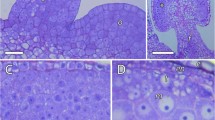
Abstract
Pollen grains have a relatively simple structure and microscopic size, with two or three cells surrounded by the protective sporoderm at maturity. The viability and efficiency of pollen transport from anther to stigma depends on pollen physiological properties, especially the relative water content of the vegetative cell. Pollen transport is a crucial fate for most angiosperms that depends on biotic pollinators and studies focusing on understanding the morpho-physiological properties of pollen grains are still scarce, especially to tropical open physiognomies as the Brazilian Cerrado. Therefore, we investigate some structural and physiological aspects of pollen grains from six native species naturally growing in one Cerrado area: Campomanesia pubescens (Myrtaceae), Caryocar brasiliense (Caryocaraceae), Erythroxylum campestre (Erythroxylaceae), Lippia lupulina (Verbenaceae), Pyrostegia venusta (Bignoniaceae), and Xylopia aromatica (Annonaceae). We selected dehiscent anthers and mature pollen grains to analyze (1) the anther wall and pollen microstructure, (2) the pollen water status at the time of anther dehiscence, and (3) the pollen chemical compounds. In all analyzed species, the anther and pollen developed in a successfully way, and except for Caryocar brasiliense, all species were able to emit pollen tubes in the germination tests. As expected for a dry and open environment, most species dispersed their pollen grains in a partially dehydrated form, as indicated by our harmomegathy experiment. As indicated by our study, the pollen ability in preventing dissection, maintaining its viability in a dry and hot environment during its transport from anther to stigma, may be related to the sporoderm apertures and to the reserve compounds, mainly carbohydrates in the form of hydrolysable starch grains.





Similar content being viewed by others
References
Albert B, Ressayre A, Nadot S (2011) Correlation between pollen aperture pattern and callose deposition in late tetrad stage in three species producing atypical pollen grains. Am J Bot 98:189–196. https://doi.org/10.3732/ajb.1000195
Ali SI (1988) The functional significance of pollen aggregates in angiosperms. Pak J Bot 20:21–44
Alonso C, Navarro-Fernández CM, Arceo-Gómez G, Meindl GA, Parra-Tabla V, Ashman T (2013) Among species differences in pollen quality and quantity limitation: implications for endemics in biodiverse hotspots. Ann Bot 112:1461–1469. https://doi.org/10.1093/aob/mct213
Alves-Ferreira M, Wellmer F, Banhara A, Kumar V, Riechmann JL, Meyerowitz EM (2007) Global expression profiling applied to the analysis of Arabidopsis stamen development. Plant Physiol 145:747–762. https://doi.org/10.1104/pp.107.104422
Araujo FD (1995) A review of Caryocar brasiliense (Caryocaraceae): an economically valuable species of the Central Brazilian Cerrados. Econ Bot 49:40–48. https://doi.org/10.1007/BF02862276
Atkins S (2004) Verbenaceae. In: Kadereit JW (ed) Flowering plants, vol. VII, Dicotyledons: Lamiales (except Acanthaceae including Avicenniaceae), 7th edn. Springer-Verlag, Berlin, pp 449–468
Baker HG, Baker I (1979) Starch in angiosperm pollen grains and its evolutionary significance. Am J Bot 66:591–600. https://doi.org/10.1002/j.1537-2197.1979.tb06262.x
Baker HG, Baker I (1982) Starchy and starchless pollen in the Onagraceae. Ann Mo Bot Gard 68:748–754. https://doi.org/10.2307/2398994
Baker HG, Baker I (1983) Some evolutionary and taxonomic implications of variation in the chemical reserves of pollen. In: Mulcahy DL, Ottaviano E (eds) Pollen: biology and implications in plant breeding. Elsevier Biomedical, New York, pp 42–51
Ballesteros-Mejia L, Lima JS, Collevatti RG (2018) Spatially-explicit analyses reveal the distribution of genetic diversity and plant conservation status in Cerrado biome. Biodivers Conserv 29:1537–1554. https://doi.org/10.1007/s10531-018-1588-9
Barros MG (1998) Sistemas reprodutivos e polinização em espécies simpátricas de Erythroxylum P. Br. (Erythroxylaceae) do Brasil. Rev Bras Bot 21:159–166. https://doi.org/10.1590/S0100-84041998000200008
Bawa KS, Kang H, Grayum MH (2003) Relationships among time, frequency, and duration of flowering in Tropical Rain Forest trees. Am J Bot 90:877–887. https://doi.org/10.3732/ajb.90.6.877
Bhandari NN (1984) The microsporangium. In: Johri BM (ed) Embryology of angiosperms. Springer, New York, pp 53–121
Bittrich V (2014) Erythroxylaceae. In: Kubitzki K (ed) Flowering plants Eudicots: Malpighiales, XI. Springer Verlag, Heidelberg, pp 43–49
BPBES/REBIPP (2019) Relatório temático sobre polinização, polinizadores e produção de alimentos no Brasil. Wolowski M et al (org). Editora Cubo, São Carlos-SP. https://doi.org/10.4322/978-85-60064-83-0
Brewbaker JL (1967) Distribution and phylogenetic significance of binucleate and trinucleate pollen grains in angiosperms. Am J Bot 54:1069–1083. https://doi.org/10.1002/j.1537-2197.1967.tb10735.x
Brewbaker JL, Kwack BH (1963) The essential role of calcium ion in pollen germination and pollen tube growth. Am J Bot 50:859–865. https://doi.org/10.1002/j.1537-2197.1963.tb06564.x
Buitink J, Walters-Vertucci C, Hoekstra FA, Leprince O (1996) Calorimetric properties of dehydrating pollen (analysis of a desiccation-tolerant and an intolerant species). Plant Physiol 111:235–242. https://doi.org/10.1104/pp.111.1.235
Cheynier V, Comte G, Davies KM, Lattanzio V, Martens S (2013) Plant phenolics: recent advances on their biosynthesis, genetics, and ecophysiology. Plant Physiol Biochem 72:1–20. https://doi.org/10.1016/j.plaphy.2013.05.009
Cortez PA, Caetano APS, Carmello-Guerreiro SM, Teixeira SP (2014) Elucidating the mechanism of poricidal anther dehiscence in Miconia species (Melastomataceae). Flora 209:571–579. https://doi.org/10.1016/j.flora.2014.07.002
Cortez PA, Caetano APS, Carmello-Guerreiro SM, Teixeira SP (2015) Anther wall and pollen development in Neotropical species-rich Miconia (Melastomataceae). Plant Syst Evol 301:217–230. https://doi.org/10.1007/s00606-014-1067-z
Cruden RW (2000) Pollen grains: why so many? Plant Syst Evol 222:143–165. https://doi.org/10.1007/BF00984100
Dafni A, Firmage D (2000) Pollen viability and longevity: practical, ecological and evolutionary implications. Plant Syst Evol 222:113–132. https://doi.org/10.1007/BF00984098
Davis GL (1966) Systematic embryology of the angiosperms. Wiley , New York
Dobson HEM (1988) Survey of pollen and pollenkitt lipids: chemical cues to flower visitors? Am J Bot 75:170–182. https://doi.org/10.1002/j.1537-2197.1988.tb13429.x
Doyle JA (2005) Early evolution of angiosperm pollen as inferred from molecular and morphological phylogenetic analyses. Grana 44:227–251. https://doi.org/10.1080/00173130500424557
Doyle JA, Thomas AL (2012) Evolution and phylogenetic significance of pollen in Annonaceae. Bot J Linn Soc 169:190–221. https://doi.org/10.1111/j.1095-8339.2012.01241.x
Edlund AF, Zheng Q, Lowe N, Kuseryk S, Ainsworth KL, Lyles RH, Sibener SJ, Preuss D (2016) Pollen from Arabidopsis thaliana and other Brassicaceae are functionally omniaperturate. Am J Bot 103:1–14. https://doi.org/10.3732/ajb.1600031
Eiten G (1972) The Cerrado vegetation of Brazil. Bot Rev 38:201–341. https://doi.org/10.1007/BF02859158
Endress PK (1996) Diversity and evolutionary biology of tropical flowers. University Press, Cambridge
Engel EC, Irwin RE (2003) Linking pollinator visitation rate and pollen receipt. Am J Bot 90:1612–1618. https://doi.org/10.3732/ajb.90.11.1612
Erdtman G (1943) An introduction to pollen analysis. Chronica Botanica Company, Waltham
Erdtman G (1952) Pollen morphology and plant taxonomy - angiosperms. Almqvist and Wiksell, Stockholm
Eschrich W, Currier HB (1964) Identification of calose by its diachrome and fluorochrome reactions. Stain Technol 39:303–307. https://doi.org/10.3109/10520296409061248
Faegri K, Iversen J (1964) Textbook of pollen analysis. Munksgaard, Copenhagen
Faegri K, van der Pijl L (1979) The principles of pollination ecology. Pergamon Press, Oxford
Firon N, Nepi M, Pacini E (2012) Water status and associated processes mark critical stages in pollen development and functioning. Ann Bot 109:1201–1213. https://doi.org/10.1093/aob/mcs070
Fisher DB (1968) Protein staining of ribboned Epon sections for light microscopy. Histochemie 16:92–96. https://doi.org/10.1007/BF00306214
Flora do Brasil 2020 em construção. Jardim Botânico do Rio de Janeiro. http://floradobrasil.jbrj.gov.br/. Accessed 15 October 2020
Forrest JRK (2015) Plant-pollinator interactions and phenological change: what can we learn about climate impacts from experiments and observations? Oikos 124:4–13. https://doi.org/10.1111/oik.01386
Franchi GG, Bellani L, Nepi M, Pacini E (1996) Types of carbohydrate reserves in pollen: localization, systematic distribution and ecophysiological significance. Flora 191:143–159. https://doi.org/10.1016/S0367-2530(17)30706-5
Franchi GG, Nepi M, Dafni A, Pacini E (2002) Partially hydrated pollen: taxonomic distribution, ecological and evolutionary significance. Plant Syst Evol 234:211–227. https://doi.org/10.1007/s00606-002-0221-1
Franchi GG, Piotto B, Nepi M, Baskin CC, Baskin JM, Pacini E (2011) Pollen and seed desiccation tolerance in relation to degree of developmental arrest, dispersal, and survival. J Exp Bot 62:5267–5281. https://doi.org/10.1093/jxb/err154
Franchi GG, Nepi M, Pacini E (2014) Is flower/corolla closure linked to decrease in viability of desiccation-sensitive pollen? Facts and hypotheses: a review of current literature with the support of some new experimental data. Plant Syst Evol 300:577–584. https://doi.org/10.1007/s00606-013-0911-x
Furness CA, Rudall PJ (2004) Pollen aperture evolution - a crucial factor for eudicot success? Trends Plant Sci 9:154–158. https://doi.org/10.1016/j.tplants.2004.01.001
Gobatto-Rodrigues AA, Stort MNS (1992) Biologia floral e reprodução de Pyrostegia venusta (Ker-Gawl) Miers (Bignoniaceae). Rev Bras Bot 15:37–41. https://doi.org/10.5216/rbn.v4i1.4656
Gottsberger G (1970) Beiträge zur Biologie von Annonaceen-Blüten. Österr Bot Zeitschrift 118:237–279. https://doi.org/10.1007/BF01377861
Gottsberger G (1999) Pollination and evolution in Neotropical Annonaceae. Plant Spec Biol 14:143–152. https://doi.org/10.1046/j.14421984,1999(00018),pp.x
Gottsberger G, Silberbauer-Gottsberger I (2018) How are pollination and seed dispersal modes in Cerrado related to stratification? Trends in a cerrado sensu stricto woodland in southeastern Brazil, and a comparison with Neotropical forests. Acta Bot Bras 32:434–445. https://doi.org/10.1590/0102-33062018abb0186
Gressler E, Pizo MA, Morellato PC (2006) Polinização e dispersão de sementes em Myrtaceae do Brasil. Rev Brasil Bot 29:509–530. https://doi.org/10.1590/S0100-84042006000400002
Gribel R, Hay JD (1993) Pollination ecology of Caryocar brasiliense (Caryocaraceae) in Central Brazil cerrado vegetation. J Trop Ecol 9:199–211. https://doi.org/10.1017/S0266467400007173
Guarnieri M, Speranza A, Nepi M, Artese D, Pacini E (2006) Ripe pollen carbohydrate changes in Trachycarpus fortunei: the effect of relative humidity. Sex Plant Reprod 19:117–124. https://doi.org/10.1007/s00497-006-0027-3
Gusman AB, Gottsberger G (1996) Differences in floral morphology, floral nectar constituents, carotenoids, and flavonoids in petals of orange and yellow Pyrotegia venusta (Bignoniaceae) flowers. Phyton 36:161–171
Halbritter H, Ulrich S, Grímsson F, Weber M, Zetter R, Hesse M, Buchner R, Svojtka M, Frosch-Radivo A (2018) Illustrated pollen terminology. Springer International Publishing, New York
Harder LD, Johnson SD (2008) Function and evolution of aggregated pollen in angiosperms. Int J Plant Sci 169:59–78. https://doi.org/10.1086/523364
Hedhly A, Hormaza JI, Herrero M (2008) Global warming and sexual plant reproduction. Trends Plant Sci 14:30–36. https://doi.org/10.1016/j.tplants.2008.11.001
Hegland SJ, Nielsen A, Lázaro A, Bjerknes A, Totland Ø (2009) How does climate warming affect plant-pollinator interactions? Ecol Lett 12:184–195. https://doi.org/10.1111/j.1461-0248.2008.01269.x
Heithaus ER (1974) The role of plant-pollinator interactions in determining community structure. Ann Mo Bot Gard 61:675–691. https://doi.org/10.2307/2395023
Heslop-Harrison Y (1979) An interpretation of the hydrodynamics of pollen. Am J Bot 66:737–743. https://www.jstor.org/stable/2442418
Hesse M (2000) Pollen wall stratification and pollination. Plant Syst Evol 222:1–17. https://doi.org/10.1007/BF00984093
Hoekstra FA, Golovina EA, Buitink J (2001) Mechanisms of plant desiccation tolerance. Trends in Plant Sci 6:431–438. https://doi.org/10.1016/S1360-1385(01)02052-0
Johansen DA (1940) Plant microtechnique. McGraw-Hill Book Company, New York
Johri BM, Ambegaokar KB, Srivastava PS (1992) Comparative embryology of angiosperms. Springer-Verlag, Berlin
Karnovsky MJ (1965) A formaldehyde-glutaraldehyde fixative of high osmolarity for use in electron microscopy. J Cell Biol 27:137A-138A
Katifori E, Alben S, Cerda E, Nelson DR, Dumais J (2010) Foldable structures and the natural design of pollen grains. PNAS 107:7635–7639. https://doi.org/10.1073/pnas.0911223107
Keijzer CJ, Hoek IHS, Willemse MTM (1987) The processes of anther dehiscence and pollen dispersal III. The dehydration of the filament tip and the anther in three monocotyledonous species. New Phytol 106:281–287. https://doi.org/10.1111/j.1469-8137.1987.tb00143.x
Kenda G (1952) Stomata an antheren. Phyton 4:83–96.
Kessler PJA (1993) Annonaceae. In: Kubitzki K, Rohwer G, Bittrich V (eds) Flowering plants Dicotyledons: Magnoliid II Hamamelid and Caryophyllid families. Springer-Verlag, Berlin, pp 93–129
Koski MH, MacQueen D, Ashman T (2020) Floral pigmentation has responded rapidly to Global Change in ozone and temperature. Curr Biol 30:1–7. https://doi.org/10.1016/j.cub.2020.08.077
Kriebel R, Khabbazian M, Sytsma KJ (2017) A continuous morphological approach to study the evolution of pollen in a phylogenetic context: an example with the order Myrtales. PLoS One 12:e0187228. https://doi.org/10.1371/journal.pone.0187228
Landrum LR (1986) Campomanesia, Pimenta, Blepharocalyx, Legrandia, Acca, Myrrhinium, and Luma (Myrtaceae). Flora Neotropica 45:1–178. http://www.jstor.org/stable/4393795
Landrum LR, Kawasaki ML (1997) The genera of Myrtaceae in Brazil: an illustrated synoptic treatment and identification keys. Brittonia 49:508–536. https://doi.org/10.2307/2807742
Maheshwari P (1950) An introduction to the embryology of angiosperms. McGrawHill Book Co, New York, pp 28–53, 154–180
Martin FW (1959) Staining and observing pollen tubes in the style by means of fluorescence. Stain Technol 34:125–128. https://doi.org/10.3109/10520295909114663
Martins AE, Camargo MGG, Morellato LPC (2021) Flowering phenology and the influence of seasonality in flower conspicuousness for bees. Front Plant Sci 11:594538. https://doi.org/10.3389/fpls.2020.594538
Matamoro-Vidal A, Raquin C, Brisset F, Colas E, Izac B, Albert B, Gouyon P (2016) Links between morphology and function of the pollen wall: an experimental approach. Bot J Linn Soc 180:478–490. https://doi.org/10.1111/boj.12378
McManus JFA (1948) Histological and histochemical uses of periodic acid. Stain Technol 23:99–108. https://doi.org/10.3109/10520294809106232
Merkel A. Dados climáticos para cidades mundiais. https://pt.climate-data.org/america-do-sul/brasil/sao-paulo/itirapina-34775/#climate-graph. Accessed 16 December 2020
Mittermeier RA, Robles-Gil P, Hoffman M, Pilgrim J, Brooks T, Mittermeier CG, Lamoreux J, Fonseca GAB (2005) Hotspots revisited: earth's biologically richest and most endangered terrestrial ecoregions. CEMEX, Mexico City
Moldenke HN (1965) Materials toward a monograph of the genus Lippia. Phytologia 12:265–271. https://www.biodiversitylibrary.org/item/50236
Mostafa NM, El-Dahshan O, Singab ANB (2013) Pyrostegia venusta (Ker Gawl.) Miers: a botanical, pharmacological and phytochemical review. Med Aromat Plants 2:1–6. https://doi.org/10.4172/2167-0412.1000123
Muller J (1979) Form and function in angiosperm pollen. Ann Mo Bot Gard 66:593–632. https://doi.org/10.2307/2398913
Nanda K, Gupta SC (1971) Endothecium in Pyrostegia venusta. Curr Sci 40:470–471
Nepi M, Franchi GG (2000) Cytochemistry of mature angiosperm pollen. Plant Syst Evol 222:45–62. https://doi.org/10.1007/978-3-7091-6306-1_3
Nepi M, Franchi GG, Pacini E (2001) Pollen hydration status at dispersal: cytophysiological features and strategies. Protoplasma 216:171–180. https://doi.org/10.1007/BF02673869
O’Brien TP, Feder N, McCully ME (1964) Polyehromatic staining of plant cell walls by Toluidine Blue O. Protoplasma 59:368–373. https://doi.org/10.1007/BF01248568
Oliveira PE, Gibbs PE (2000) Reproductive biology of woody plants in a Cerrado community of Central Brazil. Flora 195:311–329. https://doi.org/10.1016/S0367-2530(17)30990-8
Oliveira PS, Marquis RJ (2002) The Cerrados of Brazil: ecology and natural history of a Neotropical Savanna. Columbia University Press, New York
Ollerton J, Winfree R, Tarrant S (2011) How many flowering plants are pollinated by animals? Oikos 120:321–326. https://doi.org/10.1111/j.1600-0706.2010.18644.x
Pacini E (2000) From anther and pollen ripening to pollen presentation. Plant Syst Evol 222:19–43. https://doi.org/10.1007/BF00984094
Pacini E, Hesse M (2004) Cytophysiology of pollen presentation and dispersal. Flora 199:273–285. https://doi.org/10.1078/0367-2530-00156
Pacini E, Hesse M (2005) Pollenkitt - its composition, forms and functions. Flora 200:399–415. https://doi.org/10.1016/j.flora.2005.02.006
Pacini E, Dolferus R (2016) The trials and tribulations of the plant male gametophyte - understanding reproductive stage stress tolerance. In: Shanker AK, Shanker C (eds) Abiotic and biotic stress in plants - recent advances and future perspectives. InTechOpen, London, pp 703–754
Pacini E, Franchi GG (2020) Pollen biodiversity - why are pollen grains different despite having the same function? A review. Bot J Linn Soc 193:141–164. https://doi.org/10.1093/botlinnean/boaa014
Pacini E, Guarnieri M, Nepi M (2006) Pollen carbohydrates and water content during development, presentation, and dispersal: a short review. Protoplasma 228:73–77. https://doi.org/10.1007/s00709-006-0169-z
Payne WW (1972) Observations of harmomegathy in pollen of Anthophyta. Grana 12:93–98. https://doi.org/10.1080/00173137209428832
Payne WW (1981) Structure and function in angiosperm pollen wall evolution. Rev Palaeobot Palyno 35:39–59. https://doi.org/10.1016/0034-6667(81)90013-0
Pearse AGE (1985) Histochemistry. Theoretical and applied. Churchill Livingstone, Edinburgh
Piffanelli P, Ross J, Murphy D (1998) Biogenesis and function of the lipidic structures of pollen grains. Sex Plant Reprod 11:65–80. https://doi.org/10.1007/s004970050122
Polatto LP, Dutra JCS, Alves-Junior VV (2007) Biologia reprodutiva de Pyrostegia venusta (Ker-Gawl) Miers (Bignoniaceae) e comportamento de forrageamento dos visitantes florais predominantes. Rev Biol Neotrop 4:46–57. https://doi.org/10.5216/rbn.v4i1.4656
Pool A (2008) A review of the genus Pyrostegia (Bignoniaceae). Ann Mo Bot Gard 95:495–510. https://doi.org/10.3417/2003090
Prance GT (2014) Caryocaraceae. In: Kubitzki K (ed) Flowering plants Eudicots: Malpighiales, XI. Springer Verlag, Heidelberg, pp 13–16
Prance GT, Silva MF (1973) Monograph of Caryocaraceae. Flora. Neotropica 12:1–75
Proença CEB, Gibbs PE (1994) Reproductive biology of eight sympatric Myrtaceae from Central Brazil. New Phytol 126:343–354. https://doi.org/10.1111/j.1469-8137.1994.tb03954.x
Punt W, Hoen PP, Blackmore S, Nilsson S, Le Thomas A (2007) Glossary of pollen and spore terminology. 143:1-81. https://doi.org/10.1016/j.revpalbo.2006.06.008
Ribeiro VC, Leitão CAE (2020) Utilisation of Toluidine blue O pH 4.0 and histochemical inferences in plant sections obtained by free-hand. Protoplasma 257:993–1008. https://doi.org/10.1007/s00709-019-01473-0
Rossatto DR, Kolb RM (2011) Comportamento fenológico da liana Pyrostegia venusta (Ker Gawl.) Miers (Bignoniaceae) em área de cerradão na Estação Ecológica de Assis, SP Brasil. R Bras Bioci 9:289–296
Roulston TH, Buchmann SL (2000) A phylogenetic reconsideration of the pollen starch-pollination correlation. Evol Ecol Res 2:627–643
Roulston TH, Cane JH (2000) Pollen nutritional content and digestibility for animals. Plant Syst Evol 222:187–209. https://doi.org/10.1007/BF00984102
Roulston TH, Cane JH, Buchmann SL (2000) What governs protein content of pollen: pollinator preferences, pollen pistil interactions, or phylogeny? Ecol Monogr 70:617–643. https://doi.org/10.2307/2657188
Salimena FRG, França F, Silva TRS (2009) Verbenaceae In: Giulietti AM et al. (org) Plantas raras do Brasil. Conservação Internacional, Belo Horizonte, pp 399–405
Salimena FRG, Kutschenko DC, Monteiro NP, Mynssen C (2013) Verbenaceae In: Martinelli G, Moraes MA (org) Livro vermelho da flora do Brasil. Instituto de Pesquisas Jardim Botânico, Rio de Janeiro, pp 1010-1016
Sampaio ES, Almeida AA (1995) Morfologia floral e biologia reprodutiva de Pyrostegia venusta (Bignoniaceae) na região urbana de Curitiba, Paraná. Acta Biol Par 24:25–38. https://doi.org/10.5380/abpr.v24i0.701
Schmid R (1976) Filament histology and anther dehiscence. Bot J Linn Soc 73:303–315. https://doi.org/10.1111/j.1095-8339.1976.tb01811.x
Schweizer D, Ambros PF (1994) Chromosome banding. In: Gosden JR (ed) Chromosome analysis protocols. Methods in Molecular Biology, vol 29. Humana Press, New Jersey. https://doi.org/10.1385/0-89603-289-2:97
Shivanna KR, Tandon R (2020) Developmental biology of dispersed pollen grains. Int J Dev Biol 64:7–19. https://doi.org/10.1387/ijdb.190166ks
Silva L, Fernandes CL, Simão E, Nakamura AT (2016) Development of anthers and pollen in Brazilian heterostylic species of Erythroxylum (Erythroxylaceae): an ecological approach. Pak J Bot 48:2047–2055
Singh S, Chauhan S, Galetto L (2009) Reproductive biology of Pyrostegia venusta (Ker-Gawl.) Miers (Bignoniaceae) with special reference to seedlessness. J Plant Reprod Biol 1:87–92
Speranza A, Calzoni GL, Pacini E (1997) Occurrence of mono- or disaccharides and polysaccharide reserves in mature pollen grains. Sex Plant Reprod 10:110–115. https://doi.org/10.1007/s004970050076
Stroo A (2000) Pollen morphological evolution in bat pollinated plants. In: Dafni A, Hesse M, Pacini E (ed) Pollen and Pollination. Springer, Vienna. https://doi.org/10.1007/978-3-7091-6306-1_12
Swain T (1977) Secondary compounds as protective agents. Ann Rev Plant Physiol 28:479–501. https://doi.org/10.1146/annurev.pp.28.060177.002403
Tanaka N, Uehara K, Murata J (2004) Correlation between pollen morphology and pollination mechanisms in the Hydrocharitaceae. J Plant Res 117:265–276. https://doi.org/10.1007/s10265-004-0155-5
Vieira AM, Feijó JA (2016) Hydrogel control of water uptake by pectins during in vitro pollen hydration of Eucalyptus globulus. Am J Bot 103:437–451. https://doi.org/10.3732/ajb.1500373
Walker JW (1971) Pollen morphology, phytogeography, and phylogeny of the Annonaceae. Contr Gray Herb 202:1–132
Walker JW (1972) Contributions to the pollen morphology and phylogeny of the Annonaceae. II Bot J Linn Soc 65:173–178. https://doi.org/10.1111/j.1095-8339.1972.tb00931.x
Wei D, Liu M, Chen H, Zheng Y, Liu Y, Wang X, Yang S, Zhou M, Lin J (2018). Inducer of CBF expression 1 is a male fertility regulator impacting anther dehydration in Arabidopsis. PLoS Genet 14:e1007695. https://doi.org/10.1371/journal.pgen.1007695
Willmer P (2011) Pollination and floral ecology. University Press, Princeton
Wodehouse RP (1928) The phylogenetic value of pollen grain characters. Ann Bot 42:891–934. https://doi.org/10.1093/oxfordjournals.aob.a090149
Wodehouse RP (1935) Pollen grains. McGraw-Hill Book Company, New York
Wortley AH, Wang H, Lu L, Li D, Blackmore S (2015) Evolution of angiosperm pollen. 1 Introduction. Ann Mo Bot Gard 100:177–226. https://doi.org/10.3417/2012047
Zanchetta D (2006) Plano de manejo integrado das unidades de Itirapina. Secretaria do Meio Ambiente, Instituto Florestal, São Paulo
Zhang D, Yang L (2014) Specification of tapetum and microsporocyte cells within the anther. Curr Opin Plant Biol 17:49–55. https://doi.org/10.1016/j.pbi.2013.11.001
Acknowledgements
We thank all the undergraduate students of the course Reproductive Structures of Angiosperms: Anatomy and Development (Biological Sciences, Instituto de Biociências, Universidade de São Paulo, São Paulo, Brazil, 2018) for their help with field observations and plant collections.
Author information
Authors and Affiliations
Corresponding author
Additional information
Handling Editor: Benedikt Kos
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Cortez, P.A., dos Santos Silva, L.N.N., de Ornellas Paschoalini, G. et al. Light and electron microscopies reveal unknown details of the pollen grain structure and physiology from Brazilian Cerrado species. Protoplasma 259, 399–412 (2022). https://doi.org/10.1007/s00709-021-01671-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00709-021-01671-9