Abstract
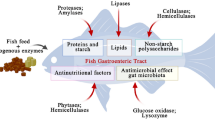
This study investigated the potential use of acid-fermented chicken silage on growth performance, digestive enzymes, liver and intestinal histology in Oreochromis mossambicus (Mozambique tilapia) (Peters 1852). Five silage-based diets were formulated by replacing fishmeal at 0% (control), 25%, 50%, 75% and 100%, thereafter denoted as D1, D2, D3, D4 and D5, respectively. The control diet (0%) contained 40% of fishmeal while D5 contained no fishmeal. The diets contained the same protein (30.06%), fat (15%) and energy levels (18 MJ kg−1). Silage (29.31% protein and 10.97% fat) fed alone and a commercial feed (35% protein and 2.97% fat) were designated as D6 and D7, respectively. Each diet was randomly assigned to triplicate groups of O. mossambicus (6.06 ± 0.27 g) stocked in 400-L fiberglass tanks (50 fish/tank). Fish were fed with the diets to apparent satiation twice daily for 45 days. Among the silage-based diets, weight gained was significantly higher (P < 0.05) in D4 (18.34 ± 1.51 g) and D5 (20.86 ± 1.95 g) than in D1 (14.28 ± 1.21 g), D2 (14.19 ± 1.51 g) and D3 (14.43 ± 1.34 g). The highest specific growth rates were found in D4 (1.10 ± 0.19%) and D5 (1.23 ± 0.04%) than in D1 (0.96 ± 0.22%), D2 (0.74 ± 0.06%) and D3 (0.92 ± 0.04%). The best feed conversion ratio was found in D4 (2.55 ± 0.27) and D5 (2.41 ± 0.32) compared to D1 (3.13 ± 0.38), D2 (3.50 ± 0.34) and D3 (3.26 ± 0.42). Apparent digestibility coefficient for protein values was higher in D4 (93.50 ± 2.96%) and D5 (92.55 ± 3.90%) than in the control (86.68 ± 2.80%). However, the best growth rates were recorded in the D6 and D7 diets. The highest protease, amylase and lipase activities were recorded in D6 and D7 followed by D5 and D4. No pathological alterations were observed in the liver and intestines of fish among dietary treatments. This study confirmed that acid-fermented chicken silage could serve as an alternative and cheaper protein source, as well as complete diet for O. mossambicus.



Similar content being viewed by others
Data availability
The data that supports the findings of this study is available on request from the corresponding author.
Code availability
Not applicable.
References
AOAC (Association of Official Analytical Chemists) International (2012) Official Methods of Analysis of AOAC International, 19th edn. Association of Analytical Communities, Gaithersburg
Baeverfjord G, Krogdahl A (1996) Development and regression of soybean meal induced enteritis in Atlantic salmon, Salmo salar L., distal intestine: a comparison with the intestines of fasted fish. J Fish Dis 19:375–387. https://doi.org/10.1046/j.1365-2761.1996.d01-92.x|
Bernfeld P (1951) Enzymes of starch degradation and synthesis. In: Nord FF (ed) Advances in Enzymology and Related Areas of Molecular Biology. John Wiley and Sons, Inc., Hoboken, NJ, USA
Bezerra RS, Lins EJF, Alencar RB, Paiva PMG, Chaves MEC, Luana CBB, Carvalho LB Jr (2005) Alkaline proteinase from intestines of Nile tilapia (Oreochromis niloticus). Process Biochem 40:1829–1834. https://doi.org/10.1016/j.procbio.2004.06.066
DAFF (Department of Agriculture, Forestry and Fisheries) (2019) Aquaculture yearbook. Department of Agriculture, Forestry and Fisheries, South Africa, pp 214
De Almeida LC, Lundstedt LM, Mores G (2006) Digestive enzyme response of Tambaqui (Colossoma macropomum) fed on different levels of protein and lipid. Aquac Nutr 12:443–450. https://doi.org/10.1111/j.1365-2095.2006.00446.x
El-Sayed AFM (2006) Tilapia Culture. CABI Publishers, Oxfordshire, UK
Fagbenro O, Jauncey K, Haylor G (1994) Nutritive value of diets containing dried lactic acid fermented fish silage and soybean meal for juvenile Oreochromis niloticus and Clarias gariepinus. Aquat Living Resour 7:79–85. https://doi.org/10.1051/alr:1994010
FAO (Food and Agriculture Organization of the UN) (2016) The State of World Fisheries and Aquaculture. Contributing to food security and nutrition for all. Italy, Rome, pp 200
Fardiaz D, Markakis P (1981) Degradation of phytic acid in oncom (fermented peanut press cake). J Food Sci 46(2):523–525. https://doi.org/10.1111/j.1365-2621.1981.tb04901.x
Folch J, Lees M, Stanley GHS (1957) A simple method for the isolation and purification of total lipids from animal tissues. J Biol Chem 226:497–509. https://doi.org/10.1016/S0021-9258(18)64849-5
Francis G, Makkar HPS, Becker K (2001) Antinutritional factors present in plant-derived alternate fish feed ingredients and their effects in fish. Aquaculture 199:197–227. https://doi.org/10.1016/S0044-8486(01)00526-9
Furukawa A, Tsukahara H (1966) On the acid digestion method for determination of chromic oxide as an indicator substance in the study of digestibility in fish. Bull Japan Soc Sci Fish 32:502–506
Gatlin DM, Barrows FT, Brown P, Dabrowski K, Gaylord TG, Hardy RW, Herman E, Hu G, Krogdahl Å, Nelson R, Overturf K, Rust M, Sealey W, Skonberg D, Souza EJ, Stone D, Wilson R, Wurtele E (2007) Expanding the utilization of sustainable plant products in aquafeeds: a review. Aquac Res 38(6):551–579. https://doi.org/10.1111/j.1365-2109.2007.01704.x
German DP, Horn MH, Gawlicka A (2004) Digestive enzyme activities in herbivorous and carnivorous prickleback fishes (Teleostei: Stichaeidae): ontogenetic, dietary, and phylogenetic effects. Physiol Biochem Zool 77:789–804. https://doi.org/10.1086/422228
Ghosh K, Roy M, Kar N, Ringø E (2010) Gastrointestinal bacteria in Rohu, Labeo rohita (Actinopterygii: cypriniformes: Cyprinidae): scanning electron microscopy and bacteriological study. Acta Ichthyol Piscat 40(2):129–135. https://doi.org/10.3750/AIP2010.40.2.05
Gupta RK, Gangoliya SS, Singh NK (2015) Reduction of phytic acid and enhancement of bioavailable micronutrients in food grains. J Food Sci Technol 52:676–684. https://doi.org/10.1007/s13197-013-0978-y
Haider MS, Ashraf M, Azmat H, Khalique A, Javid A, Atique U, Zia M, Iqbal KJ, Akram S (2016) Nutritive evaluation of fish acid silage in Labeo rohita fingerlings feed. J Appl Anim Res 44(1):158–164. https://doi.org/10.1080/09712119.2015.1021811
Hardy RW (2010) Utilization of plant proteins in fish diets: effects of global demand and supplies of fishmeal. Aquac Res 41(5):770–776. https://doi.org/10.1111/j.1365-2109.2009.02349.x
Hidalgo MC, Urea E, Sanz A (1999) Comparative study of digestive enzymes in fish with different nutritional habits. Proteolytic Amylase Act Aquac 170:267–283
Hlophe SN, Moyo NAG (2013) The aquaculture potential of Tilapia rendalli in relation to its feeding habits and digestive capabilities. Phys Chem Earth 66:33–37. https://doi.org/10.1016/j.pce.2013.09.006
Jauncey K, Tacon AGJ, Jackson AJ (1983) The quantitative essential amino acid requirements of Oreochromis (Sarotherodon) mossambicus. In: Fishelson L, Yaron Z (eds) Proceedings of the international symposium on Tilapia in Aquaculture. Tel Aviv University, Tel Aviv, pp 328–337
Kaviraj A, Mondal K, Mukhopadhyay PK, Turchini GM (2013) Impact of fermented mulberry leaf and fish silage in diet formulation of Indian major carp (Labeo rohita). Proc Zool Soc 66(1):64–73. https://doi.org/10.1007/s12595-012-0052-1
Kirk JR (1979) Nutritional changes upon processing. In: Clydesdale F (ed) Food Science and Nutrition. Current Issues and Answers. Prentice Hall, Inc., Englewood Cliffs, NJ, pp 62
Lazzari R, Neto JR, Pedron FA, Loro VL, Pretto A, Gioda CR (2010) Protein sources and digestive enzyme activities in jundiá (Rhamdia quelen). Sci Agric 67:259–266. https://doi.org/10.1590/S0103-90162010000300002
Lee SM (2002) Apparent digestibility coefficients of various feed ingredients for juvenile and grower rockfish (Sebastes schlegeli). Aquaculture 207:79–95. https://doi.org/10.1016/S0044-8486(01)00751-7
Lee YJ, Kim BK, Lee BH, Jo KI, Lee NK, Chung CH, Lee YC, Lee JW (2008) Purification and characterization of cellulase produced by Bacillus amyoliquefaciens DL-3 utilizing rice hull. Bioresour Technol 99:378–386. https://doi.org/10.1016/j.biortech.2006.12.013
Makkar HPS, Francis G, Becker K (2007) Bioactivity of phytochemicals in some lesser-known plants and their effects and potential applications in livestock and aquaculture production systems. Animal 1:1371–1391. https://doi.org/10.1017/S1751731107000298
Manikandavelu D, Neethiselvan J, Jagatheeshan G, Sundarsanam K (1992) Use of fermented ensilage based diet in enhancing the growth of common carp (Cyprinus carpio var communis). Fish Technol 29(2):111–113
Markweg H, Lang MS, Wagner F (1995) Dodecanoic acid inhibition of lipase from Acinetobacter sp. OPA 55. Enzyme Microb Technol 17:512–516. https://doi.org/10.1016/0141-0229(94)00067-2
McFadzen IRB, Coombs SH, Halliday NC (1997) Histological indices of the nutritional condition of sardine, Sardina pilchardus (Walbaum) larvae of the north coast of Spain. J Exp Mar Biol Ecol 212:239–258
Mondal S, Roy T, Sen SK, Ray AK (2008) Distribution of enzyme-producing bacteria in the digestive tracts of some freshwater fish. Acta Ichthyol Piscat 38(1):1–8. https://doi.org/10.3750/AIP2008.38.1.01
Mukhopadhyay N, Ray AK (1999) Effect of fermentation on the nutritive value of sesame seed meal in the diets for rohu, Labeo rohita (Hamilton), fingerlings. Aquac Nutr 5:229–236. https://doi.org/10.1046/j.1365-2095.1999.00101.x
Nasser N, Babikian J, Hatem MG, Saoud IP, Abiad MG (2019) Evaluation of post-consumer food waste as partial replacement of commercial feed in marbled rabbitfish, Siganus rivulatus aquaculture. Int J Environ Sci Technol 16:4059–4068. https://doi.org/10.1007/s13762-018-2051-x
National Research Council (NRC) (2011) Nutrient requirements of fish and shrimp. National Academy Press, Washington DC
Peace RW, Gilani GS (2005) Chromatographic determination of amino acids in foods. J AOAC Int 88(3):877–887. https://doi.org/10.1093/jaoac/88.3.877
Rahimnejad S, Lu K, Wang L, Song K, Mai K, Davis DA, Zhang C (2019) Replacement of fishmeal with Bacillus pumillus SE5 and Pseudozyma aphidis ZR1 fermented soybean meal in diets for Japanese seabass (Lateolabrax japonicus). Fish Shellfish Immunol 84:987–997. https://doi.org/10.1016/j.fsi.2018.11.009
Ramírez-Ramírez JC, Huerta S, Arias L, Prado A, Shirai K (2008) Utilization of fisheries by-catch and processing wastes for lactic acid fermented silage and evaluation of degree of protein hydrolysis and in vitro digestibility. Rev Mex Ing Quim 7(3):195–204
Rangacharyulu PV, Giri SS, Paul BN, Yashoda KP, Rao RJ, Mahendrakar NS, Mohanty SN, Mukhopadhyay PK (2003) Utilization of fermented silkworm pupae silage in feed for carps. Bioresour Technol 86:29–32. https://doi.org/10.1016/s0960-8524(02)00113-x
Rao RJ, Mahendrakar NS, Chakrabarthy NM, Raghavan SL (1996) Utilisation of fermented fish and poultry silages in feed for common carp (Cyprinus carpio). Seafood Export J 27(3):17–23
Saha S, Roy RN, Sen SK, Ray AK (2006) Characterization of cellulase-producing bacteria from the digestive tract of tilapia, Oreochromis mossambicus (Peters) and grass carp, Ctenopharyngodon idella (Valenciennes). Aquac Res 37:380–388. https://doi.org/10.1111/j.1365-2109.2006.01442.x
Samaddara A, Kaviraja A, Saha A (2015) Utilization of fermented animal by-product blend as fishmeal replacer in the diet of Labeo rohita. Aquac Rep 1:28–36. https://doi.org/10.1016/j.aqrep.2015.03.004
Samtiya M, Aluko RE, Puniya AK, Dhewa T (2021) Enhancing micronutrients bioavailability through fermentation of plant-based foods: a concise review. Fermentation 7(2):63. https://doi.org/10.3390/fermentation7020063
Sinha AK, Kumar V, Makkar HP, De Boeck G, Becker K (2011) Non-starch polysaccharides and their role in fish nutrition–a review. Food Chem 127:1409–1426
Sudarmadji K, Markakis P (1977) The phytate and phytase of soyabean tempeh. J Sci Food Agric 28(4):381–383. https://doi.org/10.1002/jsfa.2740280410
Sutardi A, Buckle KA (1985) Reduction in phytic acid levels in soybean during tempeh production, storage and frying. Food Sci 50(1):260–263. https://doi.org/10.1111/j.1365-2621.1985.tb13325.x|
Teutino RA, Knorr D (1985) Impact of biotechnology on nutritional quality of food plants. Food Technol 39:127–134
Van Soest PJ, Robertson JB, Lewis BA (1991) Methods for dietary fiber, neutral detergent fiber and nonstarch polysaccharides in relation to animal nutrition. J Dairy Sci 74:3583–3597. https://doi.org/10.3168/jds.S0022-0302(91)78551-2
Vidotti RM, Carneiro DJ, Viegas EMM (2002) Growth rate of Pacu, Piaractus mesopotamicus, fingerlings fed diets containing co-dried fish silage as replacement of fish meal. J Appl Aquac 12(4):77–88
Wilson RP (2002) Amino acids and proteins. In: Halver JE, Hardy RW (eds) Fish Nutrition, 3rd edn. Academic Press, San Diego, California, pp 143–179
Winberg GG (1956) Rate of metabolism and food requirements of fishes. J Fish Res Board Can 194:1–202
Zeineddine R, Ireland B, Monzer S, Saoud IP (2021) Preliminary assessment of restaurant food waste as a feed ingredient for small juvenile rainbow trout Oncorhynchus mykiss). Aquac Int 29:669–679. https://doi.org/10.1007/s10499-021-00650-4
Acknowledgements
The authors would like express their heartfelt gratitude to Mr. Nell Wiid, a fish farmer who helped to collect the chicken carcasses and process it on his farm. We are also grateful to honours students at ARU who assisted during fish sampling and dissections. We are equally thankful to the anonymous reviewers whose criticism and guidance led to an improved final product.
Funding
The Department of Forestry, Fisheries and the Environment provided funds for this study.
Author information
Authors and Affiliations
Contributions
Chris Henri Fouche and Esau M. Mbokane conceived the study and wrote the manuscript. Esau M. Mbokane and Lucia Matlale Mbokane carried out the experiments, collected data, analyzed and interpreted it.
Corresponding author
Ethics declarations
Ethics approval
This study is part of a multi-year project focusing on the use of locally-available plant and animal waste resources in an effort to develop cheaper and high-quality diets for freshwater fish species in South Africa. Ethical clearance was granted by the University of Limpopo Animal Research Ethics Committee (AREC) of the Faculty of Science and Agriculture. Handling and sacrificing of fish was executed in a humane manner, and international, national and/or institutional guidelines governing the use of animals were observed at all times.
Competing interests
The authors declare no competing interests.
Additional information
Handling Editor: Gavin Burnell.
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Mbokane, E.M., Mbokane, L.M. & Fouche, C.H. The effect of fishmeal replacement with acid-fermented chicken silage on growth, digestive enzyme activity and histology of the intestine and liver of juvenile Mozambique tilapia (Oreochromis mossambicus). Aquacult Int 30, 2491–2512 (2022). https://doi.org/10.1007/s10499-022-00916-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10499-022-00916-5