| 85.6% |
|
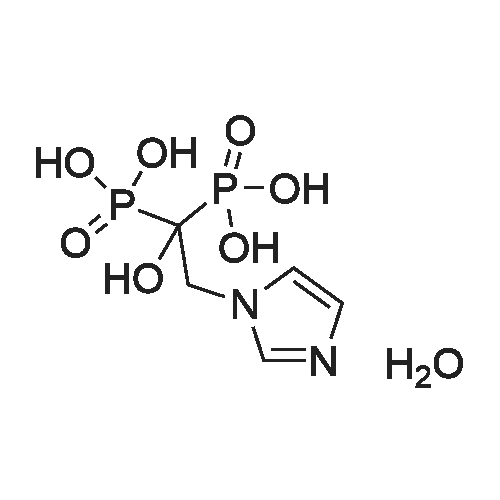
A mixture of 1-imidazolylacetic acid (25g; 0.1538mol) and H3PO3 (18.9g; 0.2306mol) in N,N'-dimethylethyleneurea (DMEU) (150ml) is heated to a temperature of from 4O0C to 500C. PCl3 (26ml; 0.3076mol) is slowly added to the resulting suspension. The resulting mixture is heated to a temperature of from 500C to 6O0C and stirred until reaction is complete by HPLC. Water is slowly added to the reaction mixture and the resulting solution is heated, with stirring, to a temperature of from 800C to 1000C until the reaction is complete. The reaction mixture is cooled to ambient temperature and the pH is adjusted to pH 8.0 to 9.0 with aqueous sodium hydroxide solution. The resulting solution is filtered and the pH of the solution is adjusted to pH 1.5 to 2.0. Ethanol is added and precipitation of solids occurs. The solid is filtered, washed and dried under vacuum at a temperature of from 45C to 550C to a constant weight. 25.7g of zoledronic acid is obtained (molar yield: 85.6%) with a HPLC purity higher than 99.5% in area. [The yield was calculated on dry basis]The product was characterized as follows:1H NMR (D2O) delta=4.71 (t, 2H, CH2); 7.28 (dd., IH3CH); 7.44 (dd., IH, CH); 8.62 (s., IH, CH)31P NMR (D2O) delta= 16.03 |
| 83% |
|
Example 1 - Zoledronic Acid; EPO <DP n="7"/>A suspension of 1-imidazol-l-yl acetic acid (200 g) in methanesulfonic acid 98-99 % (240 ml) is added slowly and blending phosphorous trichloride (856 ml).The temperature is increased until reaching 55 0C, reflux is observed.Once the phosphorous trichloride aggregate is finished, the aggregate of water (171 ml) is started, thus increasing the exothermy, which is evidenced through a larger volume of reflux.During the reaction hydrogen chloride is released.The mass in suspension slowly dissolves and the solution turns very viscous, thus making the agitation difficult.After 12 hours reaction at 55-70 0C, water is slowly added (805 ml), in a period of 2-3 hours at a temperature between 8 and 25 0C, with which a fluid solution is achieved.It is then heated at 105 - 112 0C over 3 hours and the solution is filtered to eliminate impurities.The resulting solution is partially neutralized at a temperature of 30 - 40 0C with a sodium hydroxide aqueous solution 50 % (w/v) until obtaining a pH of 0.25 +/- 0.03It is then cooled down to 0-5 0C, maintaining this temperature over at least 2 hours the precipitate being filtered.The same is washed by resuspension once in water (500 ml) and twice in methanol (500 ml each time).The precipitate may be dried in a stove at 50 - 60 0C, thus obtaining the raw zoledronic acid with a potentiometric titre equal to or exceeding 98 %.It may also be used humid to prepare the trihydrate form.The output is 83 %. |
| 75% |
|
The suspension of (1-imidazoyl)ethanoic acid (10.0 g) and phosphorous acid (19.5 g) in diphenyl ether (50 ml) was heated up to 70 C. for 1 hour. Phosphorous trichloride (20 ml) was slowly added to reaction mass at 70 C. temperature and maintained reaction temperature for another 6 hours. Reaction mass was cooled to 25 C. followed by addition of water (150 ml) and toluene (30 ml). Reaction mixture was again heated to 70 C. and charged charcoal in hazy biphasic solution, stirred, filtered through Hyflo bed, washed the bed with hot water (30 ml). Layers was separated from filtrate, aqueous layer was washed with toluene (20 ml) and combined organic layer was then back extracted with water (20 ml) and mixed with main aqueous layer. The water (140 ml) was distilled out from combined aqueous layer at atmospheric pressure in 2 hours and then refluxed the concentrated mass for 13 hours. Reaction mass was cooled to 25 C. followed by addition of methanol (50 ml) in 1 hours. The reaction mixture was stirred and again cooled to 0 C. followed filtration. The filtrate was washed with chilled 1:2 mixture (30 ml) of water and methanol and dried at 60 C. to get Zoledronic Acid. Yield: 19.0 gm (75%) |
| 74% |
|
Example 3Preparation of [1- hydroxy -2-(l H-imidazol-l -yl) ethylidenejbisphosphonic acid6.3 g (0.05 mol) imidazol-l-yl-acetic acid is dissolved in 14 ml methanesulfonic acid while stirring. After 30 minutes of stirring, 13.6 ml (0.16 mol) phosphorus trichloride is added dropwise, then the mixture is stirred for 3 hours at 80 C. The mixture is left to cool down to room temperature, then 36 ml water is added dropwise. The mixture is warmed to 105-110 C and then, stirred for 5 hours. Then the reaction mixture is purified with 0.75 g activated carbon and then filtered, washed with 5 ml water and then, 10 M NaOH solution is added droppwise until pH is 1.8. The suspension is stirred for 1 day then the precipitated product is filtered, washed with water and dried. Yield: 10.1 g (74 %), Free acid content: 23 %, Purity: 83 %The product is recrystallized with a 5 X excess of 1 N HCl. Yield: 6.3 g (46 %), Free acid content: 98 %, Purity: 98 % |
| 72% |
|
Examples; CRYSTAL FORMS OF ZOLEDRONIC ACID (ZLD-Ac); Preparation of ZLD-AC crystal form I; General procedure for the preparation of ZLD-AC crystal form I starting from 1- Imidazoleacetic acid (IAA), Phosphorous acid (H3PO3) and Phosphorous oxychloride (POC13) (Examples 1-9, see Table 1) :; A cylindrical reactor equipped with a mechanical stirrer, a thermometer, a reflux condenser and a dropping funnel, is loaded with 1-IMIDAZOLEACETIC acid (IAA), Phosphorous acid and a diluent (Toluene/Chlorobenzene/PEG-400/Silicon oil). The obtained suspension is heated to 75C-80C and Phosphorous oxychloride is added drop- wise. The reaction mixture is then heated to 75C-100C for 1-34 hours. Then water is added at 80C-100C. The mixture is stirred vigorously for about 15 minutes. [In some cases, when Silicon oil is used as a diluent, there is a need to add Toluene in order to improve the separation between the oily phase and the aqueous phase]. Then the phases are separated. The aqueous phase is put in a clean reactor and heated to 95C-100C for 13.5-19 hours. Then it is cooled to 5C and absolute Ethanol is added to obtain a precipitate after stirring at 5C for 2.5-4 hours [In some cases a precipitate of Zoledronic acid is obtained without adding absolute Ethanol as an anti-solvent]. The white product is then filtered, washed with absolute Ethanol and dried in a vacuum oven at 50C for 17-24 hours to obtain Zoledronic acid crystal form I (LOD BY TGA=6. 3%-9. 3%).; ZLD HPLC METHOD: COLUMN: PHENOMENEX PHENYL-HEXYL 5UM, 250X4.6MM MOBILE PHASE: 40MM OCTANSULFONIC ACID SODIUM SALT IN 1% HCLO4, 0.2% H3PO4 : METHANOL (85:15) DETECTION: 220NM STABILITY WAS MEASURED VERSUS THE PRESENCE OF FORM II. P THE STABILITY DATA FOR EXAMPLE 4 IN THE TABLE ABOVE IS: |
| 70.7% |
|
Preparation of zoledronic acid monohydrate A suspension of 1-imidazolylacetic acid (50g, 0. 396mol) and phosphorous acid (48.7g, 0. 594mol) in sulfolan (180ml) is heated to 75C for 30 min. The mixture is cooled to 35-40 C and then gradually introduced phosphorous trichloride (117ml, 1. 346mol) while maintaining the temperature between 35-45C. The mixture is heated to 63-67C for 3hrs, whereby white solid results. It is then cooled to 0-5C and quenched by slow addition of water (500ml) at 0-5C over a period of 1 hr. The resulting clear solution is heated at 100C for 3hrs, cooled to ambient temperature and charcoalized. To the charcoalized solution is added acetone (800ml). The mixture is then stirred for 4hrs at 20- 25C and the crystallized product is filtered, washed sequentially with chilled water (200ml), acetone (100ml) and dried in air oven at 55-60C until water content is between. |
| 70% |
|
3.8 mmol (1 equiv) of the respective carboxylic acid (658 mg 3-pyridylacetic acid hydrochloride for 1, 478 mg imidazol-1-yl-acetic acid for 2, 339 mg beta-alanine for 3, 392 mg gamma-aminobutyric acid for 4, 499 mg 6-aminohexanoic acid for 5) were added to 11.4 mmol (3 equiv) H3PO3 in a dry flask. 1.6 mL of distilled sulfolane was added and the contents were heated briefly to dissolve the solids. The solution was cooled down to approximately 25-35 C, and 11.4 mmol (3 equiv) of PCl3 were immediately added. The flask was then placed in a Milestone Ethos Synth Microwave Synthesis Labstation and fitted with a condenser through which cold water was passed. The following microwave programs were applied:For synthesis of 1: 3 min ramp to 65 C, followed by 15 s at 65 C. For synthesis of 2: 3 min ramp to 65 C, followed by 45 s at 65 C.For synthesis of 3: 3 min ramp to 65 C, followed by 15 s at 65 C.For synthesis of 4: 3 min ramp to 65 C, followed by 4 min at 65 C.For synthesis of 5: 3 min ramp to 65 C, followed by 4 min at 65 C.The power was automatically adjusted to reach and maintain the temperature designated by the program, which is determined by a built-in IR sensor in the microwave reactor. For the synthesis of intermediates 1 and 2, the power fluctuated between 0 and a max of 300-400 W, while for 3, 4, and 5, the max power was typically 200-300 W.The solid mixture after microwave irradiation consists of intermediate phosphorus compound together with a yellow-orange unwanted side product which can be removed by centrifugation before or after hydrolysis. The reaction mixture was quenched with 6 mL of H2O, yielding a clear solution that was then transferred to a 50 mL sealed quartz reaction vessel and was hydrolyzed to the bisphosphonic acid in the microwave reactor with a 6 min ramp to 150 C, followed by 4 min at 150 C. The power applied fluctuated between 0 and a max of 450-500 W. The pH of the hydrolysis mixtures for acids 1, 3, 4, and 5 was adjusted15 with NaOH and the mixture then aged at 0-5 C until crystallization of the products was complete. Acids 4 and 5 were precipitated as monosodium salts by stirring with 2-5 mL ethanol for 1-2 h at room temperature. Acid 2 was precipitated by the addition of 9 mL acetone to the acidic hydrolysis mixture and stirring for 3-4 h at room temperature. The white crystalline products were then filtered and washed with cold H2O and acetone or ethanol and then dried under vacuum at 45 C to constant weight.15 with NaOH and the mixture then aged at 0-5 C until crystallization of the products was complete. Acids 4 and 5 were precipitated as monosodium salts by stirring with 2-5 mL ethanol for 1-2 h at room temperature. Acid 2 was precipitated by the addition of 9 mL acetone to the acidic hydrolysis mixture and stirring for 3-4 h at room temperature. The white crystalline products were then filtered and washed with cold H2O and acetone or ethanol and then dried under vacuum at 45 C to constant weight. |
| 59% |
|
Examples; CRYSTAL FORMS OF ZOLEDRONIC ACID (ZLD-Ac); Preparation of ZLD-AC crystal form I; General procedure for the preparation of ZLD-AC crystal form I starting from 1- Imidazoleacetic acid (IAA), Phosphorous acid (H3PO3) and Phosphorous oxychloride (POC13) (Examples 1-9, see Table 1) :; A cylindrical reactor equipped with a mechanical stirrer, a thermometer, a reflux condenser and a dropping funnel, is loaded with 1-IMIDAZOLEACETIC acid (IAA), Phosphorous acid and a diluent (Toluene/Chlorobenzene/PEG-400/Silicon oil). The obtained suspension is heated to 75C-80C and Phosphorous oxychloride is added drop- wise. The reaction mixture is then heated to 75C-100C for 1-34 hours. Then water is added at 80C-100C. The mixture is stirred vigorously for about 15 minutes. [In some cases, when Silicon oil is used as a diluent, there is a need to add Toluene in order to improve the separation between the oily phase and the aqueous phase]. Then the phases are separated. The aqueous phase is put in a clean reactor and heated to 95C-100C for 13.5-19 hours. Then it is cooled to 5C and absolute Ethanol is added to obtain a precipitate after stirring at 5C for 2.5-4 hours [In some cases a precipitate of Zoledronic acid is obtained without adding absolute Ethanol as an anti-solvent]. The white product is then filtered, washed with absolute Ethanol and dried in a vacuum oven at 50C for 17-24 hours to obtain Zoledronic acid crystal form I (LOD BY TGA=6. 3%-9. 3%).; ZLD HPLC METHOD: COLUMN: PHENOMENEX PHENYL-HEXYL 5UM, 250X4.6MM MOBILE PHASE: 40MM OCTANSULFONIC ACID SODIUM SALT IN 1% HCLO4, 0.2% H3PO4 : METHANOL (85:15) DETECTION: 220NM STABILITY WAS MEASURED VERSUS THE PRESENCE OF FORM II. P THE STABILITY DATA FOR EXAMPLE 4 IN THE TABLE ABOVE IS: |
| 53 - 65% |
|
Example 1; [0022] An oven-dried 250 ml_ 4-neck RB flask was fitted with a mechanic stirrer, K-thermocouple, condenser, nitrogen inlet and outlet and two 1/8 inch polytetrafluoroethylene (PTFE) feeding lines. The system was flushed with nitrogen for 30 minutes. Under nitrogen protection, imidazoIe-1-ylacetic acid (15.97 g, 0.13 mole), sulfolane (70 ml_) and phosphorous acid (2.67 g, 0.033 mol) were charged to the RB flask. The reaction mixture was mixed at 210 RPM and heated to 600C. PCI3 (9.18 g, 0.067mol) was added slowly (1.3 mL/mn) via a masterflex tubing pump. Five minutes were allowed for mixing. Alternately fed were 26 wt% phosphorous acid sulfolane solution (30.7 g, 10.23 g each addition at 1.6 mL/min) and PCI3 (27.5 g, 9.18 g each portion at 1.3 mL/min). Three to five minutes of mixing were allowed between additions. The addition took 1 hr 3 min and temperature was maintained between 600C and 670C. After addition was complete, the temperature of the reaction mixture was raised to 800C and was held at this temperature for 4 hours. Then the temperature of the reaction mixture was raised to 880C and held for 30 minutes. Ambient temperature water (50 g) was added in to quench the reaction. The solution was refluxed for 3 hrs <n="13"/>. temperature, the product was vacuum-filtered and rinsed with 38 g of acetone. Zoledronic acid was obtained as a white crystalline solid (24.1 g, 95.3wt% purity by quantitative NMR, 65% yield). Example 2; [0023] An oven-dried 250 mL 4-neck RB flask was fitted with a mechanic stirrer, K-thermocouple, condenser, nitrogen inlet and outlet and two 1/8 inch PTFE feeding lines. The system was flushed with nitrogen for 30 minutes. Under nitrogen protection, imidazole-1-ylacetic acid (15.97 g, 0.13 mole), sulfolane (70 mL) and phosphorous acid (2.67 g, 0.033 mol) were charged to the RB flask. The reaction mixture was mixed at 300 RPM and heated to 600C. PCI3 (9.18 g, 0.067mol) was added in slowly (1.3 mL/min) via a masterflex tubing pump. Five minutes were allowed for mixing. Alternately fed were 26 wt% phosphorous acid sulfolane solution (30.7 g, 10.23 g each addition at 1.6 mL/min) and PCI3 (27.5 g, 9.18 g each portion at 1.3 mL/min. Three to five minutes of mixing were allowed between additions. The addition took 1 hr 6 min and temperature was maintained between 54 and 64C. After addition was complete, the temperature of the reaction mixture was raised to 800C and held for 4 hours. Then the temperature of the reaction mixture was raised to 880C and held for 30 minutes. This slurry was transferred via 3/8" PTFE tubing using nitrogen pressure into 100 mL of pre-heated (80C) water under mixing. The resultant water solution was heated to refluxing and held at that temperature for 4 hr. It was then slowly cooled to room temperature then to 1-2C and held for 1.5 hr at this temperature. The product was collected via vacuum filtration. The cake was rinsed with acetone (20 g) and zoledronic acid was obtained as a white crystalline solid (19.1 g, 98.3 wt% purity by quantitative NMR, 53% yield). Example 3; [0024] An oven-dried 250 mL 4-neck RB flask was fitted with a mechanic stirrer, K-thermocouple, condenser, nitrogen inlet and outlet and two 1/8 inch PTFE feeding lines. The system was flushed with nitrogen for 30 minutes. Under nitrogen protection, imidazole-1-ylacetic acid (15.9 g, 0.13 mole), sulfolane (70 mL) and phosphorous acid (2.67 g, 0.033 mol) were charged to the RB flask. The reaction mixture was mixed and heated to 6O0C. PCI3 (36.7 g, 0.267 mol) and phosphorous acid (8.0 g, 0.098 mol) in <n="14"/>- . mL/min respectively. Reaction temperature remained between 6O0C and 67C during feeding, After addition of PCI3 and phosphorous acid, the reaction slurry was heated to 800C and held at that temperature for 4 hours. This slurry was then transferred via 3/8" PTFE tubing using nitrogen pressure into 50 m._ of pre-heated (8O0C) water under mixing. The resultant water solution was heated to refluxing and held at that temperature for 4 hr. It was then slowly cooled to 48-500C. Acetone (200 ml_) was added in slowly and then it was cooled to 1-2C and held for 2 hr at this temperature. The product was collected via vacuum filtration. The cake was rinsed with acetone (35 g) and zoledronic acid was obtained as a white crystalline solid (22.1 g, 98.6 wt% purity by quantitative NMR, 61 % yield). Example 4; [0025] A 2.5 L resin-kettle was fitted with a mechanic stirrer, K-thermocouple, condenser, nitrogen inlet and outlet, two 1/8 inch PTFE tubing as PCI3 and phosphorous acid sulfolane solution feeding lines. The system was dried under a stream of nitrogen. Under nitrogen, imidazoleacetic acid (79.92 g, 0.63 mol), phosphorous acid (13.90 g, 0.17 mol) and sulfolane (350 ml_) were added to the resin-kettle and mixed at 200 RPM. The mixture was heated to 62C then PCI3 (36.7 g, 0.267 mol) was added in slowly (1.3 mL/min) using a masterflex tubing pump. The slurry was mixed for 5 minutes. Phosphoro... |
| 53% |
|
6.3 g (0.05 mol) of imidazol-1-yl-acetic acid was dissolved in 21 ml of methanesulfonic acid with stirring. 13.6 ml (0.16 mol) of phosphorus trichloride was added dropwise and the mixture was stirred at 80 C for 3 h. After cooling to 26 C, 36 ml of water was added dropwise, the temperature was elevated to 105-110 C and the contents of the flask were stirred at this temperature for 5 h. Next, the mixture was stirred with 0.75 g of activated carbon for 30 min, filtered and the solid washed with 5 ml of water. To the combined water phase was added dropwise 10 N NaOH solution to attain pH 1.8. The suspension obtained was stirred for 5 h, the solid filtered, washed with 5 ml of water and dried to afford 9.7 g (71%) of a crude mixture consisting of a 26-74% mixture of ZA and ZA-Na. Recrystallization from 48.5 ml of 1 N hydrochloric acid furnished 7.2 g (53%) of ZA comprising 99% of the acid (Table 1, entry 7); 31P NMR (5% NaOH/D2O) 16.0 [P8 (5% NaOD/D2O) 16.2]. |
| 50% |
|
PREPARALION OF ZLD-AC CRYSTAL FORM XNII; Example 18 :; A 3L reactor equipped with A mechanical stirrer, a thermometer, a reflux condenser and a dropping funnel, was loaded with 1-IMIDAZOLEACETIC acid (70. OG, 0. 56MOLE), Phosphorous acid (136. 7g, 1.67mole) and Silicon oil (M-350) (490ML). The suspension was heated to 80C and Phosphorous oxychloride (194. 4ML, 2.08mole) was added drop-wise during 4 hours. The reaction mixture was stirred at 80C for 22 hours. Then water (490ML) was added slowly at 80C. The mixture was stirred vigorously for about 30 minutes. Then the silicon oil phase and the aqueous phase were separated. The aqueous phase was put in a clean reactor and heated to 97C for 17.5 hours. Then absolute Ethanol (490ML) was added and the solution was stirred at reflux (87C) for 2 hours. The solution was then cooled to 70C-72C during about 1 hour and was kept at this temperature for 1 hour. After cooling to 25C during 2.5 hours and stirring at this temperature for 1 hour, half of the product was filtered, washed with small amount of cold water and dried in a vacuum oven at 50C for 20 hours to obtain 50.8g of Zoledronic acid crystal form XVIII (MS- 507-CROP I, LOD by TGA=1.9%). The rest of the suspension was cooled to 0C during 2 hours and was stirred at this temperature for about 16 hours. Then the product was filtered and dried in a vacuum oven at 50C for 24 hours to obtain 26g of Zoledronic acid crystal form XVIII (MS-507-CROP II, LOD by TGA=1. 0%). The overall yield of the process is 50% purity by HPLC 97.7%. |
| 49% |
|
Example II. Preparation of [l-hydroxy-2-(lH-imidazol-l-yl)- ethylidene]bisphosrhohonic acid.A solution of lH-imidazole-1 -acetic acid [26 g (0.206 moles) of lH-imidazole-1- acetic acid in the mixture of 18 ml of water and 18 ml of hydrochloric acid (35%)] is added dropwise for 30 minutes at the temperature 0-5 0C to 108 ml of phosphorus trichloride (PCl3) cooled to the temperature 0-5 0C. The mixture is mixed at 0-50C for 1 hour. Then, the mixture is heated to 800C and maintained at this temperature for 1 hour. The excess of phosphorus trichloride is then distilled off under the reduced pressure. 200 ml of water is added to the reaction residues and the hydrolysis is performed while maintaining boiling for 6 hours, afterwards 1 g of activated charcoal, 5 g of Hyflo Super CeI are added and mixed while boiling for 30 minutes. The mixture is filtered and the filter is washed with 20 ml of water. The filtrate is concentrated under the reduced pressure to the volume of 100 ml. 150 ml of 95% ethanol is added to the concentrated filtrate at 70C. The mixture is cooled while mixing to 250C and crystallization is performed until the temperature reaches 20-250C for 4 hours. The formed precipitate is filtered off, washed twice with 30 ml of water-ethanol mixture (1:1.5) and dried at 5O0C. 29.3 g (49%) of [l-hydroxy-2-(lH-imidazol-l-yl)-ethylidene]bisrhohosphonic acid monohydrate is obtained. EtaPLC 100.00% , TGA 6.30%XPRD: 12.05; 12.77; 15.69; 18.80; 20.84; 21.25; 21.71; 22.09; 25.71; 27.50; 29.19; 32.42; 32.88 (+,- 0,02 ) 2 theta1H NMR (D2O): 5=4.670-4.709 ppm (t, 2H, J=9.65); 7.379 (s,lH); 7.540 (s, IH);8.719 (s, IH)13C NMR: 5=55.56 ppm; 74.81-76.90 (t); 121.13; 126.83; 138.7131P NMR: 5=14.36 ppm15 g of [l-hydroxy-2-(l/-r-imidazol-l-yl)-ethylidene]bisphosphonic acid is suspended in 300 ml of water, heated to the boiling point and mixed while boiling for 15 minutes. Then, the mixture is cooled to 70C and 300 ml of ethanol is added for about 1-1.5 hour. The mixture is cooled to 20-25C. After 14 hours of mixing in20-25C the precipitate is filtered off, washed twice with 15 ml of water and once with 15 ml of ethanol and dried for 6 hours at 55C. 12.78 g (85%) of [l-hydroxy-2-(lH-imidazol-l-yl)-ethylidene]bisphosphonic acid monohydrate is obtained.EtaPLC 100.00%, TGA 6.24%XPRD: 12.05; 12.78; 15.72; 18.70; 20.82; 21.23; 21.70; 22.08; 25.70; 27.51; 29.14;32.32; 32.88 (+,- 0.02 ) 2 theta 1H NMR (D2O): 5=4.470-4.497 ppm (t, 2H, J=9.62); 7.164 (s,lH); 7.367 (s, IH);8.421 (s, IH)13C NMR: 5=53.00 ppm; 72.59 (t); 118.47; 123.66; 138.7131P NMR: 5=14.21 ppm |
| 49% |
|
EXAMPLE II Preparation of [1-hydroxy-2-(1H-imidazol-1-yl)-ethylidene]bisphosphonic acid A solution of 1H-imidazole-1-acetic acid [26 g (0.206 moles) of 1H-imidazole-1-acetic acid in the mixture of 18 ml of water and 18 ml of hydrochloric acid (35%)] is added dropwise for 30 minutes at the temperature 0-5 C. to 108 ml of phosphorus trichloride (PCl3) cooled to the temperature 0-5 C. The mixture is mixed at 0-5 C. for 1 hour. Then, the mixture is heated to 80 C. and maintained at this temperature for 1 hour. The excess of phosphorus trichloride is then distilled off under the reduced pressure. 200 ml of water is added to the reaction residues and the hydrolysis is performed while maintaining boiling for 6 hours, afterwards 1 g of activated charcoal, 5 g of Hyflo Super Cel are added and mixed while boiling for 30 minutes. The mixture is filtered and the filter is washed with 20 ml of water. The filtrate is concentrated under the reduced pressure to the volume of 100 ml. 150 ml of 95% ethanol is added to the concentrated filtrate at 70 C. The mixture is cooled while mixing to 25 C. and crystallization is performed until the temperature reaches 20-25 C. for 4 hours. The formed precipitate is filtered off, washed twice with 30 ml of water-ethanol mixture (1:1.5) and dried at 50 C. 29.3 g (49%) of [1-hydroxy-2-(1H-imidazol-1-yl)-ethylidene]bisphosphonic acid monohydrate is obtained. HPLC 100.00% , TGA 6.30% XPRD: 12.05; 12.77; 15.69; 18.80; 20.84; 21.25; 21.71; 22.09; 25.71; 27.50; 29.19; 32.42; 32.88 (+,-0.02) 2theta 1H NMR (D2O): delta=4.670-4.709 ppm (t, 2H, J=9.65); 7.379 (s, 1H); 7.540 (s, 1H); 8.719 (s, 1H) 13C NMR: delta32 55.56 ppm; 74.81-76.90 (t); 121.13; 126.83; 138.71 |
| 28% |
With phosphoric acid; phosphorus trichloride; In diethylene glycol dimethyl ether; water; at 50 - 100℃; for 8 - 11h;Heating / reflux;Product distribution / selectivity; |
Example 3:Preparation of Zoledronic Acid:[0019] A 1.5 liter kettle reactor, fitted with a heating mantle, mechanical stirrer, dropping funnel, thermocouple and condenser with nitrogen inlet adapter, was charged with imidazoleacetic acid (100 g, 0.793 mol), diglyme (400 ml), and 85% phosphoric acid (55 ml). Phosphorus trichloride (330 g, 2.41 mol) was slowly added to the reaction mass resulting in an exotherm and the evolution of hydrogen chloride. The temperature was allowed to rise to 700C and the solution was stirred until the evolution of HCl subsided. The temperature of the reaction mass was increased to 85C and a white solid began to form, float and adhere to the stirrer shaft. After about 1 hour, stirring became impossible and the stirring motor was stopped. The reaction mass was heated for 5 more hours at 85C and then cooled to ambient temperature, producing a solid homogeneous white mass. <n="8"/>[0020] Water was slowly added to the white mass (320 ml) that resulted in an exotherm and HCl evolution. The water slowly dissolved the mass in a gradual and uniform fashion, eventually liberating the stirrer. After the mass substantially dissolved, the solution was refluxed for 5 hours, then cooled and stripped to a gum with a rotary evaporator, collecting 420 g of water (pH 0.65). More water (250 ml) was added and stripped, collecting 166 g of water (pH 1.87). Water (250 ml) was again added and stripped, collecting 316 g (pH 2.14). The flask was removed from the rotary evaporator, water (150 ml) was added and the mixture was heated to 90-950C during which time all solids dissolved. The solution was seeded with zoledronic acid monohydrate crystals and slowly cooled to room temperature then chilled to 30C with an ice bath. The resulting crystalline solid was filtered, rinsed with acetone (200 + 100 ml) and dried under a nitrogen stream giving a crop of 52.4 g. Acetone was also added to the filtrate (200 ml) and the solution was left in a freezer overnight giving a second crop of crystals (12.0 g) which, after washing with acetone and drying, was combined with the first crop for a total yield of 64.4 g (28%). The NMR indicated the presence of traces of diglyme, acetone and H3PO3 impurities. Example 4:Preparation of Zoledronic Acid:[0021 ] A 5 liter cylindrical jacketed reactor was fitted with a mechanical stirrer, thermocouple, nitrogen inlet adapter and a condenser with a caustic scrubber. This was charged with imidazoleacetic acid (0.333 kg, 2.64 mol) and diglyme (1.00 1). The slurry was heated to 500C while stirring (100 rpm) under a slow nitrogen purge (1 1/min). Additional diglyme (0.26 1) and 85% phosphoric acid (0.304 kg) were added to the reaction mass. Using a Masterflex pump and Teflon tubing, phophorus trichloride (1.04 kg total, 7.57 mol) was pumped into the reaction mass, slowly (2 ml/min) at first and then at an increased rate (40 ml/min), after the water in the phosphoric acid had been depleted. During addition of the PCl3, the temperature was raised to about 65C and a white mass gradually formed, causing the stirrer to bind. The jacket temperature was increased to 850C causing PCl3 to reflux. The refluxing slowed and then stopped as the white mass expanded. The reactor was allowed to stand at about 80C for four hours, after which the jacket temperature was set at 150C overnight.[0022] The reactor jacket temperature was increased to 500C and water (0.95 kg total) was slowly (2-5 ml/min) added with a Masterflex pump. The water dissolved the white mass on contact, liberating HCl in an exothermic reaction. After about 250 g of water was added to the reaction mass, the stirrer became unbound and stirring was resumed (100 rpm). The water <n="9"/>addition rate was slowly increased to 40 ml/min. The reaction mass was then heated at about 1000C for 4 hours and then cooled to room temperature.[0023] The reaction mass was drained and rotary evaporated to yield a gum. Water was added to the gum and stripped several times until the distillates pH rose above 1. The resulting aqueous solution (1.2 kg, 1.6 1) was stirred in a beaker and acetone (1.5 1) was slowly added. The mixture was allowed to stand 16 hours to complete crystallization. The solid was filtered, thoroughly washed with acetone and dried in a nitrogen stream to give crude Zoledronic acid (0.202 g, 0.74 mol, 28% yield). |
| 7% |
With phosphoric acid; phosphorus trichloride; In water; PEG 400; at 70 - 85℃; for 11h;Heating / reflux;Product distribution / selectivity; |
Example 6:(Comparative) Preparation of Zoledronic Acid in PEG-400:[0025] Example 3 was repeated substituting PEG-400 (400ml) for diglyme. After the addition of phosphorus trichloride and increased temperature of the reaction mass, a solid formed that eventually returned to solution upon further heating. The yield of zoledronic acid was 7 % (isolated yield). 1HNMR (D2O/NaOD): 7.72 (s, 1 H); 7.22 (s, 1 H); 6.87 (s, 1 H); 4.82 (O-H, 7.02 H); 4.45 (m, 2 H). 31P NMR (D2O/NaOD): 17.0 (m). Not only was there a substantial decrease in yield, but the product purity deteriorated as well. |
|
|
A suspension of imidazol-1-ylacetic acid, compound of formula 2 (50g, 0. 396mol) and phosphorous acid (48.7g, 0. 594mol) in sulfolan (180ml) was heated to 75 C for 30 min. The mixture was cooled to 35-40 C and phosphorous trichloride (117ml, 1. 346mol) was gradually introduced while maintaining the temperature between 35-45 C. The mixture was heated to 63-67 C for 3 hours, whereby white solid results. It was then cooled to 0- 5'C and quenched by slow addition of water (500ml) at 0-5 C over a period of 1 hour. The resulting clear solution was heated at 100 C for 3 hours, cooled to ambient temperature and charcoalized. Acetone was added to the charcoalized solution. The mixture was then stirred for 4 hours at 20-25 C and the crystallized product was filtered, washed sequentially with chilled water, acetone and dried to obtain zoledronic acid. |
|
|
Preparation of zoledronic acid monohydrate A suspension of 1-imidazolylacetic acid (20g, 0. 159mol) and phosphorous acid (19.6g, 0. 239mol) in 1,2-dimethoxyethane (72m1) is heated to 75 C for 30 minutes. The mixture is cooled to 35-40 C and then gradually introduced phosphorous trichloride (48ml, 0. 543mol) while maintaining the temperature between 35-45 C. The mixture is heated to 63-67 C for 3 hrs, whereby white solid results. It is then cooled to 0-5 C and quenched by slow addition of water (160ml) at 0-5 C over a period of 1 hr. The resulting clear solution is heated at 100 C for 3 hrs, cooled to ambient temperature and charcoalized. To the charcoalized solution is added acetone (320ml). The mixture is then stirred for 4 hours at 20-25 C, the crystallized product is filtered, washed sequentially with chilled water (80ml), acetone (80ml) and dried in air oven at 55-60 C until water content is between 6.2-7. 2% w/w. Appearance: white crystalline solid, purity >99.5%, meeting specification as per IHS. |
|
|
Imidazol-1-ylacetic acid (50 gm), phosphorous acid (150 gm) and n-octane (1000 mL) were taken in a four necked round bottom flask fitted with an addition funnel, mechanical stirrer, condenser and thermometer pocket and allowed to stir at 90-950C. Phosphorus trichloride (250 gm) was then added to the reaction mixture and allowed to heat at 90- 950C. The reaction mixture was cooled and distilled water (500 mL) was added to it. The reaction mixture was further heated to 90-950C and then cooled to room temperature, filtered through celite bed. Aqueous layer was separated and methanol (2000 mL) was EPO <DP n="11"/>added to it. The solution was cooled to 0-5 C and stirred for 4-5 hrs. The precipitated solid was filtered, washed with methanol and dried under vacuum yielding 70 gm of product. |
|
|
Imidazol-1-ylacetic acid (50 gm), phosphorous acid (150 gm) and 1,4-dioxane (1000 mL) were taken in a four necked round bottom flask fitted with an addition funnel, mechanical stirrer, condenser and thermometer pocket and allowed to stir at 90-950C. Phosphorus trichloride (250 gm) was then added to the reaction mixture and allowed to heat at 90- 95C. The reaction mixture was cooled and distilled water (500 mL) was added to it. The reaction mixture was further heated to 90-950C and then cooled to RT and filtered through celite bed. Methanol (2000 mL) was added to the filtrate and cooled to 0-50C and stirred for 4-5 hrs. The precipitated solid was filtered, washed with methanol and dried under vacuum to yield 56.0 gm of product. |
|
|
Example- 3: Preparation of zoledronic acid; A mixture of imidazol-1-yl acetic acid (100 gm) and phosphorous acid (324 gm) was heated under stirring to about 60-800C to get a homogeneous solution. Phosphorous trichloride (324 gm) was added slowly at a temperature of about 750C to the homogeneous solution. The resultant mass was stirred for 6 hours and cooled. A solution of hydrochloric acid (9N, 465 ml) was added over 30 minutes and the entire mass was heated at 75-8O0C for about 12 hours, treated with activated carbon (3.8 gm) and filtered. Acetone (1200 ml) was added to the filtrate and the resultant mixture was cooled to 15-2O0C. After complete precipitation of the product, the mass was filtered and the wet cake was washed with acetone (300 ml) and dried at 50-6O0C to get zoledronic acid as a white crystalline solid. Yield: 161 gmHPLC Purity: 99.92% |
|
|
Example-5: Preparation of zoledronic acid; A mixture of imidazol-1-yl acetic acid (100 gm) and phosphorous acid (324 gm) was heated under stirring to about 60-800C to get a homogeneous solution. Phosphorous oxychloride (364 gm) was added slowly at a temperature of about 750C to the homogeneous solution. The resultant mass was stirred for 5 hours and cooled. A solution of hydrochloric acid (9N, 465 ml) was added over 30 minutes and the entire mass was heated at 9O0C for about 12 hours, cooled and filtered. Acetone (1200 ml) was added to the filtrate and the resultant mixture was cooled to 15-2O0C. After complete precipitation of the product, the mass was filtered and the <n="10"/>wet cake was washed with acetone (300 ml) and dried at 50-600C to get crystals of zoledronic acid.Yield: 170 gmHPLC Purity: 99.92% |
|
|
Imidazol-1-ylacetic acid (50 gm), phosphorous acid (150 gm) and n-octane (1000 mL) were taken in a four necked round bottom flask fitted with an addition funnel, mechanical stirrer, condenser and thermometer pocket and allowed to stir at 90-95 C. Phosphorus trichloride (250 gm) was then added to the reaction mixture and allowed to heat at 90-95 C.The reaction mixture was cooled and distilled water (500 mL) was added to it. The reaction mixture was further heated to 90-95 C. and then cooled to room temperature, filtered through celite bed. Aqueous layer was separated and methanol (2000 mL) was added to it. The solution was cooled to 0-5 C. and stirred for 4-5 hrs. he precipitated solid was filtered, washed with methanol and dried under vacuum yielding 70 gm of product. |
|
|
Take the above compounds Intermediate 2 3.15g (0.025mol), H3PO4 2.45g (0.025mol) and polyethylene glycol 400 0.5h the reaction at 100 , after cooling to 65 deg.] C of 15g (0.037mol), was slowly added dropwise phosphorus trichloride was added 0.025 mol (2.2 mL), addition was complete, the reaction was continued 3h 100 , followed by addition of 30mL of concentrated hydrochloric acid was refluxed for 5h, the reaction was completed, cooled to room temperature, the reaction solution was poured into 3 of ethanol with a white solid was precipitated, filtered, and dried in vacuo to give the compound . |

 Chemistry
Chemistry
 Pharmaceutical Intermediates
Pharmaceutical Intermediates
 Inhibitors/Agonists
Inhibitors/Agonists
 Material Science
Material Science














 For Research Only
For Research Only
 110K+ Compounds
110K+ Compounds
 Competitive Price
Competitive Price
 1-2 Day Shipping
1-2 Day Shipping