Abstract
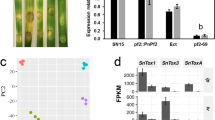
Magnaporthe oryzae is notorious for its efficient evolution as a plant pathogen. Analysis of well organised fungal pathogenic mechanisms would enable the scientific understanding to fight against pathogens. Histidine phosphotransferase in fungal systems are good targets for antifungals since they are component of SAPK pathway, which is essential for survival of the fungus. The histidine Phosphotransferase YPD1 in M. oryzae is known to be light responsive and important for both asexual reproduction as well as pathogenicity. In this study, the knock-down (KD) mutant of YPD1 was highly sensitive to cell wall perturbing agents and were highly deficient in laccase expression and activity. A RNA seq transcriptome screening of KD mutant compared to wild type revealed altered expression of about 146 genes important in biotrophic and necrotrophic phases of the pathogen. About 96 genes were found to be down regulated, of which several plant cell wall degrading genes vital for pathogenicity and fungal cell wall maintenance genes important for growth and fungal development were identified. Most of the genes identified are involved in biotrophic invasion and metabolism. Our results explain the reason for the deficiency of the KD mutant in asexual reproduction as well as pathogenicity. Analysis of differentially expressed genes in the KD YPD1 mutant suggests that YPD1 can be a good target for development of novel antifungal compounds, as YPD1 expression affects genes required for pathogenicity.




Similar content being viewed by others
Data Availability
All the reads were deposited in National Center for Biotechnology Information (NCBI) under Gene Expression Ominibus (GEO) database (GEO accession No. GSE192934).
References
Adams DJ (2004) Fungal cell wall chitinases and glucanases. Microbiology 150:2029–2035. https://doi.org/10.1099/mic.0.26980-0
Alonso-Monge R, Román E, Arana DM et al (2009) Fungi sensing environmental stress. Clin Microbiol Infect 15(Suppl 1):17–19. https://doi.org/10.1111/j.1469-0691.2008.02690.x
Bahn Y-S (2008) Master and commander in fungal pathogens: the two-component system and the HOG signaling pathway. Eukaryot Cell 7:2017–2036. https://doi.org/10.1128/EC.00323-08
Bahn Y, Kojima K, Cox GM, Heitman J (2006) A Unique Fungal Two-Component System Regulates Stress Responses, Drug Sensitivity, Sexual Development, and Virulence of Cryptococcus neoformans. Mol Biol Cell 17:3122–3135. https://doi.org/10.1091/mbc.e06-02-0113
Bicknell A, Tourtellotte J, Niwa M (2010) Late phase of the endoplasmic reticulum stress response pathway is regulated by Hog1 MAP kinase. J Biol Chem 285:17545–17555. https://doi.org/10.1074/jbc.M109.084681
Bigeard J, Colcombet J, Hirt H (2015) Signaling Mechanisms in Pattern-Triggered Immunity ( PTI ). Mol Plant 8:521–539. https://doi.org/10.1016/j.molp.2014.12.022
Chen C, Chen H, Zhang Y et al (2020) TBtools: An Integrative Toolkit Developed for Interactive Analyses of Big Biological Data. Mol Plant 13:1194–1202. https://doi.org/10.1016/j.molp.2020.06.009
Desjardins AE, Hohn TM (1997) Mycotoxins in Plant Pathogenesis. Mol Plant-Microbe Interact 10:147–152. https://doi.org/10.1094/MPMI.1997.10.2.147
Divon HH, Fluhr R (2007) Nutrition acquisition strategies during fungal infection of plants. FEMS Microbiol Lett 266:65–74. https://doi.org/10.1111/j.1574-6968.2006.00504.x
Ebbole DJ (2007) Magnaporthe as a Model for Understanding Host-Pathogen Interactions. Annu Rev Phytopathol 45:18.1–18.20. https://doi.org/10.1146/annurev.phyto.45.062806.094346
Eckardt NA (2008) Chitin signaling in plants: insights into the perception of fungal pathogens and rhizobacterial symbionts. Plant Cell 20:241–243. https://doi.org/10.1105/tpc.108.058784
Fassler JS, West AH (2013) Histidine phosphotransfer proteins in fungal two-component signal transduction pathways. Eukaryot Cell 12:1052–1060. https://doi.org/10.1128/EC.00083-13
Fuchs BB, Mylonakis E (2009) Our Paths Might Cross : the Role of the Fungal Cell Wall Integrity Pathway in Stress Response and Cross Talk with Other Stress Response Pathways. Eukaryot Cell 8:1616–1625. https://doi.org/10.1128/EC.00193-09
Geoghegan I, Steinberg G, Gurr S (2017) The Role of the Fungal Cell Wall in the Infection of Plants. Trends Microbiol 25:957–967. https://doi.org/10.1016/j.tim.2017.05.015
Guo M, Chen Y, Du Y et al (2011) The bZIP transcription factor MoAP1 mediates the oxidative stress response and is critical for pathogenicity of the rice blast fungus Magnaporthe oryzae. PLoS Pathog 7:e1001302. https://doi.org/10.1371/journal.ppat.1001302
Harris LJ, Desjardins AE, Plattner RD et al (2017) Possible Role of Trichothecene Mycotoxins in Virulence of Fusarium graminearum on Maize. Plant Dis 83:954–960. https://doi.org/10.1094/PDIS.1999.83.10.954
Howlett BJ (2006) Secondary metabolite toxins and nutrition of plant pathogenic fungi. Curr Opin Plant Biol 9:371–375. https://doi.org/10.1016/j.pbi.2006.05.004
Iyer G, Chattoo B (2003) Purification and characterization of laccase from the rice blast fungus, Magnaporthe grisea. FEMS Microbiol Lett 227:121–126. https://doi.org/10.1016/S0378-1097(03)00658-X
Izumitsu K, Yoshimi A, Tanaka C (2007) Two-component response regulators Ssk1p and Skn7p additively regulate high-osmolarity adaptation and fungicide sensitivity in Cochliobolus heterostrophus. Eukaryot Cell 6:171–181. https://doi.org/10.1128/EC.00326-06
Jacob S, Foster AJ, Yemelin A, Thines E (2015) High osmolarity glycerol (HOG) signalling in Magnaporthe oryzae: Identification of MoYPD1 and its role in osmoregulation, fungicide action, and pathogenicity. Fungal Biol 119:580–594. https://doi.org/10.1016/j.funbio.2015.03.003
Kachroo P, Leong SA, Chattoo BB (1994) Pot2, an inverted repeat transposon from the rice blast fungus Magnaporthe grisea. Mol Gen Genet 245:339–348. https://doi.org/10.1007/BF00290114
Kahmann R, Basse C (2001) Fungal gene expression during pathogenesis-related development and host plant colonization. Curr Opin Micr 4:374–380. https://doi.org/10.1016/S1369-5274(00)00220-4
Krzywinski M, Schein J, Birol I et al (2009) Circos: An information aesthetic for comparative genomics. Genome Res 19:1639–1645. https://doi.org/10.1101/gr.092759.109
Kuźniak E (2020) Kopczewski T (2020) The chloroplast reactive oxygen species-redox system in plant immunity and disease. Front Plant Sci 11:572686. https://doi.org/10.3389/fpls.2020.572686
Lamb TM, Goldsmith CS, Bennett L et al (2011) Direct transcriptional control of a p38 MAPK pathway by the circadian clock in Neurospora crassa. PLoS ONE 6:e27149. https://doi.org/10.1371/journal.pone.0027149
Langner T, Białas A, Kamoun S (2018) The blast fungus decoded: Genomes in flux. MBio 9:1–4. https://doi.org/10.1128/mBio.00571-18
Lee JW, Ko YJ, Kim SY, Bahn YS (2011) Multiple roles of Ypd1 phosphotransfer protein in viability, stress response, and virulence factor regulation in Cryptococcus neoformans. Eukaryot Cell 10:998–1002. https://doi.org/10.1128/EC.05124-11
Lee K, Singh P, Chung W-C et al (2006) Light regulation of asexual development in the rice blast fungus, Magnaporthe oryzae. Fungal Genet Biol 43:694–706. https://doi.org/10.1016/j.fgb.2006.04.005
Livak KJ, Schmittgen TD (2001) Analysis of Relative Gene Expression Data Using Real- Time Quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods 25:402–408. https://doi.org/10.1006/meth.2001.1262
Lu L, Rong W, Massart S, Zhang Z (2018) Genome-wide identification and expression analysis of cutinase gene family in Rhizoctonia cerealis and functional study of an active cutinase RcCUT1 in the fungal-wheat interaction. Front Microbiol 9:1–13. https://doi.org/10.3389/fmicb.2018.01813
Mavrianos J, Berkow EL, Desai C et al (2013) Mitochondrial two-component signaling systems in Candida albicans. Eukaryot Cell 12:913–922. https://doi.org/10.1128/EC.00048-13
McCarthy MW, Walsh TJ (2018) Amino acid metabolism and transport mechanisms as potential antifungal targets. Int J Mol Sci 19.https://doi.org/10.3390/ijms19030909
Menke J, Dong Y, Kistler HC (2012) Fusarium graminearum Tri12p Influences Virulence to Wheat and Trichothecene Accumulation. Mol Plant-Microbe Interact 25:1408–1418. https://doi.org/10.1094/MPMI-04-12-0081-R
Missall TA, Moran JM, Corbett JA, Lodge JK (2005) Distinct stress responses of two functional laccases in Cryptococcus neoformans are revealed in the absence of the thiol-specific antioxidant Tsa1. Eukaryot Cell 4:202–208. https://doi.org/10.1128/EC.4.1.202-208.2005
Mohanan VC, Chandarana PM, Chattoo BB et al (2017) Fungal Histidine Phosphotransferase Plays a Crucial Role in Photomorphogenesis and Pathogenesis in Magnaporthe oryzae. Front Chem 5:31. https://doi.org/10.3389/fchem.2017.00031
Montibus M, Ducos C, Bormann J et al (2013) The bZIP Transcription Factor Fgap1 Mediates Oxidative Stress Response and Trichothecene Biosynthesis But Not Virulence in Fusarium graminearum. PLoS ONE 8:1–12. https://doi.org/10.1371/journal.pone.0083377
Motoyama T, Kadokura K, Ohira T et al (2005) A two-component histidine kinase of the rice blast fungus is involved in osmotic stress response and fungicide action. Fungal Genet Biol 42:200–212. https://doi.org/10.1016/j.fgb.2004.11.002
Qi Z, Wang QI, Dou X et al (2012) MoSwi6, an APSES family transcription factor, interacts with MoMps1 and is required for hyphal and conidial morphogenesis, appressorial function and pathogenicity of Magnaporthe oryzae. Mol Plant Pathol 13:677–689. https://doi.org/10.1111/J.1364-3703.2011.00779.X
Reichmann D, Voth W, Jakob U (2018) Maintaining a Healthy Proteome during Oxidative Stress. Mol Cell 69:203–213. https://doi.org/10.1016/j.molcel.2017.12.021
Saint-Macary ME, Barbisan C, Gagey MJ et al (2015) Methionine biosynthesis is essential for infection in the rice blast fungus Magnaporthe oryzae. PLoS ONE 10:1–22. https://doi.org/10.1371/journal.pone.0111108
Singh D, Gupta P, Singla-Pareek SL, Siddique KHM, Pareek A (2021) The Journey from Two-Step to Multi-Step Phosphorelay Signaling Systems. Curr Genomics 22(1):59–74. https://doi.org/10.2174/1389202921666210105154808
Skamnioti P, Gurr SJ (2007) Magnaporthe grisea cutinase2 mediates appressorium differentiation and host penetration and is required for full virulence. Plant Cell 19:2674–2689. https://doi.org/10.1105/tpc.107.051219
Snoeijers SS, Alejandro P, Joosten MHA, de Wit PJGM (2000) The effect of nitrogen on disease development and gene expression in bacterial and fungal plant pathogens. Eur J Plant Pathol 106:493–506. https://doi.org/10.1023/A:1008720704105
Solomon PS, Tan K-C, Oliver RP (2003) The nutrient supply of pathogenic fungi; a fertile field for study. Mol Plant Pathol 4:203–210. https://doi.org/10.1046/j.1364-3703.2003.00161.x
Tan K, Oliver RP (2017) Regulation of proteinaceous effector expression in phytopathogenic fungi. PLoS Pathog 13:e1006241. https://doi.org/10.1371/journal.ppat.1006241
Tanaka C, Izumitsu K (2010) Two-Component Signaling System in Filamentous Fungi and the Mode of Action of Dicarboximide and Phenylpyrrole Fungicides. In: Carisse O (ed) Fungicides. IntechOpen
Taylor BL, Zhulin IB (1999) PAS domains: internal sensors of oxygen, redox potential, and light. Microbiol Mol Biol Rev 63:479–506. https://doi.org/10.1128/MMBR.63.2.479-506.1999
Trapnell C, Pachter L, Salzberg SL (2009) TopHat : discovering splice junctions with RNA-Seq. Bioinformatics 25:1105–1111. https://doi.org/10.1093/bioinformatics/btp120
Trapnell C, Roberts A, Goff L et al (2014) Differential gene and transcript expression analysis of RNA-seq experiments with TopHat and Cufflinks. Nat Protoc 7:562–578. https://doi.org/10.1038/nprot.2012.016
Upadhya R, Baker LG, Lam WC et al (2018) Cryptococcus neoformans cda1 and its chitin deacetylase activity are required for fungal pathogenesis. MBio 9. https://doi.org/10.1128/mBio.02087-18
Van Vu B, Itoh K, Nguyen QB et al (2012) Cellulases belonging to glycoside hydrolase families 6 and 7 contribute to the virulence of Magnaporthe oryzae. Mol Plant Microbe Interact 25:1135–1141. https://doi.org/10.1094/MPMI-02-12-0043-R
Xu JR, Staiger CJ, Hamer JE (1998) Inactivation of the mitogen-activated protein kinase Mps1 from the rice blast fungus prevents penetration of host cells but allows activation of plant defense responses. Proc Natl Acad Sci U S A 95:12713–12718. https://doi.org/10.1073/pnas.95.21.12713
Yoshimi A, Kojima K, Takano Y, Tanaka C (2005) Group III Histidine Kinase Is a Positive Regulator of Hog1-Type Mitogen-Activated Protein Kinase in Filamentous Fungi. 4:1820–1828. https://doi.org/10.1128/EC.4.11.1820
Zhang H, Liu K, Zhang X et al (2010) A two-component histidine kinase, MoSLN1, is required for cell wall integrity and pathogenicity of the rice blast fungus, Magnaporthe oryzae. Curr Genet 56:517–528. https://doi.org/10.1007/s00294-010-0319-x
Zhang H, Zheng X, Zhang Z (2016) The Magnaporthe grisea species complex and plant pathogenesis. Mol Plant Pathol 17:796–804. https://doi.org/10.1111/mpp.12342
Zhou J (2016) Plant Pathology : A Life and Death Struggle in Rice Blast Disease. Curr Biol 26:R843–R845. https://doi.org/10.1016/j.cub.2016.08.038
Acknowledgements
This research work was funded by Department of Biotechnology, Ministry of Science and Technology, India.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interests
The authors have declared that no competing interests exist.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Mohanan, V.C., Chandarana, P., Prasad, B.D. et al. Transcriptome analysis of Magnaporthe oryzae reveals the role of histidine phosphotransferase in pathogenesis and stress adaptation. Australasian Plant Pathol. 51, 155–165 (2022). https://doi.org/10.1007/s13313-022-00851-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13313-022-00851-2