Abstract
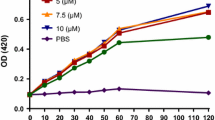
Antimicrobial peptides are promising anti-infective agent candidates because they have a broad antimicrobial spectrum and bioactivity and are unlikely to elicit antibiotic resistance. The bogorols represent a new cationic antibiotic peptide and possess great therapeutic potential because of their bioactivity and precise mode of action. Here, we report that Bogorol B-JX (BBJX), a peptide previously isolated from Brevibacillus laterosporus JX-5 by us, has significant antibacterial and antitumor activities in vitro. BBJX was found to inhibit methicillin-resistant Staphylococcus aureus (MRSA) at 2.5 µg/mL with distinct mechanisms of action from those against Bacillus bombyseptieus and Escherichia coli. It penetrates MRSA membrane with little visible destruction and binds to genomic DNA. BBJX could inhibit the proliferation of human histiocytic lymphoma cell line U-937 and ConA-activated spleen cells at 5 µg/mL, but was not cytotoxic to the Jurkat cells, resting spleen cells or differentiated macrophage-like U-937 immunocytes. Moreover, BBJX caused apoptosis of U-937 cells by opening the mitochondrial permeability transition pore and stimulating the production of reactive oxygen species. Taken together, these studies provided basis for future medical application of the bogorols.








Similar content being viewed by others
References
Andrews JM, Andrews JM (2001) Determination of minimum inhibitory concentrations. J Antimicrob Chemother 1 (48 Suppl):5–16. doi: 10.1093/jac/48.suppl_1.5
Barsby T, Kelly MT, Gagné SM, Andersen RJ (2001) Bogorol a produced in culture by a Marine Bacillus sp. Reveals a novel template for cationic peptide antibiotics. Org Lett 3:437–440. doi: 10.1021/ol006942q
Barsby T, Warabi K, Sørensen D et al (2006) The bogorol family of antibiotics: template-based structure elucidation and a new approach to positioning enantiomeric pairs of amino acids. J Org Chem 71:6031–6037. doi: 10.1021/jo060667p
Brogden KA (2005) Antimicrobial peptides: pore formers or metabolic inhibitors in bacteria? Nat Rev Microbiol 3:238–250. doi: 10.1038/nrmicro1098
Feliu L, Oliveras G, Cirac AD et al (2010) Antimicrobial cyclic decapeptides with anticancer activity. Peptides 31:2017–2026. doi: 10.1016/j.peptides.2010.07.027
Gaspar D, Salomé Veiga A, Castanho MARB (2013) From antimicrobial to anticancer peptides. A review. Front Microbiol. 4
Guo TL, Miller MA, Shapiro IM, Shenker BJ (1998) Mercuric chloride induces apoptosis in human T lymphocytes: evidence of mitochondrial dysfunction. Toxicol Appl Pharmacol 153:250–257. doi: 10.1006/taap.1998.8549
Hancock RE (1997) Peptide antibiotics. The Lancet 349:418–422. doi: 10.1016/S0140-6736(97)80051-7
Hancock REW, Diamond G (2000) The role of cationic antimicrobial peptides in innate host defences. Trends Microbiol 8:402–410. doi: 10.1016/S0966-842X(00)01823-0
Insug O, Datar S, Koch CJ et al (1997) Mercuric compounds inhibit human monocyte function by inducing apoptosis: evidence for formation of reactive oxygen species, development of mitochondrial membrane permeability transition and loss of reductive reserve. Toxicology 124:211–224. doi: 10.1016/S0300-483X(97)00153-4
Jiang H, Wang X, Xiao C et al (2015) Antifungal activity of Brevibacillus laterosporus JX-5 and characterization of its antifungal components. World J Microbiol Biotechnol. doi: 10.1007/s11274-015-1912-4
Kim H, Jang JH, Kim SC, Cho JH (2014) De novo generation of short antimicrobial peptides with enhanced stability and cell specificity. J Antimicrob Chemother 69:121–132. doi: 10.1093/jac/dkt322
Krauss S, Buttgereit F, Brand MD (1999) Effects of the mitogen concanavalin A on pathways of thymocyte energy metabolism. Biochim Biophys Acta 1412:129–138. doi: 10.1016/S0005-2728(99)00058-4
Leuschner C, Hansel W (2004) Membrane disrupting lytic peptides for cancer treatments. Curr Pharm Des 10:2299–2310. doi: 10.2174/1381612043383971
Lopes G, Pinto E, Andrade PB, Valentão P (2013) Antifungal activity of phlorotannins against dermatophytes and yeasts: approaches to the mechanism of action and influence on Candida albicans virulence factor. PLoS ONE. doi: 10.1371/journal.pone.0072203
Lowy F (2003) Antimicrobial resistance: the example of Staphylococcus aureus. J Clin Invest 111:1265–1273. doi: 10.1172/JCI200318535.In
Lu Y, Zhang T-F, Shi Y et al (2016) PFR peptide, one of the antimicrobial peptides identified from the derivatives of lactoferrin, induces necrosis in leukemia cells. Sci Rep 6:20823. doi: 10.1038/srep20823
Meyer JE, Harder J (2007) Antimicrobial peptides in oral cancer. Curr Pharm Des 13:3119–3130. doi: 10.2174/138161207782110372
Moniruzzaman M, Alam JM, Dohra H, Yamazaki M (2015) Antimicrobial peptide lactoferricin B-induced rapid leakage of internal contents from single giant unilamellar vesicles. BioChemistry 54:5802–5814. doi: 10.1021/acs.biochem.5b00594
Muñoz A, López-García B, Marcos JF (2006) Studies on the mode of action of the antifungal hexapeptide PAF26. Antimicrob Agents Chemother 50:3847–3855. doi: 10.1128/AAC.00650-06
Nguyen LT, Haney EF, Vogel HJ (2011) The expanding scope of antimicrobial peptide structures and their modes of action. Trends Biotechnol 29:464–472. doi: 10.1016/j.tibtech.2011.05.001
O’Connell KMG, Hodgkinson JT, Sore HF et al (2013) Combating multidrug-resistant bacteria: current strategies for the discovery of novel antibacterials. Angew Chemie Int Ed Engl 52:10706–10733. doi: 10.1002/anie.201209979
Oreopoulos J, Epand RF, Epand RM, Yip CM (2010) Peptide-induced domain formation in supported lipid bilayers: direct evidence by combined atomic force and polarized total internal reflection fluorescence microscopy. Biophys J 98:815–823. doi: 10.1016/j.bpj.2009.12.4327
Paphitou NI (2013) Antimicrobial resistance: Action to combat the rising microbial challenges. Int J Antimicrob Agents 42:S25–S28. doi: 10.1016/j.ijantimicag.2013.04.007
Papo N, Shai Y (2005) Host defense peptides as new weapons in cancer treatment. Cell Mol Life Sci 62:784–790. doi: 10.1007/s00018-005-4560-2
Ran S, Thorpe PE, Horpe PHET (2002) Phosphatidylserine is a marker of tumor vasculature and a potential target for cancer imaging and therapy. Int J Radiat Oncol Biol Phys 54:1479–1484
Riedl S, Rinner B, Schaider H et al (2014) Killing of melanoma cells and their metastases by human lactoferricin derivatives requires interaction with the cancer marker phosphatidylserine. BioMetals 27:981–997. doi: 10.1007/s10534-014-9749-0
Risso A, Zanetti M, Gennaro R (1998) Cytotoxicity and apoptosis mediated by two peptides of innate immunity. Cell Immunol 189:107–115. doi: 10.1006/cimm.1998.1358
Rivas-Santiago B, Santiago CE, Castañeda-Delgado JE et al (2013) Activity of LL-37, CRAMP and antimicrobial peptide-derived compounds E2, E6 and CP26 against Mycobacterium tuberculosis. Int J Antimicrob Agents 41:143–148. doi: 10.1016/j.ijantimicag.2012.09.015
Rokitskaya TI, Kolodkin NI, Kotova EA, Antonenko YN (2011) Indolicidin action on membrane permeability: carrier mechanism versus pore formation. Biochim Biophys Acta 1808:91–97. doi: 10.1016/j.bbamem.2010.09.005
Schreer A, Tinson C, Sherry JP, Schirmer K (2005) Application of Alamar Blue/5-carboxyfluorescein diacetate acetoxymethyl ester as a noninvasive cell viability assay in primary hepatocytes from rainbow trout. Anal Biochem 344:76–85. doi: 10.1016/j.ab.2005.06.009
Schröder-Borm H, Bakalova R, Andrä J (2005) The NK-lysin derived peptide NK-2 preferentially kills cancer cells with increased surface levels of negatively charged phosphatidylserine. FEBS Lett 579:6128–6134. doi: 10.1016/j.febslet.2005.09.084
Spector AA, Yorek MA (1985) Membrane lipid composition and cellular function. J Lipid Res 26:1015–1035
Stoian I, Oros A, Moldoveanu E (1996) Minireview apoptosis and free radicals. Biochem Mol Med 59:93–97
Walsh C (2003) Where will new antibiotics come from? Nat Rev Microbiol 1:65–70. doi: 10.1038/nrmicro727
Yamashita T, Kuranaga T, Inoue M (2015) Solid-phase total synthesis of bogorol A: Stereocontrolled construction of thermodynamically unfavored (E)-2-amino-2-butenamide. Org Lett 17:2170–2173. doi: 10.1021/acs.orglett.5b00769
Yan J, Wang K, Dang W et al (2013) Two hits are better than one: membrane-active and DNA binding-related double-action mechanism of NK-18, a novel antimicrobial peptide derived from mammalian NK-lysin. Antimicrob Agents Chemother 57:220–228. doi: 10.1128/AAC.01619-12
Yang L, Harroun TA, Weiss TM et al (2001) Barrel-Stave model or Toroidal model? A case study on melittin pores. Biophys J 81:1475–1485. doi: 10.1016/S0006-3495(01)75802-X
Acknowledgements
We thank Dr. Carla Jarrett (VBDI, UGA) for providing the Jurkat cell lines (original Jurkat and J45.01 ATCCCRL-1990) and mouse spleen cells used in the antitumor experiment. This work was supported by the Agriculture Public Welfare Program of China [Grant No. 201503112-10], Funds of Shandong “Double Tops” Program, and the Medicine and Health Science Technology Development Program of Shan dong, China [Grant No.2016WS0612].
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Jiang, H., Ji, C., Sui, J. et al. Antibacterial and antitumor activity of Bogorol B-JX isolated from Brevibacillus laterosporus JX-5. World J Microbiol Biotechnol 33, 177 (2017). https://doi.org/10.1007/s11274-017-2337-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11274-017-2337-z