Abstract
Background
Interferon regulatory factor 6 (IRF6) has a key function in palate fusion during palatogenesis during embryonic development, and mutations in IRF6 cause orofacial clefting disorders.
Methods and results
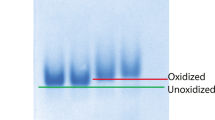
The in silico analysis of IRF6 is done to obtain leads for the domain boundaries and subsequently the sub-cloning of the N-terminal domain of IRF6 into the pGEX-2TK expression vector and successfully optimized the overexpression and purification of recombinant glutathione S-transferase-fused NTD-IRF6 protein under native conditions. After cleavage of the GST tag, NTD-IRF6 was subjected to protein folding studies employing Circular Dichroism and Intrinsic fluorescence spectroscopy at variable pH, temperature, and denaturant. CD studies showed predominantly alpha-helical content and the highest stability of NTD-IRF6 at pH 9.0. A comparison of native and renatured protein depicts loss in the secondary structural content. Intrinsic fluorescence and quenching studies have identified that tryptophan residues are majorly present in the buried areas of the protein and a small fraction was on or near the protein surface. Upon the protein unfolding with a higher concentration of denaturant urea, the peak of fluorescence intensity decreased and red shifted, confirming that tryptophan residues are majorly present in a more polar environment. While regulating IFNβ gene expression during viral infection, the N-terminal domain binds to the promoter region of Virus Response Element-Interferon beta (VRE-IFNβ). Along with the protein folding analysis, this study also aimed to identify the DNA-binding activity and determine the binding affinities of NTD-IRF6 with the VRE-IFNβ promoter region. The protein–DNA interaction is specific as demonstrated by gel retardation assay and the kinetics of molecular interactions as quantified by Biolayer Interferometry showed a strong affinity with an affinity constant (KD) value of 7.96 × 10–10 M.
Conclusion
NTD-IRF6 consists of a mix of α-helix and β-sheets that show temperature-dependent cooperative unfolding between 40 °C and 55 °C. Urea-induced unfolding shows moderate tolerance to urea as the mid-transition concentration of urea (Cm) is 3.2 M. The tryptophan residues are majorly buried as depicted by fluorescence quenching studies. NTD-IRF6 has a specific and high affinity toward the promoter region of VRE-IFNβ.





Similar content being viewed by others
Availability of data and materials
Not applicable.
Abbreviations
- NTD:
-
N-terminal domain
- SEC:
-
Size exclusion chromatography
- GST:
-
Glutathione S-transferase
- IFNβ:
-
Interferon beta
References
Taniguchi T, Ogasawara K, Takaoka A, Tanaka N (2001) IRF family of transcription factors as regulators of host defense. Annu Rev Immunol 19:623–655
Kousa YA, Schutte BC (2016) Toward an orofacial gene regulatory network. Dev Dyn 245(3):220–232
Ingraham CR, Kinoshita A, Kondo S, Yang B, Sajan S et al (2006) Abnormal skin, limb and craniofacial morphogenesis in mice deficient for interferon regulatory factor 6 (Irf6). Nat Genet 38(11):1335–1340
Oberbeck N, Pham VC, Webster JD, Reja R, Huang CS et al (2019) The RIPK4–IRF6 signalling axis safeguards epidermal differentiation and barrier function. Nature 574(7777):249–253
Rhea L, Canady FJ, Le M, Reeb T, Canady JW et al (2020) Interferon regulatory factor 6 is required for proper wound healing in vivo. Dev Dyn 249(4):509–522
Bailey CM, Hendrix MJ (2008) IRF6 in development and disease: a mediator of quiescence and differentiation. Cell Cycle 7(13):1925–1930
Yan Y et al (2023) The NOTCH-RIPK4-IRF6-ELOVL4 Axis Suppresses Squamous Cell Carcinoma. Cancers (Basel) 15(3):737
Botti E, Spallone G, Moretti F, Marinari B, Pinetti V et al (2011) Developmental factor IRF6 exhibits tumor suppressor activity in squamous cell carcinomas. Proc Natl Acad Sci USA 108(33):13710–13715
Kwa MQ, Nguyen T, Huynh J, Ramnath D, De Nardo D et al (2014) Interferon regulatory factor 6 differentially regulates Toll-like receptor 2-dependent chemokine gene expression in epithelial cells. JBC 289(28):19758–19768
Ramnath D, Tunny K, Hohenhaus DM, Pitts CM, Bergot AS et al (2015) TLR3 drives IRF6-dependent IL-23p19 expression and p19/EBI3 heterodimer formation in keratinocytes. Immunol Cell Bio 93(9):771–779
Kondo S, Schutte BC, Richardson RJ, Bjork BC, Knight AS et al (2002) Mutations in IRF6 cause Van der Woude and popliteal pterygium syndromes. Nat Genet 32(2):285–289
Escalante CR, Yie J, Thanos D, Aggarwal AK (1998) Structure of IRF-1 with bound DNA reveals determinants of interferon regulation. Nature 391(6662):103–106
Escalante CR, Nistal-Villán E, Shen L, García-Sastre A, Aggarwal AK (2007) Structure of IRF-3 bound to the PRDIII-I regulatory element of the human interferon-beta enhancer. Mol Cell 26(5):703–716
Fujii Y, Shimizu T, Kusumoto M, Kyogoku Y, Taniguchi T, Hakoshima T (1999) Crystal structure of an IRF-DNA complex reveals novel DNA recognition and cooperative binding to a tandem repeat of core sequences. Embo J 18(18):5028–5041
De Ioannes P, Escalante CR, Aggarwal AK (2011) Structures of apo IRF-3 and IRF-7 DNA binding domains: effect of loop L1 on DNA binding. Nucleic Acids Res 39(16):7300–7307
Goodbourn S, Maniatis T (1988) Overlapping positive and negative regulatory domains of the human beta-interferon gene. Proc Natl Acad Sci USA 85(5):1447–1451
Thanos D, Maniatis T (1995) Virus induction of human IFN beta gene expression requires the assembly of an enhanceosome. Cell 83(7):1091–1100
Little HJ, Rorick NK, Su LI, Baldock C, Malhotra S et al (2009) Missense mutations that cause Van der Woude syndrome and popliteal pterygium syndrome affect the DNA-binding and transcriptional activation functions of IRF6. Hum Mol Genet 18(3):535–545
Sinha BK, Meher P, Singh SK, Snehi S, Kashav T (2023) Homology modeling, cloning, overexpression, and GST-tagged protein purification of full-length and C-terminal domain of Human Interferon Regulatory Factor 6. Indian J Appl Pure Biol 38(2):908–919
Laskowski RA, MacArthur MW, Moss DS, Thornton JM (1993) PROCHECK: a program to check the stereochemical quality of protein structures. J Appl Crystallogr 26(2):283–291
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227(5259):680–685
Apiyo DO (2017) Biolayer interferometry (Octet) for label-free biomolecular interaction sensing, In: Schasfoort R.B.M., editor. Handbook of Surface Plasmon Resonance, 2nd edn. Royal Society of Chemistry London, UK, pp. 356–3 97
Lakowicz JR (2006) Principles of fluorescence spectroscopy. Springer, New York
Francis DM, Page R (2010) Strategies to optimize protein expression in E. coli. Curr Protoc Protein Sci 61(1):5.24.1-5.24.29
Ghisaidoobe AB, Chung SJ (2014) Intrinsic tryptophan fluorescence in the detection and analysis of proteins: a focus on Förster resonance energy transfer techniques. Int J Mol Sci 15(12):22518–22538
Eftink MR, Hagaman KA (1985) Fluorescence quenching of the buried tryptophan residue of cod parvalbumin. Biophys Chem 22(3):173–180
Albrecht C, Lakowicz JR (2008) Principles of fluorescence spectroscopy. Springer, New York
Barrows JK, Van Dyke MW (2022) Biolayer interferometry for DNA-protein interactions. PLoS ONE 17(2):e0263322
Acknowledgements
Human IRF6 cloned in pUC57 vector, a kind gift by Prof. W. Royer (UMASS, U.S.A.), and Human VRE-IFNβ cloned in pTZ57R/T vector was kindly gifted by Dr. Krishna Prakash, CUSB, Bihar, India. For funding, B.K.S. is thankful to the CSIR fellowship, Dr. T.K. is thankful to the DBT Bio-CARe project, and P.M. is thankful to the UGC non-net fellowship for supporting this work.
Funding
Not applicable.
Author information
Authors and Affiliations
Contributions
BKS has performed all the experiments and in silico work along with writing the original draft of the manuscript. DK gave valuable suggestions for purification, CD analysis, and fluorescence experiments. PM and SK have contributed to NTD cloning. KP gave valuable suggestions for the EMSA assay. SG guided and provided the facility for purification and characterization studies. TK was responsible for the concept, planning the experiments, and discussing and finalizing the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflicts of interest.
Ethical approval
This article does not contain any studies with human participants performed by any of the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Sinha, B.K., Kumar, D., Meher, P. et al. Biophysical and functional characterization of N-terminal domain of Human Interferon Regulatory Factor 6. Mol Biol Rep 51, 380 (2024). https://doi.org/10.1007/s11033-024-09205-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11033-024-09205-1