solvent afforded 233 mg (36.8%) product as a yellow oil. TLC (silica: 5% methanol in chloroform; rf=0.51). H1 NMR delta8.05 (d, 4 H), 7.75 (m, 4H), 4.55 (t, 4H), 2.85 (t, 4H). |
|
In 1,4-dioxane; methanol; ethanol; chloroform; |
Synthesis of Activated Disulfide Containing Co-monomers Synthesis of 5,5'-dithiobis(2-nitrobenzoate)propionitrile: <strong>[69-78-3]5,5'-dithiobis(2-nitrobenzoic acid)</strong> [Ellman's reagent] (500 mg, 1.26 mmol) was dissolved in 4.0 ml dioxane. Dicylohexylcarbodiimide (540 mg, 2.6 mmol) and 3-hydroxypropionitrile (240 muL, 188 mg, 2.60 mmol) were added. The reaction mixture was stirred overnight at room temperature. The urea precipitate was removed by centrifugation. The dioxane was removed on rotary evaporator. The residue was washed with saturated bicarbonate, water, and brine; and dried over magnesium sulfate. Solvent removal yielded 696 mg yellow/orange foam. The residue was purified using normal phase HPLC (Alltech econosil, 250*22 mn), flow rate=9.0 ml/min, mobile phase=1% ethanol in chloroform, retention time=13 min. Removal of solvent afforded 233 mg (36.8%) product as a yellow oil. TLC (silica: 5% methanol in chloroform; rf=0.51). H1 NMR delta8.05 (d, 4 H), 7.75 (m, 4H), 4.55 (t, 4H), 2.85 (t, 4H). |
|
In 1,4-dioxane; methanol; ethanol; chloroform; |
Synthesis of Activated Disulfide Containing Co-monomers Synthesis of 5,5'-dithiobis(2-nitrobenzoate)propionitrile: <strong>[69-78-3]5,5'-dithiobis(2-nitrobenzoic acid)</strong> [Ellman's reagent] (500 mg,1.26 mmol) was dissolved in 4.0 ml dioxane. Dicylohexylcarbodiimide (540 mg, 2.6 mmol) and 3-hydroxypropionitrile (240 muL, 188 mg, 2.60 mmol) were added. The reaction mixture was stirred overnight at room temperature. The urea precipitate was removed by centrifugation. The dioxane was removed on rotary evaporator. The residue was washed with saturated bicarbonate, water, and brine; and dried over magnesium sulfate. Solvent removal yielded 696 mg yellow/orange foam. The residue was purified using normal phase HPLC (Alltech econosil, 250*22 nm), flow rate=9.0 ml/min, mobile phase=1% ethanol in chloroform, retention time=13 min. Removal of solvent afforded 233 mg (36.8%) product as a yellow oil. TLC (silica: 5% methanol in chloroform; rf=0.51). H1NMR ? 8.05 (d, 4H), 7.75 (m, 4H), 4.55 (t, 4H), 2.85 (t, 4H). |
|
In 1,4-dioxane; ethanol; chloroform; |
Synthesis of 5,5'-dithiobis(2-nitrobenzoate)propionitrile <strong>[69-78-3]5,5'-dithiobis(2-nitrobenzoic acid)</strong> [Ellman's reagent] (500 mg,1.26 mmol) was dissolved in 4.0 ml dioxane. Dicylohexylcarbodiimide (540 mg, 2.6 mmol) and 3-hydroxypropionitrile (240 muL, 188 mg, 2.60 mmol) were added. The reaction mixture was stirred overnight at RT. The urea precipitate was removed by centrifugation. The dioxane was removed on rotary evaporator. The residue was washed with saturated bicarbonate, water, and brine; and dried over magnesium sulfate. Solvent removal yielded 696 mg yellow/orange foam. The residue was purified using normal phase HPLC (Alltech econosil, 250*22 nm), flow rate=9.0 ml/min, mobile phase=1% ethanol in chloroform, retention time=13 min. Removal of solvent afforded 233 mg (36.8%) product as a yellow oil. TLC (silica: 5% methanol in chloroform; rf=0.51). H1 NMR ?8.05 (d, 4 H), 7.75 (m, 4H), 4.55 (t, 4H), 2.85 (t, 4H). |
| 233 mg (36.8%) |
In 1,4-dioxane; ethanol; chloroform; |
Example 11 Synthesis of 5,5'-Dithiobis(2-nitrobenzoate)propionitrile <strong>[69-78-3]5,5'-dithiobis(2-nitrobenzoic acid)</strong> (500 mg, 1.26 mmol, Aldrich Chemical Company) was taken up in 4.0 mL dioxane. Dicylohexylcarbodiimide (540 mg, 2.6 mmol, Aldrich Chemical Company) and 3-hydroxypropionitrile (240 muL, 188 mg, 2.60 mmol, Aldrich Chemical Company) were added. The reaction mixture was stirred overnight at room temperature. The precipitate was removed by centrifugation, and the solvent concentrated under reduced pressure. The residue was washed with saturated sodium bicarbonate, water, and brine; and dried over magnesium sulfate. Solvent removal (aspirator) yielded 696 mg yellow/orange foam. The residue was purified using normal phase HPLC (Alltech econosil, 250*22 nm), flow rate=9.0 mL/min, mobile phase=1% ethanol in chloroform, retention time=13 min. Removal of solvent (aspirator) afforded 233 mg (36.8%) of 5,5'-dithiobis(2-nitrobenzoate)propionitrile as a yellow oil. TLC (silica: 5% methanol in chloroform; Rf=0.51). H1NMR ? 8.05 (d, 4 H), 7.75 (m, 4H), 4.55 (t, 4H), 2.85 (t, 4H). |
|
In 1,4-dioxane; ethanol; chloroform; |
Synthesis of 5,5'-dithiobis(2-nitrobenzoate)propionitrile: <strong>[69-78-3]5,5'-dithiobis(2-nitrobenzoic acid)</strong> [Ellman's reagent] (500 mg,1.26 mmol) was dissolved in 4.0 ml dioxane. Dicylohexylcarbodiimide (540 mg, 2.6 mmol) and 3-hydroxypropionitrile (240 muL, 188 mg, 2.60 mmol) were added. The reaction mixture was stirred overnight at room temperature. The urea precipitate was removed by centrifugation. The dioxane was removed on rotary evaporator. The residue was washed with saturated bicarbonate, water, and brine; and dried over magnesium sulfate. Solvent removal yielded 696 mg yellow/orange foam. The residue was purified using normal phase HPLC (Alltech econosil, 250*22 nm), flow rate=9.0 mlmin, mobile phase=1% ethanol in chloroform, retention time=13 min. Removal of solvent afforded 233 mg (36.8%) product as a yellow oil. TLC (silica: 5% methanol in chloroform; rf=0.51). H1NMR ?8.05 (d, 4 H), 7.75 (m, 4H), 4.55 (t, 4H), 2.85 (t, 4H). |

 Chemistry
Chemistry
 Pharmaceutical Intermediates
Pharmaceutical Intermediates
 Inhibitors/Agonists
Inhibitors/Agonists
 Material Science
Material Science














 For Research Only
For Research Only
 110K+ Compounds
110K+ Compounds
 Competitive Price
Competitive Price
 1-2 Day Shipping
1-2 Day Shipping