Abstract
Serious manifestations of respiratory virus infections such as influenza and coronavirus disease 2019 (COVID-19) are associated with a dysregulated immune response and systemic inflammation. Treating the immunological/inflammatory dysfunction with glucocorticoids, Janus kinase inhibitors, and monoclonal antibodies against the interleukin-6 receptor has significantly reduced the risk of respiratory failure and death in hospitalized patients with severe COVID-19, but the proportion of those requiring invasive mechanical ventilation (IMV) and dying because of respiratory failure remains elevated. Treatment of severe influenza-associated pneumonia and acute respiratory distress syndrome (ARDS) with available immunomodulators and anti-inflammatory compounds is still not recommended. New therapies are therefore needed to reduce the use of IMV and the risk of death in hospitalized patients with rapidly increasing oxygen demand and systemic inflammation who do not respond to the current standard of care. This paper provides a critical assessment of the published clinical trials that have tested the investigational use of intravenously administered allogeneic mesenchymal stem/stromal cells (MSCs) and MSC-derived secretome with putative immunomodulatory/antiinflammatory/regenerative properties as add-on therapy to improve the outcome of these patients. Increased survival rates are reported in 5 of 12 placebo-controlled or open-label comparative trials involving patients with severe and critical COVID-19 and in the only study concerning patients with influenza-associated ARDS. Results are encouraging but inconclusive for the following reasons: small number of patients tested in each trial; differences in concomitant treatments and respiratory support; imbalances between study arms; differences in MSC source, MSC-derived product, dosing and starting time of the investigational therapy; insufficient/inappropriate reporting of clinical data. Solutions are proposed for improving the clinical development plan, with the aim of facilitating regulatory approval of the MSC-based investigational therapy for life-threatening respiratory virus infections in the future. Major issues are the absence of a biomarker predicting responsiveness to MSCs and MSC-derived secretome and the lack of pharmacoeconomic evaluations.
Similar content being viewed by others
New therapeutic options are needed to treat life-threatening manifestations of respiratory virus infections such as coronavirus disease-19 (COVID-19) and influenza that do not resolve despite appropriate respiratory support and management with the currently recommended antivirals, immunomodulators, and antiinflammatory agents. |
The therapeutic potential of mesenchymal stem/stromal cells (MSCs) and MSC-derived products is under evaluation in a huge number of clinical trials, on the basis of the favorable results of preclinical studies. |
Although it is unclear how this investigational therapy could be integrated into the approved clinical management protocol for serious respiratory virus infections in the future, its use as an adjunctive therapy would fulfill the currently unmet need. |
This paper therefore focuses on the critical assessment of published clinical studies that have specifically tested MSCs and MSC-derived products as add-on therapy to reduce mortality and the requirement for invasive mechanical ventilation in hospitalized patients with severe and critical COVID-19 and influenza. |
Results are promising but inconclusive and solutions are proposed for improving the clinical development plan for the currently most needed add-on-therapy indication, with adequate consideration of regulatory and pharmacoeconomic issues. |
Introduction
Acute respiratory tract infections are among the commonest infectious diseases [1, 2]. Until December 2019, the most serious and prolonged outbreaks of these diseases had been observed with infections caused by strains of the influenza viruses type A and type B and by coronaviruses such as the severe acute respiratory syndrome coronavirus and the Middle East respiratory syndrome coronavirus [1,2,3]. Nonetheless, respiratory mortality associated with seasonal influenza has remained elevated worldwide even outside periods of major outbreaks caused by new strains, with global influenza-associated respiratory deaths ranging between 291,243 and 654,832 annually (4.0–8.8 deaths per 100,000 individuals), according to the latest estimate published in March 2018 [4].
The first outbreak of a pneumonia associated with a new coronavirus, the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), was reported in China in December 2019 and was followed by a rapid spreading of the infection worldwide [1, 5]. At the time of this writing (22 September 2022) the illness caused by SARS-CoV-2 and termed coronavirus disease 2019 (COVID-19) has already killed more than 6.5 million individuals and the pandemic is not over yet [6]. The devastating impacts on healthcare systems, economies, and education and social relationships have resulted in a global crisis with no precedent since the Second World War [7,8,9]. Highly efficacious vaccines have been developed in less than 1 year from SARS-CoV-2 identification [10] and the speed at which many countries have rolled out the vaccination program is unparalleled [9,10,11]. High levels of immunity induced by the mass vaccination efforts and by natural infections have greatly changed the course of the COVID-19 pandemic, but the continued generation of highly transmissible and virulent SARS-CoV-2 genetic variants capable of evading the existing level of immunity and still causing multiple waves of infections is hampering transition of COVID-19 from the pandemic to an endemic phase [12].
Like influenza viruses, SARS-CoV-2 can cause severe pneumonitis and acute respiratory distress syndrome (ARDS) with high frequency in the elderly, in immunocompromised patients, and in those with comorbidities such as obesity, diabetes, chronic cardiovascular disorders, and kidney and liver diseases [1, 2, 4, 13,14,15]. Invasive mechanical ventilation (IMV) is commonly needed in critically ill patients admitted to intensive care units (ICUs) for seasonal influenza and COVID-19, but patients with COVID-19 require longer duration of IMV and are at greater risk of mortality during the hospitalization than patients with influenza, irrespective of age, sex, and comorbidities [15]. The survivors may not recover completely and may suffer from disabling symptoms for the rest of their lives.
Because the influenza viruses and SARS-CoV-2 can cause serious pneumonitis and ARDS in the same groups of individuals, even minor outbreaks of COVID-19 occurring with a simultaneous influenza wave in the Northern Hemisphere in late autumn and winter could lead to another surge in admissions to ICUs and deaths. In addition, the prevalence of coinfections, which are associated with increased odds of ICU admission and death in those individuals [16], may also escalate because of the easing of non-pharmaceutical measures that greatly reduced the circulation of SARS-CoV-2 as well as the circulation of influenza viruses in 2020–2021, the current absence of a systematic virologic surveillance [17,18,19], and the limited effectiveness of influenza vaccines in high-risk subjects [19].
Rationale for the Potential Use of MSCs and MSC-Derived Secretome as Add-On Therapy
The severe life-threatening manifestations of influenza and COVID-19 are associated with a dysregulated immune response and hyperproduction of proinflammatory cytokines and chemokines such as interleukin (IL)-1β, IL-6, tumor necrosis factor (TNF)-α, interferon (IFN)-γ inducible protein-10, monocyte chemoattractant protein-1, and IL-8 [20,21,22,23,24,25]. The unchecked immunological/inflammatory alterations lead to further tissue damage [20,21,22,23,24,25], in addition to that caused by virus replication alone, and to increased risk of thrombosis [26] not responding to anticoagulation alone [27]. Targeting the immunological/inflammatory dysfunction with glucocorticoids, Janus kinase inhibitors, and humanized monoclonal antibodies against the IL-6 receptor (IL-6R) [28,29,30], in addition to providing maximal supportive therapy [31], significantly reduces the risk of respiratory failure and death in hospitalized patients with severe COVID-19-associated pneumonia, hypoxia, and evidence of systemic inflammation, but the residual numbers of individuals requiring IMV and dying because of respiratory failure remain elevated in the clinical trial setting [28,29,30] and in clinical practice [32]. The evaluation of the efficacy of immunomodulators and antiinflammatory compounds in severe influenza-associated pneumonia and ARDS has generated conflicting results and there is evidence of detrimental effects of glucocorticoids in influenza-related ARDS [31, 33, 34]. Effective therapeutic options are therefore needed to reduce the use of IMV and the risk of death in hospitalized patients with rapidly increasing oxygen demand and systemic inflammation who do not respond to the evidence-based therapeutic regimen currently recommended by international guidelines (Table 1) [19, 35].
Because of this unmet need, the therapeutic potential of mesenchymal stem/stromal cells (MSCs) and MSC-derived products is under evaluation in a huge number of clinical trials [36,37,38,39,40] on the basis of the results of preclinical studies that have demonstrated the ability of intravenously injected MSCs to transiently accumulate in the pulmonary circulation and to exert multiple beneficial effects, including the modulation of immunological responses, the prevention of bacterial superinfections, the promotion of the repair of damaged alveolo-capillary barriers, and the alleviation of fibrosis in the injured lungs [40,41,42,43,44,45,46], mainly through paracrine signaling [42,43,44,45]. The MSCs under evaluation are a heterogeneous population of self-renewable multipotent cells that are most commonly harvested from the perinatal tissues (umbilical cord tissue, umbilical cord blood, or placenta), the menstrual blood, adult bone marrow or adult adipose tissue of one or more healthy unrelated donor(s) and are expanded in culture to large quantities for treating many patients [39, 42, 47, 48]. The investigational therapy is either the allogeneic population of MSCs expanded in culture or its secretome, which is composed of soluble factors and extracellular vesicles such as exosomes and microvesicles [38, 45].
It is widely recognized that the allogeneic MSCs under evaluation only acquire immunomodulatory properties in inflammatory conditions [49]. The induction of the expression of a predominant immunosuppressive phenotype is known as MSC licensing and has been reported to be elicited in the circulation and at the tissue sites by IFN-γ [50, 51], particularly in the concomitant presence of one of the proinflammatory cytokines TNF-α, IL-1α, and IL-1β [52]. The importance of the licensing activity of IFN-γ is supported by the results of studies in an animal model of graft versus host disease (GVHD), where the recipients of IFN-γ–/– T lymphocytes did not respond to treatment with bone-marrow-derived MSCs and died [53].
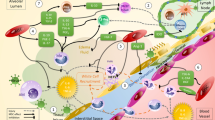
Licensed MSCs acquire the ability to generate powerful immunoregulatory effects by modulating the proliferation and function of diverse cells involved in the innate and adaptive immunity through the release of biologically active soluble molecules and extracellular vesicles, and the transfer of mitochondria via intercellular communication [49, 54] (Fig. 1). Soluble factors with immunomodulatory properties include indoleamine 2,3-dioxygenase (IDO), prostaglandin E2 (PGE2), transforming growth factor (TGF)-β, and IL-1R antagonist (IL-RA). Through the release of IDO and PGE2, MSCs can reduce the proliferation, cytotoxic activity, and cytokine production of effector T lymphocytes, and the proliferation of B lymphocytes. Importantly, MSCs can favor the differentiation and expansion of functional regulatory T lymphocytes (Treg) through IDO, PGE2, cyclooxygenase (COX)-2, and TGF-β [49, 54] (Fig. 1). Furthermore, they can promote the generation of IL-10-producing regulatory B cells (Breg) that inhibit the differentiation of effector T cells into T helper-17 (Th-17) lymphocytes [49, 54] (Fig. 1). MSCs can also block the activation of effector immune cells via cell-to-cell interaction through the association of the programmed death (PD)-1 and its ligand PD-L1 [49]. In presence of macrophage colony-stimulating factor (M-CSF), MSCs promote the differentiation of monocytes and type 1 macrophages with proinflammatory activity into M2 type macrophages with antiinflammatory and regenerative properties, which produce IL-10 and TGF-β. MSCs also inhibit the differentiation and maturation of dendritic cells and switch their profile toward a tolerogenic one by reducing their expression of the costimulatory molecules HLA-DR, CD1a, CD80, and CD83, downregulating their production of IL-12 and increasing their expression of PD-L1 [54] (Fig. 1).
Immunomodulatory properties of mesenchymal stem/stromal cells. Copyright 2021 Müller, Tunger, Wobus, von Bonin, Towers, Bornhäuser, Dazzi, Wehner, and Schmitz [54], Figure reproduced without changes under the terms of the Creative Commons Attribution License, https://creativecommons.org/licenses/by/4.0/, which permits unrestricted use, distribution, and reproduction in any medium, provided the original authors(s) and the copyright owner(s) are credited, the original publication is cited, the link to the license is given, and it is indicated if changes were made. CD cluster of differentiation, CTL cytotoxic T lymphocytes, EVs extracellular vesicles, FasL Fas ligand, HGF hepatocyte growth factor, IDO indoleamine 2,3-dioxygenase, IFN interferon, IL interleukin, IL1-RA interleukin 1 receptor antagonist, MHC major histocompatibility complex, M-CSF macrophage colony-stimulating factor, miRNA microRNA, OX40L OX40 ligand, PD-L1 programmed death-ligand 1, PGE2 prostaglandin 2, Th T helper lymphocyte, TGF transforming growth factor, TNF tumor necrosis factor, Treg T regulatory lymphocyte, TSG tumor necrosis factor-stimulated gene
In addition to the process of MSC licensing described above, another mechanism has been recently proposed to explain the immunomodulatory function of intravenously injected allogeneic MSCs [49, 55]. The infused cells would undergo apoptosis by interaction with the granules released by cytotoxic CD8 lymphocytes and natural killer (NK) cells of the host, and the apoptotic cells would be taken up by the circulating mononuclear phagocytes. This efferocytosis would induce a sort of reprogramming of the phagocytic cells of the MSC recipient, which would produce PGE2 and IDO themselves and in this manner mediate the immunosuppressive effects of MSCs. The two mechanisms may coexist because there is evidence that viable MSCs cannot be replaced with apoptotic or dead MSCs from a therapeutic perspective [56], and more studies are required to clarify this issue. In addition, it would be important to understand if the reprogramming of phagocytic cells depends at least in part on the biological activity of the extracellular vesicles of the apoptotic MSCs and whether it can also occur with the infusion of MSC-derived extracellular vesicles, which are in large part taken up by the phagocytes of the reticuloendothelial system because of their size.
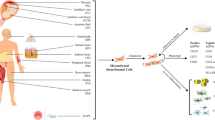
MSC tracking experiments in an animal model of infectious pneumonitis [57], using real-time, intravital imaging of the kinetics of MSCs in lung vessels, demonstrated the immediate influx of MSCs following their intravascular injection and their persistence in the alveolar capillaries for more than 24 h. Moreover, MSC administration was associated with improvements in the gas-exchange function of the alveolar-capillary barrier, resulting in increased arterial oxygen levels [57]. Figure 2 schematically shows the possible mechanisms through which intravenously injected allogeneic MSCs, or MSC-derived extracellular vesicles, can restore the impaired gas exchange and counteract the effects of the dysregulated immune response and persisting inflammation in severe and critical COVID-19 and influenza. The indicated effects of MSCs and MSC-derived extracellular vesicles are based on the immunomodulatory properties of MSCs discussed above and the results of preclinical studies in animal models of lung injury associated with influenza virus infection [58,59,60,61] or in experimentally-induced ARDS [62]. The figure also shows a possible mechanism through which MSCs become or remain not permissive to viral growth in the inflamed alveoli. This is related to their ability to express INF-stimulated genes in response to INFs such as the type I IFNs (IFN-α/β) produced by infected alveolar epithelial cells and the IFN-γ present in the inflammatory infiltrate [63].
The currently approved pharmacological treatment for severe and critical COVID-19 includes the glucocorticoid dexamethasone as standard of care, the humanized monoclonal antibody of the IgG1 class against the IL-6R tocilizumab, and the Janus kinase inhibitor of the JAK/1/JAK2 subtype baricitinib as adjunctive therapies (Table 1) [19, 35]. Dexamethasone is known to have a broad antiinflammatory activity, and the transcriptomic data on pulmonary and circulating immune cells from patients with severe COVID-19 has suggested that the therapeutic effect of this glucocorticoid in this disease may be specifically related to TNF-α, IL-1a, IL-1b, IFN-α, and IFN-γ signaling but does not involve the IL-6 pathway [64]. Tocilizumab targets IL-6-mediated signal transduction by binding to both the transmembrane and the soluble receptors, and in so doing it irreversibly blocks the proinflammatory and prothrombotic effects of IL-6 for 2–3 weeks, as well as its still desirable effects on the development of an acute-phase response against infections and on the enhancement of bacterial phagocytosis [65]. Baricitinib predominantly blocks IL-6 and INF-γ signaling and IL-10 and IFN-α signaling to a lesser extent [66], and similarly to dexamethasone, has a short half-life. Both dexamethasone and baricitinib inhibit the function of type I IFNs involved in viral clearance and must be administered in combination with antivirals in immunocompromised patients. Baricitinib also reduces, albeit to a lesser extent than other Janus kinase inhibitors [66], the desirable regulatory activity of IL-10, which is related to its ability to promote the emergence of Tregs while suppressing the development of Th-17 lymphocytes. None of these therapeutic agents has the direct effects mediated by whole MSCs and by their extracellular vesicles on the viability of alveolar epithelial cells, the repair of the alveolar-capillary barrier, and the prevention of the development of secondary bacterial infections (Fig. 2). In severe and critical influenza, the potential biological effects of whole MSCs or of their extracellular vesicles are unrivaled, because a combination of antivirals and antiinflammatory or immunoregulators is not allowed (Table 1) [19, 35].
Mechanistic rationale for investigating the clinical use of mesenchymal stem/stromal cells and their products as adjunctive therapy for the management of severe and critical coronavirus disease 2019 and influenza. The pathological mechanisms leading to alveolar damage, hypoxemia, and systemic inflammation are highlighted in red, and the counteracting effects of intravenously injected allogeneic mesenchymal stem/stromal cells that have been induced to express an antiinflammatory/immunosuppressive phenotype systemically and in the inflamed alveoli are highlighted in green. In severe and critical coronavirus disease 2019, virus replication, the proliferation of effector T lymphocytes, the release of proinflammatory cytokines, and the recruitment of leukocytes from the peripheral blood are inhibited by the recommended treatment with antivirals and the glucocorticoid dexamethasone in combination with a Janus kinase inhibitor of the JAK1/JAK2 subtype, such as baricitinib, or with the humanized antibody against the interleukin-6 receptor tocilizumab. This combination also reduces the systemic effects of viral replication and excessive inflammation, but in abolishing the acute-phase response and the IL-6 mediated enhancement of bacterial phagocytosis, the combination of antiinflammatory agents concurs to render the host more vulnerable to pulmonary and systemic infections. Key adjunctive effects of mesenchymal stem/stromal cells are the following: reestablishment of the regulatory function of subpopulations of T and B lymphocytes (Treg and Breg cells) that normally suppress excessive and deleterious immunological/inflammatory responses; activation of the mechanisms involved in the repair of the alveolar-capillary barrier via the release of soluble factors (Ang-1, HGF, and KGF) and extracellular vesicles delivering microRNAs; enhancement of the viability of alveolar epithelial cells through the transfer of healthy mitochondria by intercellular communication; prevention of the development of secondary bacterial infections by producing antimicrobial peptides and by enhancing the phagocytic activity of neutrophils and macrophages through the release of prostaglandin E2. In severe and critical influenza, where a combination of antivirals and antiinflammatory or immunoregulators is not allowed, most of the biological effects of mesenchymal stem/stromal cells highlighted in this figure would be desirable. Generated using in part ScienceSlides graphics from VisiScience Corp., licensed use. AEC alveolar epithelial cell, AMPs antimicrobial peptides, Ang angiopoietin, Breg B regulatory lymphocytes, CRP C-reactive protein, CoV coronavirus, COX cyclooxygenase, CTL cytotoxic T lymphocytes, EVs extracellular vesicles, FGF fibroblast growth factor, GCs glucocorticoids, HGF hepatocyte growth factor, IAV influenza virus, IDO indoleamine 2,3-dioxygenase, IFN interferon, IL interleukin, IL1-RA interleukin 1 receptor antagonist, IL-6R interleukin-6 receptor, ISGs interferon-stimulated genes, JAKis Janus kinase inhibitors, KGF keratinocyte growth factor, miRNA microRNA, NK natural killer, PGE2 prostaglandin 2, RLR retinoic acid-inducible gene-1-like receptor, TCR T cell receptor, TGF transforming growth factor, Th T helper lymphocyte, TLR toll-like receptor, TNF tumor necrosis factor, Treg T regulatory lymphocyte, TSG tumor necrosis factor-stimulated gene
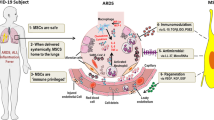
A mechanistic rationale therefore emerges for the use of MSC-based therapy as an adjunctive therapy in patients with severe and critical COVID-19 who do not respond to dexamethasone, and in patients with severe and critical influenza who show increasing oxygen demand and systemic inflammation on treatment with antivirals alone. The combination of an MSC-based therapy and glucocorticoids has been already used for the treatment of GVHD and excellent results have been reported in terms of safety and efficacy [67]. Moreover, glucocorticoids at high doses are used as standard of care in studies testing the potential additional benefits of MSC-based therapies in GVHD [68]. The potential future integration of investigational MSC-based therapy into the currently recommended therapeutic management of hospitalized adult patients with severe and critical COVID-19 or influenza is illustrated in Fig. 3.
Potential future integration of investigational therapy into the recommended therapeutic management of hospitalized adult patients with severe and critical COVID-19 or influenza, which is based on published international guidelines [19, 35, 90]. The arrows indicate potential sequential treatment strategies, where allogeneic mesenchymal stem/stromal cells or their products are administered intravenously as an adjunctive or alternative add-on therapy to prevent further disease progression and death in patients not responding to the recommended first-line treatment with dexamethasone, while receiving adequate respiratory support and the required critical care management. The asterisk indicates the allowed addition of the antiviral remdesivir in immunocompromised patients with severe coronavirus disease 2019 who require oxygen through a high-flow device or noninvasive ventilation and are receiving dexamethasone. ANI antiviral neuraminidase inhibitor, COVID-19 coronavirus disease 2019, ECMO extracorporeal membrane oxygenation, ICU intensive care unit, IL-6 interleukin-6, IMV invasive mechanical ventilation, IV intravenous, MSC mesenchymal stem/stromal cells, NIV noninvasive ventilation, WHO CPS World Health Organization Clinical Progression Scale
Aims and Methodological Approach of this Review
The focus of this review was on the status of the clinical investigations testing the potential use of MSCs and MSC-derived secretome to improve the outcome of patients with severe and critical diseases already managed according to the evidence-based therapeutic approach outlined in Table 1. The main objectives were the following: to identify progresses in the assessment of the potential added value of the investigational therapy in clinical trials, to highlight unresolved issues, and to discuss how to address them.
An extensive literature search was conducted to retrieve all articles reporting on the clinical use of MSCs and MSC-derived products as investigational therapy for lung conditions related to COVID-19 and influenza as described in Table 2. To assess if investigational therapy significantly accelerated the recovery and decreased the mortality of patients with severe or critical diseases in comparison with the recommended/standard therapeutic regimen, controlled prospective clinical trials on the use of the investigational therapy as add-on therapy and single-arm uncontrolled clinical trials on the use of the investigational therapy when the recommended/standard treatments have failed were taken into consideration. Clinical studies in patients in stable conditions on the recommended/standard treatment, clinical studies where the recommended/standard treatment was not described, clinical studies where the recommended/standard treatment did not include antivirals or glucocorticoids or immunomodulators, case series, and case reports were excluded and are reviewed elsewhere [40], together with registered but still unpublished studies.
This review was based on previously conducted studies and does not contain any new studies with human participants or animals performed by any of the authors.
Overview and Critical Assessment of Published Clinical Trial Results
As of 31 July 2022, 21 published reports were found [69,70,71,72,73,74,75,76,77,78,79,80,81,82,83,84,85,86,87,88,89], including 6 randomized, double-blind, placebo-controlled trials [71, 74, 75, 81, 82, 84], 3 randomized, open-label parallel-group studies [76, 77, 85], 6 nonrandomized prospective studies with control groups [69, 70, 72, 78, 86, 89], and 6 prospective, uncontrolled single-arm studies [73, 79, 80, 83, 87, 88] conducted in diverse countries worldwide (Table 3). A total of 20 reports [69,70,71,72,73,74,75,76,77,78,79,80,81,82,83,84,85,86,87,88] concerned treatment of patients with severe and or critical illness caused by laboratory-confirmed SARS-CoV-2 infection, but 6 of the reported studies also included patients with mild [85] or moderate [69, 70, 78, 79, 86] disease, and 1 study [87] was conducted in patients with a condition defined as moderate pneumonia by the investigators, although the reported clinical and laboratory data at baseline (Table 3) reflected a more severe stage according to international guidelines [35, 90]. The total number of patients treated with MSCs or MSC-derived products in these studies was 343 (Table 3). One report [89] concerned treatment of patients with ARDS caused by laboratory-confirmed H7N9 influenza virus infection: 17 out of 61 individuals received the investigational therapy, in addition to standard care including antivirals and glucocorticoids, while the others served as control (Table 3). The characteristics of all studies and their main findings are summarized in Tables 3, 4, 5, and 6.
The most frequent source of MSCs was the umbilical cord tissue [70,71,72,73,74,75,76, 80,81,82,83, 85,86,87], followed by the menstrual blood [78, 84, 89] and bone marrow [83, 88] (Table 4). In one study the MSC source was adipose tissue [79] (Table 4). In the report of another study [69] the MSC origin was not mentioned (Table 4). Seven reports [77, 79, 80, 83, 85, 87, 89] lacked the required description of the surface markers and multilineage differentiation ability of MSCs [91], and the only report of a study using extracellular vesicles of the exosome subtype as investigational therapy [83] did not contain the required minimal information about the isolation, analysis, and quantification of the MSC-derived product [92]. The report of the study using the secretome of menstrual-blood-derived MSCs as investigational treatment [84] lacked information about the composition of the MSC-conditioned medium, which likely contained a mix of extracellular vesicles and soluble factors [93]. The putative immunomodulatory /immunosuppressive properties of the administered MSCs were confirmed by mixed lymphocyte reaction assay only in two studies [81, 88], employing umbilical-cord- and bone-marrow-derived MSCs, respectively (Table 4). The susceptibility to virus infection of the MSCs to be injected into the circulation of an infected host was tested in a minority of the studies, although infected MSCs may have reduced survival and function as a virus reservoir in the body of the treated patients. The expression of the main cell entry receptor for SARS-CoV-2, the angiotensin-converting enzyme 2 (ACE2), by the infused MSCs was only tested in 2 out of 20 studies concerning COVID-19 [69, 78], but the results were reassuring because cells from menstrual blood [78] and those from an unreported origin [69] were marginally ACE2-positive [69] or ACE2-negative [78] (Table 4). The MSCs from an unreported origin [69] also did not express a protease involved in viral cell entry, the transmembrane serine protease type 2 (TMPRSS2). These results would be in keeping with the in vitro observations that human MSCs from fetal and adult tissues are indeed ACE2- and TMPRSS2-negative and may not be permissive to SARS-CoV-2 [94], but diverse studies have uncovered new mechanisms of viral entry into human host cells [95,96,97,98,99], and these data should be taken into account in clinical trials testing the therapeutic potential of MSCs in COVID-19. The previously demonstrated susceptibility of human MSCs of bone marrow and cord blood origin to infection with avian influenza A H5N1 virus [100] was not excluded in the only study evaluating the potential therapeutic effectiveness of MSCs in H7N9-influenza-induced ARDS [89]. Thus, the possibility that the various MSC populations infused in the studies reviewed here were infected by the hosted viruses, once injected, cannot be excluded. Nonetheless, the viral load over time was not significantly affected by MSC treatment in the randomized, double-blind, placebo-controlled trials that properly evaluated this outcome in COVID-19-associated ARDS [74, 82].
The studies testing MSCs in COVID-19 differed greatly in terms of patient selection, MSC dosage, and infusion schedule (Tables 3 and 4). The outcome measures and follow-up periods also varied greatly (Tables 3, 6), precluding the possibility to perform meaningful meta-analyses, as previously recognized by the authors of recent systematic reviews not specifically focusing on the assessment of MSCs and MSC-derived products as add-on therapy to the currently recommended standard of care [101,102,103,104]. Considering the safety outcomes, most reports did not contain complete and convincing information about the incidence of serious and non-serious adverse events and their relation to treatment (Table 5). For example, in the prospective single-arm study on severe ARDS not responding to standard treatment [73] and in the prospective single-arm study on moderate pneumonitis [87], there was a transient increase in the circulating levels of the fibrin degradation product D-dimer after MSC infusions (Table 6), which was not reported as a treatment-related adverse event although it may reflect the effects of the procoagulant activity of tissue factor (TF)/CD142-expressing MSCs from umbilical cord and placenta [105] in patients who already have an increased risk of thrombosis because of the infection. In one of these studies [87], the observed transient increase in the levels of the proinflammatory chemokine IL-6 following MSC infusion (Table 6) was also not reported as an adverse event. It is, however, reassuring to know that in the controlled clinical trial with an open-label follow-up period of up to 1 year [71, 72], patients with less severe disease who had received three doses of viable 4 × 107 umbilical-cord-derived MSCs/kg of body weight (Table 4) did not show significant reductions in the inhibition rate of neutralizing antibodies against SARS-CoV-2, changes in pulmonary function, alterations of laboratory parameters, or evidence of tumor developments in comparison with the controls (Table 6).
In terms of concomitant treatment, some investigators included drugs that were empirically used during the initial phases of the COVID-19 pandemic and that are no longer recommended by current guidelines [35, 90], such as hydroxychloroquine and various antivirals used for other infections (Table 3). Nonetheless, in most studies patients received glucocorticoids (Table 3), the current standard of care in severe and critical COVID-19 [35, 90], albeit with substantial differences in terms of glucocorticoid type, dosage, and duration of treatment. Three of these studies were randomized, double-blind, placebo-controlled trials of umbilical-cord-derived MSCs in ARDS [74, 81, 82], and only one [74] demonstrated significantly higher survival rate by day 28 and shorter time to recovery in the group of patients who received MSCs as investigational add-on therapy than in the control group (Table 6). Possible explanations for the discrepancy were MSC dosing (Table 4) and imbalance of the patients’ condition at baseline (Table 3). In the study by Lanzoni and colleagues [74], the mean number of infused cells at each infusion and in total was much higher than in the other two trials, and the cell viability was comparable to that detected in the negative study by Rebellato and colleagues [82], despite the use of thawed cells from frozen cell samples (Table 4). In the same study by Lanzoni and colleagues [74], only 4 of 12 patients (33.33%) in the MSC-treated group versus 7 of 12 patients (58.33%) in the placebo group received IMV, and the baseline levels of IL-6 were significantly higher in the control group (Table 3), suggesting that control patients were at higher risk of death than MSC-treated patients. In the study by Rebelatto and colleagues [82], in which all MSC-treated and placebo-treated patients required IMV in the ICU and received glucocorticoids and anticoagulants as concomitant therapy, the mortality rate was even higher in the group of MSC-treated patients (45.45%) than in the group of patients who received placebo (16.66%), and the MSC-treated patients also showed increased levels of the cardiac troponin I and of creatinine, suggesting further cardiac damage and renal insufficiency between day 4 and day 14 post-treatment (three infusions of 5 × 105 cells/kg of body weight every other day, starting 10.7 days, on average, after symptom onset). In the study by Monsel and colleagues [81], low level of alloimmunization developed in 3 of 21 patients (14.3%) on day 14 post-infusion but an additional 6 of the 21 MSC recipients (28.6%) already had pre-formed antibodies against the human leukocyte antigen of the infused cells before treatment. In this study, antibody-mediated loss of functional cells may contribute to explain the lack of significant effects of the investigational therapy on most efficacy outcome measures, except for a significant decrease of plasma inflammatory markers at day 14 post-infusion (Table 6), but the relative impact cannot be estimated because none of the other studies reviewed here reported data concerning possible alloimmunization.
In another randomized, double-blind, placebo-controlled trial of umbilical-cord-derived MSCs in critical COVID-19 [75], there was a significantly higher survival rate in patients treated with MSCs than in the controls (2.5 times higher overall and 4.5 times higher in patients with more than two comorbidities known to worsen disease outcome), which was associated with a significant decrease in the circulating levels of IL-6 (Table 6), but the concomitant treatment only included oseltamivir and azithromycin (Table 3), drugs not considered as effective in severe or critical COVID-19 [35, 90], and there were no significant differences between groups in terms of length of stay in the ICU and ventilator usage (Table 6). Thus, MSC treatment significantly improved survival in critical COVID-19, but this effect is known to be achievable in similar patients with the very less expensive and easier to use glucocorticoids [35, 90].
Taking into account the putative immunomodulatory/immunosuppressive properties of MSCs, it is worth noting that in most placebo-controlled studies involving severely and critically ill patients with COVID-19, investigational therapy was found to significantly reduce the levels of inflammatory markers in comparison with placebo (Table 6). This was not the case for studies involving patients with less severe COVID-19, such as the controlled clinical trial with 1-year follow-up mentioned above [71, 72], in which the administration of MSCs as add-on therapy resulted in significant radiological improvements not accompanied by significant functional or laboratory changes by day 28 (Table 6).
Considering the other comparative trials testing MSCs as investigational add-on therapy in COVID-19 [69, 70, 76,77,78, 85, 86, 88], significantly higher survival rates in the treated groups than in the control groups were reported in three of the eight studies [77, 78, 88], and the increase in survival rates was more pronounced in critically ill patients than in patients with severe disease (Table 6). In one of these studies [77], which was a randomized open-label parallel-group trial, the length of stay in ICU was also significantly lower in the MSC-treated group than in the control group of critically ill patients. In another randomized, single-blind, placebo-controlled study involving patients with mild, severe, and critical disease [85], the duration of hospital stays (primary endpoint) was significantly shorter in the MSC-treated group than in the placebo group (Table 6). For the patients with severe and critical COVID-19 enrolled in the comparative trials cited above, MSC treatment was associated with significant decreases in the levels of circulating inflammatory markers and/or coagulation markers, and with significantly shorter time to normalization of the total lymphocyte counts in MSC-treated patients than in the controls, although these improvements in laboratory parameters did not consistently translate into reduced mortality and reduced use of IMV (Table 6), possibly because of the low number of enrolled patients. Promising preliminary results were provided by Grégoire and colleagues [88], with the initial analysis of data from the only still ongoing study testing bone-marrow-derived MSCs as add-on investigational therapy in severe ARDS (Tables 3, 4). The concomitant treatment with glucocorticoids and anticoagulants was in keeping with current guidelines [35, 90] and efficacy outcome measures were in accordance with those proposed by the World Health Organization (WHO) for COVID-19 clinical research, including the use of the WHO Clinical Progression Scale to evaluate patient trajectory over the course of disease [106]. MSC-treated patients (n = 8) required high-flow oxygen therapy (n = 7) or IMV (n = 1) within 24 h of ICU admission and received three infusions of 1.5–3 × 106 clinical-grade cells/kg body weight at an average interval of 3 days starting within 2 days of ICU admission. Matched controls (n = 24) were only retrospectively selected among the patients admitted to the ICU in the same hospital, the major limitation of this study. Although a progression of disease severity was initially observed in two of the seven patients after MSC infusion, survival rate was significantly higher in the MSC-treated group than in the matched control group at day 28 (100% versus 79.2%) and at day 60 (100% versus 70.8%) (Table 6). The risk of thrombosis was also significantly reduced by the investigational treatment, as indicated by the levels of the fibrin degradation product D-dimer, which were much lower in this group than in the matched control group by day 7 (Table 6).
The two studies testing MSC-derived products in severe COVID-19 [83, 84] differed greatly in terms of study design, source of MSCs, type of MSC-derived products, and concomitant treatments. In the randomized, double-blind placebo-controlled trial testing the conditioned medium from menstrual-blood-derived MSCs as investigational therapy in severe disease [84], concomitant treatment agreed with current guidelines [35, 90] and the investigational therapy was an effective add-on as it significantly reduced mortality at day 28 and the need for intubation in the treated group in comparison with the control group (Table 6). Treatment was not apparently associated with the occurrence of adverse events, but the adverse event reporting was incomplete, and one patient discontinued after first dose for unknown causes (Table 5). The second study [83] was a prospective uncontrolled cohort study testing a single intravenous infusion of exosomes from bone-marrow-derived MSCs in patients with mild, moderate, or severe COVID-19-associated ARDS, who showed clinical deterioration for more than 72 h on treatment with the institutional standard treatment, consisting in oxygen support and administration of hydroxychloroquine and azithromycin, two drugs not recommended for the management of similar patients by current guidelines [35, 90]. During the follow-up period of 14 days, the overall survival rate was 83%. The recovery rate in the cohort of 20 patients not requiring IMV at baseline was 75%, but the other patients worsened to the point of requiring IMV. In the cohort of patients with severe ARDS at baseline, all three patients remained critically ill, still requiring IMV (Table 6). Overall, a single exosome infusion was associated with a significant improvement of laboratory data, including the markers of inflammation and markers of thrombosis, such as D-dimer (Table 6). The occurrence of treatment-related adverse events was specifically evaluated over a period of 72 h after exosome administration and no adverse events were reported. The cases of pulmonary embolism, acute renal failure, worsening of hypoxic respiratory failure requiring intubation, and four deaths, all occurring at post-treatment days 4–13, were not reported as related to the exosome infusion (Table 5). Thus, investigational therapy was apparently safe and effective at improving the outcome of patients with mild or moderate COVID-19-associated ARDS in comparison with the outcome reported in literature for similar patients [107], even though a large fraction of intravenously injected exosomes is immediately taken up by the mononuclear phagocytes of the reticuloendothelial system in the liver and spleen [108]. However, better outcomes have been reported in clinical trials, for all levels of ARDS severity, with the use of glucocorticoids [107], the current standard of care in severe and critical COVID-19 [35, 90], and the addition of Janus kinase inhibitors and monoclonal antibodies against IL-6R, if required [35, 90].
Concerning the nonrandomized, open-label, parallel-group study that tested the therapeutic potential of MSCs from menstrual blood when added to standard care in H7N9-influenza-virus-induced ARDS [89] (Table 3), investigational therapy consisted in the intravenous administration of 1 × 106 90–95% viable MSCs 3–4 times (Table 4), depending on patients’ consent. The group of MSC-treated patients had a significant increase in survival rate and more marked improvement of the inflammatory parameters and D-dimer at discharge than the control group that received standard care alone (Table 6). In addition to the supportive therapy for critically ill patients with multiorgan dysfunction, the concomitant treatment in the MSC-treated group and in the control group included the antivirals recommended by international guidelines (oseltamivir or peramivir) [19] in all patients. Over 50% of them in both study arms also received glucocorticoids, which are not recommended for the treatment of severe influenza because of possible detrimental effects on the outcome [19]. However, the mortality rate in the control group (54.5%) was only slightly higher than that reported in literature [107] for similar patients with severe ARDS [107]. The more than threefold lower mortality rate (17.6%) in the MSC-treated group is impressive but may be explained at least in part by the lower proportion of critically ill patients with severe renal injury at study entry in this group in comparison with the control group (Tables 3 and 6). There was no report of adverse events related to the MSC infusions (Table 5). A 5-year follow-up period was limited to four survivors in the MSC-treated arm and no harmful effects of the MSC transplantation were observed in these subjects (Table 6).
Conclusions
The results of published clinical studies on the therapeutic potential of MSCs and MSC-derived products in COVID-19 and influenza suggest that MSCs and MSC-derived products may significantly increase the survival of hospitalized patients with severe and critical disease and that this beneficial effect may be related to the putative immunomodulatory/immunosuppressive properties of MSCs and their secretome. However, in COVID-19-associated ARDS, similar or better outcomes have been reported in clinical trials with glucocorticoids alone [107], the current standard of care in severe and critical COVID-19 [35, 90], with further improvement attainable with the addition of Janus kinase inhibitors and monoclonal antibodies against IL-6R if required to block disease progression according to current guidelines [90, 107]. The studies reviewed here have not consistently demonstrated that adding MSCs or MSC-derived products to this currently recommended therapeutic regimen can reduce the use of IMV and the risk of death in patients still showing rapidly increasing oxygen demand and systemic inflammation despite appropriate therapeutic management. Overall, increased survival rates were observed in 5 of 12 prospective comparative trials that tested MSCs or MSC-derived secretome as add-on therapy in severe and critical COVID-19. Four of these trials were randomized, double-blind, and placebo controlled. The standard of care included glucocorticoids and anticoagulants in all these trials and remdesivir, glucocorticoids, monoclonal antibodies against IL-6R, and anticoagulants in some recent studies, but the proportion of patients receiving these therapeutics in each study varied greatly across studies, and most reports do not contain information about dosing and duration of treatment. The results of the only available study about the use of MSCs as add-on therapy for influenza-associated ARDS are promising, particularly because no immunomodulator/antiinflammatory treatment is currently recommended for severe and critical disease, but the study is a small open-label trial with imbalances between the arms, which may contribute to explaining the superior efficacy outcome attributed to the MSC infusion. The positive results reported in this study and in 5 of the 12 comparative studies that evaluated the therapeutic added values of MSCs and MSC-derived products in severe and critical COVID-19 must be confirmed in controlled clinical trials conducted in compliance with the current Good Manufacturing Practice and Good Clinical Practice guidelines. Compliance with these guidelines is a condition for the generation of data that can be submitted to the regulatory authorities when seeking the mandatory authorization for the use of a therapeutic candidate outside the setting of a clinical investigation [109].
Implications for Future Research
To comply with the current international guidelines for the clinical development of cell and cell-based therapies, several issues need to be addressed in the design and conduction of future clinical trials and in the reporting of the clinical data. Possible solutions for improving the clinical development plan are proposed in tabular form (Table 7). The key issue is how to choose the starting time of the MSC-based add-on therapy. This challenge is difficult to overcome because the studies reviewed here suggest that blood biomarkers of ongoing inflammation recognized thus far are not consistently predictive of a response to MSCs and MSC-derived secretome when the recommended therapeutic regimen is insufficient to block disease progression. It may be useful to evaluate one of the most recently identified specific biomarkers of prolonged inflammation and dysregulated immune activation, the elevated level of plasma soluble urokinase plasminogen activator receptor, which has been found to predict disease progression in hospitalized patients with COVID-19 and other viral pneumonia more accurately than other inflammatory parameters [109,110,111]. Alternatively, or concomitantly, a composite score such as the COVID-19-associated Hyperinflammation Syndrome score [112, 113] may be introduced to evaluate the risk of further disease progression in critically ill patients already receiving the recommended immunomodulatory/antiinflammatory regimen (Table 7), although this score system still needs to be fully validated.
Finally, it should be considered that the high costs of MSC-based therapy would still represent an obstacle to its clinical acceptance [114], even if its effectiveness at reducing the healthcare expenditures associated with the prolonged hospitalizations of critically ill patients were conclusively demonstrated. The costs of obtaining clinical-grade allogeneic MSCs varies depending on the MSC source [114], and the use of cell-free MSC-derived products, such as exosomes and other extracellular vesicles, can greatly increase these costs. The use of properly isolated MSC-derived exosomes would render the costs of the MSC-based therapy particularly high because of the expensive isolation procedure and the necessity to produce a huge number of clinical-grade vesicles to overcome problems with the biodistribution of these nanoparticles after intravenous infusion [108, 115]. One of the possible solutions may be to avoid their systemic administration [116], but the first published small single-arm study on the investigational use of repeated doses of aerosolized MSC-derived extracellular vesicles in severe COVID-19 [117] has not provided favorable results in terms of efficacy outcome, and the total dosage of extracellular vesicles (2 × 108 nanoparticles per day for 5 consecutive days) is higher than that used for intravenous administration (6 × 108) [83, 118].
Another possible solution would be the systemic administration of exosomes that have been engineered to escape phagocytosis and/or to specifically target the lungs [119], but these manipulations would likely affect some of their desired biological activities against viral respiratory tract infections and would require a lot of preclinical research work before first investigational use in humans, without reducing or even increasing the costs of the final product for the indication discussed here. Thus, the decision to go ahead with a rigorous clinical development of an MSC-based therapy as add-on therapy for the treatment of severe and critical COVID-19, influenza, and other severe viral respiratory infections should also be based on pharmacoeconomic considerations, and studies with an adequate evaluation of the cost-effectiveness of these investigational therapies are highly demanded (Table 7).
References
Clementi N, Ghosh S, De Santis M, et al. Viral respiratory pathogens and lung injury. Clin Microbiol Rev. 2021;34(3):e00103-e120. https://doi.org/10.1128/CMR.00103-20.
Khan M, Adil SF, Alkhathlan HZ, et al. COVID-19: a global challenge with old history, epidemiology and progress so far. Molecules. 2021;26(1):39. https://doi.org/10.3390/molecules26010039.
V’kovski P, Kratzel A, Steiner S, Stalder H, Thiel V. Coronavirus biology and replication: implications for SARS-CoV-2. Nat Rev Microbiol. 2021;19(3):155–70. https://doi.org/10.1038/s41579-020-00468-6.
Iuliano AD, Roguski KM, Chang HH, Global Seasonal Influenza-associated Mortality Collaborator Network, et al. Estimates of global seasonal influenza-associated respiratory mortality: a modelling study. Lancet. 2018;391(10127):1285–300. https://doi.org/10.1016/S0140-6736(17)33293-2.
To KK, Sridhar S, Chiu KH, et al. Lessons learned 1 year after SARS-Cov-2 emergence leading to COVID-19 pandemic. Emerg Microbes Infect. 2021;10(1):507–35. https://doi.org/10.1080/22221751.2021.1898291.
Johns Hopkins University and Medicine. Johns Hopkins Coronavirus Resource Center. Updated 22 September. https://coronavirus.jhu.edu/map.html. Accessed 22 Sept 2022.
Han E, Tan MMJ, Turk E, et al. Lessons learnt from easing COVID-19 restrictions: an analysis of countries and regions in Asia Pacific and Europe. Lancet. 2020;396(10261):1525–34. https://doi.org/10.1016/S0140-6736(20)32007-9.
Nicola M, Alsafi Z, Sohrabi C, et al. The socio-economic implications of the coronavirus pandemic (COVID-19): a review. Int J Surg. 2020;78:185–93. https://doi.org/10.1016/j.ijsu.2020.04.018.
Mattoli S. Filling the gap until full vaccine deployment in the war on coronavirus disease-19. Infect Dis Ther. 2021;10(1):27–34. https://doi.org/10.1007/s40121-020-00394-3.
Fauci AS. The story behind COVID-19 vaccines. Science. 2021;372(6538):109. https://doi.org/10.1126/science.abi8397.
Ledford H. Six months of COVID vaccines: what 1.7 billion doses have taught scientists. Nature. 2021;594:164–7. https://doi.org/10.1038/d41586-021-01505-x.
Cascella M, Rajnik M, Aleem A, Dulebohn SC, Di Napoli R. Features, evaluation, and treatment of coronavirus (COVID-19). 2022 Jun 30. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2022.
Short KR, Kroeze EJBV, Fouchier RAM, Kuiken T. Pathogenesis of influenza-induced acute respiratory distress syndrome. Lancet Infect Dis. 2014;14(1):57–69. https://doi.org/10.1016/S1473-3099(13)70286-X.
Tzotzos SJ, Fischer B, Fischer H, Zeitlinger M. Incidence of ARDS and outcomes in hospitalized patients with COVID-19: a global literature survey. Crit Care. 2020;24(1):516. https://doi.org/10.1186/s13054-020-03240-7.
Cobb NL, Sathe NA, Duan KI, et al. Comparison of clinical features and outcomes in critically ill patients hospitalized with COVID-19 versus influenza. Ann Am Thorac Soc. 2021;18(4):632–40. https://doi.org/10.1513/AnnalsATS.202007-805OC.
Swets MC, Russell CD, Harrison EM, et al. SARS-CoV-2 co-infection with influenza viruses, respiratory syncytial virus, or adenoviruses. Lancet. 2022;399(10334):1463–4. https://doi.org/10.1016/S0140-6736(22)00383-X.
Oh DY, Buda S, Biere B, et al. Trends in respiratory virus circulation following COVID-19-targeted nonpharmaceutical interventions in Germany, January–September 2020: analysis of national surveillance data. Lancet Reg Health Eur. 2021;6:100112. https://doi.org/10.1016/j.lanepe.2021.100112.
Olsen SJ, Winn AK, Budd AP, et al. Changes in influenza and other respiratory virus activity during the COVID-19 pandemic—United States, 2020–2021. MMWR Morb Mortal Wkly Rep. 2021;70(29):1013–9. https://doi.org/10.15585/mmwr.mm7029a1.
Uyeki TM, Bernstein HH, Bradley JS, et al. Clinical practice guidelines by the Infectious Diseases Society of America: 2018 update on diagnosis, treatment, chemoprophylaxis, and institutional outbreak management of seasonal influenza. Clin Infect Dis. 2019;68(6):e1–47. https://doi.org/10.1093/cid/ciy866 [Erratum in: Clin Infect Dis. 2019 May 2;68(10):1790. https://doi.org/10.1093/cid/ciz044].
Darwish I, Mubareka S, Liles WC. Immunomodulatory therapy for severe influenza. Expert Rev Anti Infect Ther. 2011;9(7):807–22. https://doi.org/10.1586/eri.11.56.
Fajgenbaum DC, June CH. Cytokine storm. N Engl J Med. 2020;383:2255–73. https://doi.org/10.1056/NEJMra2026131.
Del Valle DM, Kim-Schulze S, Huang HH, et al. An inflammatory cytokine signature predicts COVID-19 severity and survival. Nat Med. 2020;26(10):1636–43. https://doi.org/10.1038/s41591-020-1051-9.
Laing AG, Lorenc A, Del Molino Del Barrio I, et al. A dynamic COVID-19 immune signature includes associations with poor prognosis. Nat Med. 2020;26(10):1623–35. https://doi.org/10.1038/s41591-020-1038-6 [Erratum in: Nat Med. 2020 Oct; 26(10):1663. https://doi.org/10.1038/s41591-020-1079-x. Erratum in: Nat Med. 2020 Dec;26(12):1951. https://doi.org/10.1038/s41591-020-01186-5].
Lucas C, Wong P, Klein J, et al. Longitudinal analyses reveal immunological misfiring in severe COVID-19. Nature. 2020;584(7821):463–9. https://doi.org/10.1038/s41586-020-2588-y.
Reynolds D, Vazquez Guillamet C, Day A, et al. Comprehensive immunologic evaluation of bronchoalveolar lavage samples from human patients with moderate and severe seasonal influenza and severe COVID-19. J Immunol. 2021;4(207):1229–38. https://doi.org/10.4049/jimmunol.2100294.
Stals MAM, Grootenboers MJJH, van Guldener C, Dutch COVID & Thrombosis Coalition (DCTC), et al. Risk of thrombotic complications in influenza versus COVID-19 hospitalized patients. Res Pract Thromb Haemost. 2021;5(3):412–20. https://doi.org/10.1002/rth2.12496.
Chowdary P. COVID-19 coagulopathy—what should we treat? Exp Physiol. 2022;107(7):749–58. https://doi.org/10.1113/EP089404.
RECOVERY Collaborative Group. Dexamethasone in hospitalized patients with Covid-19. N Engl J Med. 2021;384:693–704. https://doi.org/10.1056/NEJMoa2021436.
World Health Organization (WHO) Rapid Evidence Appraisal for COVID-19 Therapies (REACT) Working Group. Association between administration of IL-6 antagonists and mortality among patients hospitalized for COVID-19: a meta-analysis. JAMA. 2021;326(6):499–518. https://doi.org/10.1001/jama.2021.11330.
Ely EW, Ramanan AV, Kartman CE, et al., COV-BARRIER Study Group. Efficacy and safety of baricitinib plus standard of care for the treatment of critically ill hospitalised adults with COVID-19 on invasive mechanical ventilation or extracorporeal membrane oxygenation: an exploratory, randomised, placebo-controlled trial. Lancet Respir Med. 2022;10(4):327–36. https://doi.org/10.1016/S2213-2600(22)00006-6 [Erratum in: Lancet Respir Med. 2022 Feb 11. https://doi.org/10.1016/S2213-2600(22)00057-1].
Meyer NJ, Gattinoni L, Calfee CS. Acute respiratory distress syndrome. Lancet. 2021;398:622–37. https://doi.org/10.1016/S0140-6736(21)00439-6.
Lambermont B, Rousseau AF, Seidel L, et al. Outcome improvement between the first two waves of the coronavirus disease 2019 pandemic in a single tertiary-care hospital in Belgium. Crit Care Explor. 2021;3(5):e0438. https://doi.org/10.1097/CCE.0000000000000438.
Liu Q, Zhou YH, Yang ZQ. The cytokine storm of severe influenza and development of immunomodulatory therapy. Cell Mol Immunol. 2016;13(1):3–10. https://doi.org/10.1038/cmi.2015.74.
Hui DS, Lee N, Chan PK, Beigel JH. The role of adjuvant immunomodulatory agents for treatment of severe influenza. Antiviral Res. 2018;150:202–16. https://doi.org/10.1016/j.antiviral.2018.01.002.
COVID-19 treatment guidelines panel. Coronavirus disease 2019 (COVID-19) treatment guidelines. National Institutes of Health. https://www.covid19treatmentguidelines.nih.gov/. Accessed 12 Feb 2023.
Khoury M, Cuenca J, Cruz FF, Figueroa FE, Rocco PRM, Weiss DJ. Current status of cell-based therapies for respiratory virus infections: applicability to COVID-19. Eur Respir J. 2020;55(6):2000858. https://doi.org/10.1183/13993003.00858-2020.
Qu W, Wang Z, Hare JM, et al. Cell-based therapy to reduce mortality from COVID-19: systematic review and meta-analysis of human studies on acute respiratory distress syndrome. Stem Cells Transl Med. 2020;9(9):1007–22. https://doi.org/10.1002/sctm.20-0146.
Gorman E, Millar J, McAuley D, O’Kane C. Mesenchymal stromal cells for acute respiratory distress syndrome (ARDS), sepsis, and COVID-19 infection: optimizing the therapeutic potential. Expert Rev Respir Med. 2021;15(3):301–24. https://doi.org/10.1080/17476348.2021.1848555.
Spinetti G, Avolio E, Madeddu P. Treatment of COVID-19 by stage: any space left for mesenchymal stem cell therapy? Regen Med. 2021;16(5):477–94. https://doi.org/10.2217/rme-2020-0189.
Yao W, Shi L, Zhang Y, Dong H, Zhang Y. Mesenchymal stem/stromal cell therapy for COVID-19 pneumonia: potential mechanisms, current clinical evidence, and future perspectives. Stem Cell Res Ther. 2022;13(1):124. https://doi.org/10.1186/s13287-022-02810-6.
Gotts JE, Matthay MA. Mesenchymal stem cells and acute lung injury. Crit Care Clin. 2011;27(3):719–33. https://doi.org/10.1016/j.ccc.2011.04.004.
Barczyk M, Schmidt M, Mattoli S. Stem cell-based therapy in idiopathic pulmonary fibrosis. Stem Cell Rev Rep. 2015;11(4):598–620. https://doi.org/10.1007/s12015-015-9587-7.
Pittenger MF, Discher DE, Péault BM, Phinney DG, Hare JM, Caplan AI. Mesenchymal stem cell perspective: cell biology to clinical progress. NPJ Regen Med. 2019;2(4):22. https://doi.org/10.1038/s41536-019-0083-6.
Chow L, Johnson V, Impastato R, Coy J, Strumpf A, Dow S. Antibacterial activity of human mesenchymal stem cells mediated directly by constitutively secreted factors and indirectly by activation of innate immune effector cells. Stem Cells Transl Med. 2020;9(2):235–49. https://doi.org/10.1002/sctm.19-0092.
Xiong J, Chen L, Zhang L, Bao L, Shi Y. Mesenchymal stromal cell-based therapy: a promising approach for severe COVID-19. Cell Transplant. 2021;30:963689721995455. https://doi.org/10.1177/0963689721995455.
Samarelli AV, Tonelli R, Heijink I, et al. Dissecting the role of mesenchymal stem cells in idiopathic pulmonary fibrosis: cause or solution. Front Pharmacol. 2021;12:692551. https://doi.org/10.3389/fphar.2021.692551.
Samsonraj RM, Raghunath M, Nurcombe V, Hui JH, van Wijnen AJ, Cool SM. Concise review: multifaceted characterization of human mesenchymal stem cells for use in regenerative medicine. Stem Cells Transl Med. 2017;6(12):2173–85. https://doi.org/10.1002/sctm.17-0129.
Fitzsimmons REB, Mazurek MS, Soos A, Simmons CA. Mesenchymal stromal/stem cells in regenerative medicine and tissue engineering. Stem Cells Int. 2018;2018:8031718. https://doi.org/10.1155/2018/8031718.
Cheung TS, Bertolino GM, Giacomini C, Bornhäuser M, Dazzi F, Galleu A. Mesenchymal stromal cells for graft versus host disease: mechanism-based biomarkers. Front Immunol. 2020;25(11):1338. https://doi.org/10.3389/fimmu.2020.01338.
Ryan JM, Barry F, Murphy JM, Mahon BP. Interferon-gamma does not break, but promotes the immunosuppressive capacity of adult human mesenchymal stem cells. Clin Exp Immunol. 2007;149(2):353–63. https://doi.org/10.1111/j.1365-2249.2007.03422.x.
Krampera M, Cosmi L, Angeli R, et al. Role for interferon-gamma in the immunomodulatory activity of human bone marrow mesenchymal stem cells. Stem Cells. 2006;24(2):386–98. https://doi.org/10.1634/stemcells.2005-0008.
Ren G, Zhang L, Zhao X, et al. Mesenchymal stem cell-mediated immunosuppression occurs via concerted action of chemokines and nitric oxide. Cell Stem Cell. 2008;2(2):141–50. https://doi.org/10.1016/j.stem.2007.11.014.
Polchert D, Sobinsky J, Douglas G, et al. IFN-gamma activation of mesenchymal stem cells for treatment and prevention of graft versus host disease. Eur J Immunol. 2008;38(6):1745–55. https://doi.org/10.1002/eji.200738129.
Müller L, Tunger A, Wobus M, et al. Immunomodulatory properties of mesenchymal stromal cells: an update. Front Cell Dev Biol. 2021;9:637725. https://doi.org/10.3389/fcell.2021.637725.
Pang SHM, D’Rozario J, Mendonca S, et al. Mesenchymal stromal cell apoptosis is required for their therapeutic function. Nat Commun. 2021;12(1):6495. https://doi.org/10.1038/s41467-021-26834-3.
Weiss DJ, English K, Krasnodembskaya A, Isaza-Correa JM, Hawthorne IJ, Mahon BP. The neurobiology of mesenchymal stromal cells affects therapeutic efficacy. Front Immunol. 2019;4(10):1228. https://doi.org/10.3389/fimmu.2019.01228.
Masterson CH, Tabuchi A, Hogan G, et al. Intra-vital imaging of mesenchymal stromal cell kinetics in the pulmonary vasculature during infection. Sci Rep. 2021;11(1):5265. https://doi.org/10.1038/s41598-021-83894-7.
Li Y, Xu J, Shi W, et al. Mesenchymal stromal cell treatment prevents H9N2 avian influenza virus-induced acute lung injury in mice. Stem Cell Res Ther. 2016;7:159. https://doi.org/10.1186/s13287-016-0395-z.
Chan MC, Kuok DI, Leung CY, et al. Human mesenchymal stromal cells reduce influenza A H5N1-associated acute lung injury in vitro and in vivo. Proc Natl Acad Sci USA. 2016;113:3621–6. https://doi.org/10.1073/pnas.1601911113.
Khatri M, Richardson LA, Meulia T. Mesenchymal stem cell-derived extracellular vesicles attenuate influenza virus-induced acute lung injury in a pig model. Stem Cell Res Ther. 2018;9:17. https://doi.org/10.1186/s13287-018-0774-8.
Loy H, Kuok DIT, Hui KPY, et al. Therapeutic implications of human umbilical cord mesenchymal stromal cells in attenuating influenza A (H5N1) virus-associated acute lung injury. J Infect Dis. 2019;219:186–96. https://doi.org/10.1093/infdis/jiy478.
Dutra Silva J, Su Y, Calfee CS, et al. Mesenchymal stromal cell extracellular vesicles rescue mitochondrial dysfunction and improve barrier integrity in clinically relevant models of ARDS. Eur Respir J. 2021;58(1):2002978. https://doi.org/10.1183/13993003.02978-2020.
Wu X, Dao Thi VL, Huang Y, et al. Intrinsic immunity shapes viral resistance of stem cells. Cell. 2018;172(3):423-438.e25. https://doi.org/10.1183/13993003.02978-2020.
Sharma A. Inferring molecular mechanisms of dexamethasone therapy in severe COVID-19 from existing transcriptomic data. Gene. 2021;788:145665. https://doi.org/10.1016/j.gene.2021.145665.
Tanaka T, Narazaki M, Kishimoto T. IL-6 in inflammation, immunity, and disease. Cold Spring Harb Perspect Biol. 2014;6(10):a016295. https://doi.org/10.1101/cshperspect.a016295.
McInnes IB, Byers NL, Higgs RE, et al. Comparison of baricitinib, upadacitinib, and tofacitinib mediated regulation of cytokine signaling in human leukocyte subpopulations. Arthritis Res Ther. 2019;21(1):183. https://doi.org/10.1186/s13075-019-1964-1.
Kebriaei P, Isola L, Bahceci E, et al. Adult human mesenchymal stem cells added to corticosteroid therapy for the treatment of acute graft-versus-host disease. Biol Blood Marrow Transplant. 2009;15(7):804–11. https://doi.org/10.1016/j.bbmt.2008.03.012.
Kurtzberg J, Prockop S, Teira P, et al. Allogeneic human mesenchymal stem cell therapy (remestemcel-L, Prochymal) as a rescue agent for severe refractory acute graft-versus-host disease in pediatric patients. Biol Blood Marrow Transplant. 2014;20(2):229–35. https://doi.org/10.1016/j.bbmt.2013.11.001.
Leng Z, Zhu R, Hou W, et al. Transplantation of ACE2- mesenchymal stem cells improves the outcome of patients with COVID-19 pneumonia. Aging Dis. 2020;11(2):216–28. https://doi.org/10.14336/AD.2020.0228.
Meng F, Xu R, Wang S, et al. Human umbilical cord-derived mesenchymal stem cell therapy in patients with COVID-19: a phase 1 clinical trial. Signal Transduct Target Ther. 2020;5(1):172. https://doi.org/10.1038/s41392-020-00286-5.
Shi L, Huang H, Lu X, et al. Effect of human umbilical cord-derived mesenchymal stem cells on lung damage in severe COVID-19 patients: a randomized, double-blind, placebo-controlled phase 2 trial. Signal Transduct Target Ther. 2021;6(1):58. https://doi.org/10.1038/s41392-021-00488-5.
Shi L, Yuan X, Yao W, et al Human mesenchymal stem cells treatment for severe COVID-19: 1-year follow-up results of a randomized, double-blind, placebo-controlled trial. EBioMedicine. 2022;75:103789. https://doi.org/10.1016/j.ebiom.2021.103789.
Iglesias M, Butrón P, Torre-Villalvazo I, et al. Mesenchymal stem cells for the compassionate treatment of severe acute respiratory distress syndrome due to COVID 19. Aging Dis. 2021;12(2):360–70. https://doi.org/10.14336/AD.2020.1218.
Lanzoni G, Linetsky E, Correa D, et al. Umbilical cord mesenchymal stem cells for COVID-19 acute respiratory distress syndrome: a double-blind, phase 1/2a, randomized controlled trial. Stem Cells Transl Med. 2021;10(5):660–73. https://doi.org/10.1002/sctm.20-0472.
Dilogo IH, Aditianingsih D, Sugiarto A, et al. Umbilical cord mesenchymal stromal cells as critical COVID-19 adjuvant therapy: a randomized controlled trial. Stem Cells Transl Med. 2021;10:1279–87. https://doi.org/10.1002/sctm.21-0046.
Shu L, Niu C, Li R, et al. Treatment of severe COVID-19 with human umbilical cord mesenchymal stem cells. Stem Cell Res Ther. 2020;11(1):361. https://doi.org/10.1186/s13287-020-01875-5.
Adas G, Cukurova Z, Yasar KK, et al. The systematic effect of mesenchymal stem cell therapy in critical covid-19 patients: a prospective double controlled trial. Cell Transplant. 2021;30:9636897211024942. https://doi.org/10.1177/09636897211024942.
Xu X, Jiang W, Chen L, et al. Evaluation of the safety and efficacy of using human menstrual blood-derived mesenchymal stromal cells in treating severe and critically ill COVID-19 patients: an exploratory clinical trial. Clin Transl Med. 2021;11(2):e297. https://doi.org/10.1002/ctm2.297.
Sánchez-Guijo F, García-Arranz M, López-Parra M, et al. Adipose-derived mesenchymal stromal cells for the treatment of patients with severe SARS-CoV-2 pneumonia requiring mechanical ventilation. A proof of concept study. EClinicalMedicine. 2020;25:100454. https://doi.org/10.1016/j.eclinm.2020.100454.
Guo Z, Chen Y, Luo X, He X, Zhang Y, Wang J. Administration of umbilical cord mesenchymal stem cells in patients with severe COVID-19 pneumonia. Crit Care. 2020;24(1):420. https://doi.org/10.1186/s13054-020-03142-8.
Monsel A, Hauw-Berlemont C, Mebarki M, APHP STROMA–CoV-2 Collaborative Research Group, et al. Treatment of COVID-19-associated ARDS with mesenchymal stromal cells: a multicenter randomized double-blind trial. Crit Care. 2022;26(1):48. https://doi.org/10.1186/s13054-022-03930-4.
Rebelatto CLK, Senegaglia AC, Franck CL, et al. Safety and long-term improvement of mesenchymal stromal cell infusion in critically COVID-19 patients: a randomized clinical trial. Stem Cell Res Ther. 2022;13(1):122. https://doi.org/10.1186/s13287-022-02796-1.
Sengupta V, Sengupta S, Lazo A, Woods P, Nolan A, Bremer N. Exosomes derived from bone marrow mesenchymal stem cells as treatment for severe COVID-19. Stem Cells Dev. 2020;29(12):747–54. https://doi.org/10.1089/scd.2020.0080.
Fathi-Kazerooni M, Fattah-Ghazi S, Darzi M, et al. Safety and efficacy study of allogeneic human menstrual blood stromal cells secretome to treat severe COVID-19 patients: clinical trial phase I and II. Stem Cell Res Ther. 2022;13(1):96. https://doi.org/10.1186/s13287-022-02771-w.
Zhu R, Yan T, Feng Y, et al. Mesenchymal stem cell treatment improves outcome of COVID-19 patients via multiple immunomodulatory mechanisms. Cell Res. 2021;31(12):1244–62. https://doi.org/10.1038/s41422-021-00573-y.
Wei F, Kong D, Li T, et al. Efficacy and safety of umbilical cord mesenchymal stem cells for the treatment of patients with COVID-19. Clinics (São Paulo). 2021;76:e2604. https://doi.org/10.6061/clinics/2021/e2604.
Sharma A, Kulkarni R, Sane H, et al. Phase 1 clinical trial for intravenous administration of mesenchymal stem cells derived from umbilical cord and placenta in patients with moderate COVID-19 virus pneumonia: results of stage 1 of the study. Am J Stem Cells. 2022;11(3):37–55.
Grégoire C, Layios N, Lambermont B, et al. Bone marrow-derived mesenchymal stromal cell therapy in severe COVID-19: preliminary results of a phase I/II clinical trial. Front Immunol. 2022;13:932360. https://doi.org/10.3389/fimmu.2022.932360.
Chen J, Hu C, Chen L, et al. Clinical study of mesenchymal stem cell treatment for acute respiratory distress syndrome induced by epidemic influenza A (H7N9) infection: a hint for COVID-19 treatment. Engineering (Beijing). 2020;6(10):1153–61. https://doi.org/10.1016/j.eng.2020.02.006.
Agarwal A, Rochwerg B, Lamontagne F, et al. A living WHO guideline on drugs for covid-19. BMJ. 2020;370:m3379. https://doi.org/10.1136/bmj.m3379.
Dominici M, Le Blanc K, Mueller I, et al. Minimal criteria for defining multipotent mesenchymal stromal cells. The International Society for Cellular Therapy position statement. Cytotherapy. 2006;8(4):315–7. https://doi.org/10.1080/14653240600855905.
Théry C, Witwer KW, Aikawa E, et al. Minimal information for studies of extracellular vesicles 2018 (MISEV2018): a position statement of the International Society for Extracellular Vesicles and update of the MISEV2014 guidelines. J Extracell Vesicles. 2018;7(1):1535750. https://doi.org/10.1080/20013078.2018.1535750.
Bozorgmehr M, Gurung S, Darzi S, et al. Endometrial and menstrual blood mesenchymal stem/stromal cells: biological properties and clinical application. Front Cell Dev Biol. 2020;9(8):497. https://doi.org/10.3389/fcell.2020.00497.
Avanzini MA, Mura M, Percivalle E, et al. Human mesenchymal stromal cells do not express ACE2 and TMPRSS2 and are not permissive to SARS-CoV-2 infection. Stem Cells Transl Med. 2021;10(4):636–42. https://doi.org/10.1002/sctm.20-0385.
Cantuti-Castelvetri L, Ojha R, Pedro LD, et al. Neuropilin-1 facilitates SARS-CoV-2 cell entry and infectivity. Science. 2020;370(6518):856–60. https://doi.org/10.1126/science.abd2985.
Daly JL, Simonetti B, Klein K, et al. Neuropilin-1 is a host factor for SARS-CoV-2 infection. Science. 2020;370(6518):861–5. https://doi.org/10.1126/science.abd3072.
Wang K, Chen W, Zhang Z, et al. CD147-spike protein is a novel route for SARS-CoV-2 infection to host cells. Signal Transduct Target Ther. 2020;5(1):283. https://doi.org/10.1038/s41392-020-00426-x.
Wang S, Qiu Z, Hou Y, et al. AXL is a candidate receptor for SARS-CoV-2 that promotes infection of pulmonary and bronchial epithelial cells. Cell Res. 2021;31(2):126–40. https://doi.org/10.1038/s41422-020-00460-y.
Gu Y, Cao J, Zhang X, et al. Receptome profiling identifies KREMEN1 and ASGR1 as alternative functional receptors of SARS-CoV-2. Cell Res. 2022;32(1):24–37. https://doi.org/10.1038/s41422-021-00595-6 [Erratum in: Cell Res. 2022 Jun;32(6):600. https://doi.org/10.1038/s41422-022-00654-6].
Thanunchai M, Kanrai P, Wiboon-Ut S, Puthavathana P, Hongeng S, Thitithanyanont A. Tropism of avian influenza A (H5N1) virus to mesenchymal stem cells and CD34+ hematopoietic stem cells. PLoS ONE. 2013;8(12):e81805. https://doi.org/10.1371/journal.pone.0081805.
Zhang M, Yan X, Shi M, et al. Safety and efficiency of stem cell therapy for COVID-19: a systematic review and meta-analysis. Glob Health Res Policy. 2022;7(1):19. https://doi.org/10.1186/s41256-022-00251-5.
Chen CY, Chen WC, Hsu CK, Chao CM, Lai CC. The clinical efficacy and safety of mesenchymal stromal cells for patients with COVID-19: a systematic review and meta-analysis of randomized controlled trials. J Infect Public Health. 2022;15(8):896–901. https://doi.org/10.1016/j.jiph.2022.07.001.
Yao W, Dong H, Qi J, Zhang Y, Shi L. Safety and efficacy of mesenchymal stem cells in severe/critical patients with COVID-19: a systematic review and meta-analysis. EClinicalMedicine. 2022;51:101545. https://doi.org/10.1016/j.eclinm.2022.101545.
Kirkham AM, Bailey AJM, Monaghan M, et al. Updated living systematic review and meta-analysis of controlled trials of mesenchymal stromal cells to treat COVID-19: a framework for accelerated synthesis of trial evidence for rapid approval-FASTER approval. Stem Cells Transl Med. 2022;11(7):675–87. https://doi.org/10.1093/stcltm/szac038.
Moll G, Ankrum JA, Olson SD, Nolta JA. Improved MSC minimal criteria to maximize patient safety: a call to embrace tissue factor and hemocompatibility assessment of MSC products. Stem Cells Transl Med. 2022;11(1):2–13. https://doi.org/10.1093/stcltm/szab005.
WHO Working Group on the Clinical Characterisation and Management of COVID-19 infection. A minimal common outcome measure set for COVID-19 clinical research. Lancet Infect Dis. 2020;20(8):e192–7. https://doi.org/10.1016/S1473-3099(20)30483-7 [Erratum in: Lancet Infect Dis. 2020 Oct;20(10):e250. https://doi.org/10.1016/S1473-3099(20)30637-X].
Gorman EA, O’Kane CM, McAuley DF. Acute respiratory distress syndrome in adults: diagnosis, outcomes, long-term sequelae, and management. Lancet. 2022;400(10358):1157–70. https://doi.org/10.1016/S0140-6736(22)01439-8.
Wiklander OP, Nordin JZ, O’Loughlin A, et al. Extracellular vesicle in vivo biodistribution is determined by cell source, route of administration and targeting. J Extracell Vesicles. 2015;20(4):26316. https://doi.org/10.3402/jev.v4.26316.
Levy O, Kuai R, Siren EMJ, et al. Shattering barriers toward clinically meaningful MSC therapies. Sci Adv. 2020;6(30):eaba6884. https://doi.org/10.1126/sciadv.aba6884.
Velissaris D, Dimopoulos G, Parissis J, et al. Prognostic role of soluble urokinase plasminogen activator receptor at the emergency department: a position paper by the Hellenic Sepsis Study Group. Infect Dis Ther. 2020;9(3):407–16. https://doi.org/10.1007/s40121-020-00301-w.
Arnold DT, Attwood M, Barratt S, et al. Predicting outcomes of COVID-19 from admission biomarkers: a prospective UK cohort study. Emerg Med J. 2021;38(7):543–8. https://doi.org/10.1136/emermed-2020-210380.
Velissaris D, Lagadinou M, Paraskevas T, et al. Evaluation of plasma soluble urokinase plasminogen activator receptor levels in patients with COVID-19 and non-COVID-19 pneumonia: an observational cohort study. J Clin Med Res. 2021;13(9):474–8. https://doi.org/10.14740/jocmr4579.
Webb BJ, Peltan ID, Jensen P, et al. Clinical criteria for COVID-19-associated hyperinflammatory syndrome: a cohort study. Lancet Rheumatol. 2020;2(12):e754–63. https://doi.org/10.1016/S2665-9913(20)30343-X.
Yildirim M, Halacli B, Yuce D, Gunegul Y, Ersoy EO, Topeli A. Assessment of admission COVID-19 associated hyperinflammation syndrome score in critically-ill COVID-19 patients. J Intensive Care Med. 2023;38(1):70–7. https://doi.org/10.1177/08850666221131265.
Golchin A. Cell-based therapy for severe COVID-19 patients: clinical trials and cost-utility. Stem Cell Rev Rep. 2021;17(1):56–62. https://doi.org/10.1007/s12015-020-10046-1.
Patel S, Schmidt KF, Farhoud M, et al. In vivo tracking of [89Zr]Zr-labeled engineered extracellular vesicles by PET reveals organ-specific biodistribution based upon the route of administration. Nucl Med Biol. 2022;112–113:20–30. https://doi.org/10.1016/j.nucmedbio.2022.06.004.
Zhu YG, Shi MM, Monsel A, et al. Nebulized exosomes derived from allogenic adipose tissue mesenchymal stromal cells in patients with severe COVID-19: a pilot study. Stem Cell Res Ther. 2022;13(1):220. https://doi.org/10.1186/s13287-022-02900-5.
Sengupta V, Sengupta S, Lazo A Jr, Hicok KC, Moseley T. Response to Lim et al. re: “Exosomes derived from bone marrow mesenchymal stem cells as treatment for severe COVID-19.” Stem Cells Dev. 2020;29(14):879–81. https://doi.org/10.1089/scd.2020.0095.
He J, Ren W, Wang W, et al. Exosomal targeting and its potential clinical application. Drug Deliv Transl Res. 2022;12(10):2385–402. https://doi.org/10.1007/s13346-021-01087-1.
Mocchi M, Bari E, Marrubini G, et al. Freeze-dried mesenchymal stem cell-secretome pharmaceuticalization: optimization of formulation and manufacturing process robustness. Pharmaceutics. 2021;13(8):1129. https://doi.org/10.3390/pharmaceutics13081129.
Acknowledgements
Funding
No external funding or sponsorship was received for this review or publication of this article. The journal’s Rapid Service fee was funded by the authors.
Authorship
All named authors meet the International Committee of Medical Journal Editors (ICMJE) criteria for authorship for this article, take responsibility for the integrity of the work as a whole, and have given approval for this version to be published.
Author Contributions
Sabrina Mattoli conceptualized the review paper. Matthias Schmidt performed the literature search. Both authors independently assessed the retrieved articles and agreed on the final evaluation. Sabrina Mattoli wrote the original draft. Matthias Schmidt critically revised the work. Both authors read and approved the final manuscript.
Disclosures
Sabrina Mattoli is consultant to the European Commission and to the Eureka Association and is shareholder in Novartis Pharma AG and Pfizer Inc. Matthias Schmidt is named as co-inventor on a submitted patent application concerning the generation of engineered exosomes for targeted delivery of biologics to the lungs.
Compliance with Ethics Guidelines
This review was based on previously conducted studies and does not contain any new studies with human participants or animals performed by any of the authors.
Data Availability
All data generated or analyzed during this study are included in this published article.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Mattoli, S., Schmidt, M. Investigational Use of Mesenchymal Stem/Stromal Cells and Their Secretome as Add-On Therapy in Severe Respiratory Virus Infections: Challenges and Perspectives. Adv Ther 40, 2626–2692 (2023). https://doi.org/10.1007/s12325-023-02507-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12325-023-02507-z