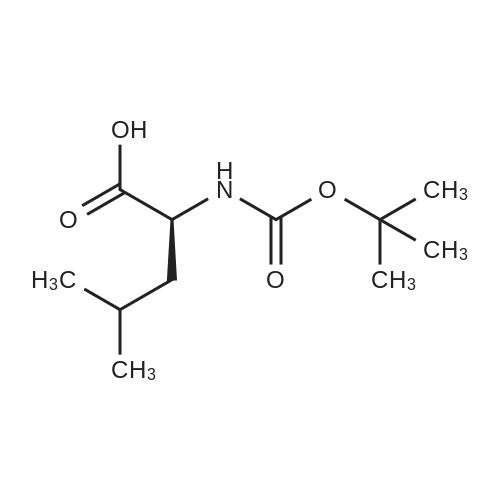
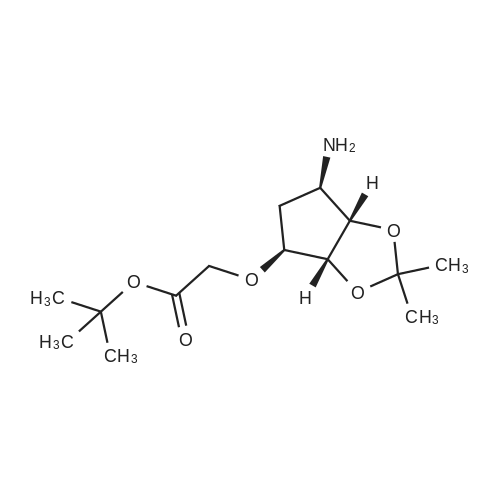
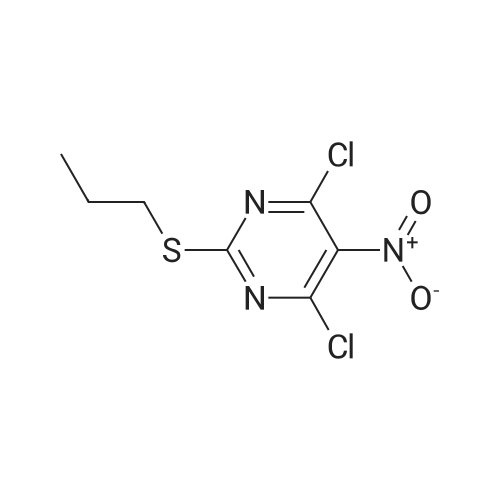
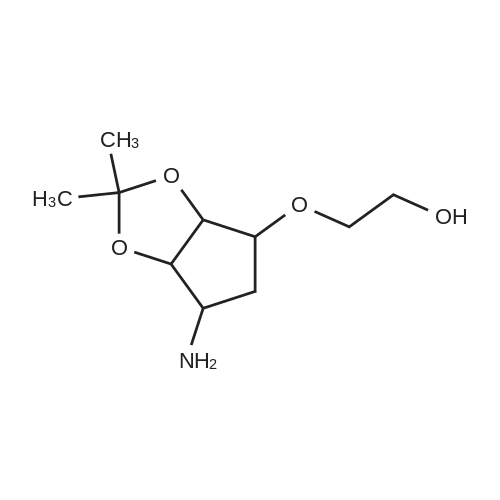
| 91% |
With N-ethyl-N,N-diisopropylamine In 1,4-dioxane at 100℃; for 16h; |
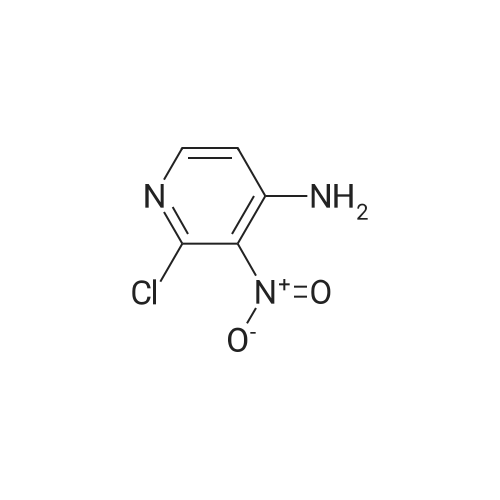
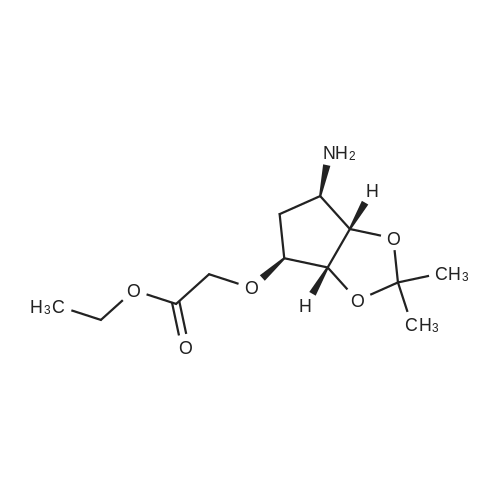
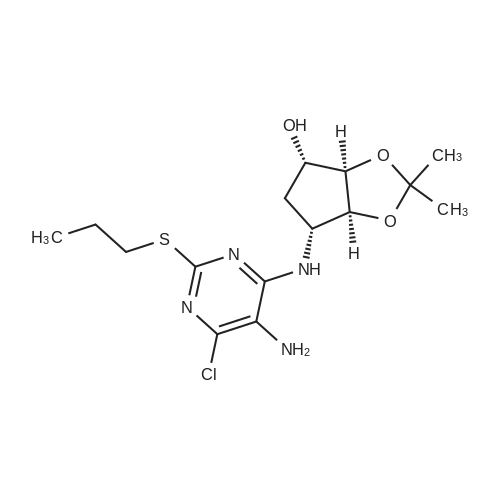
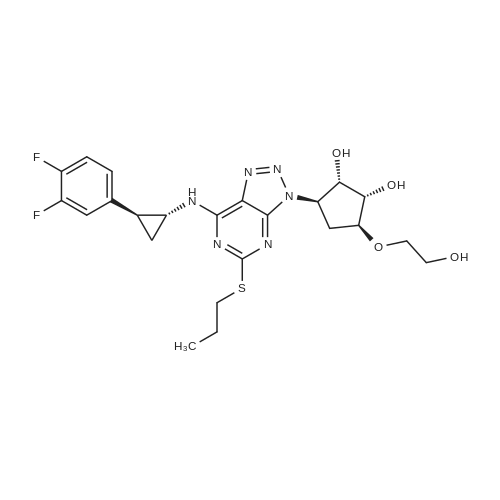
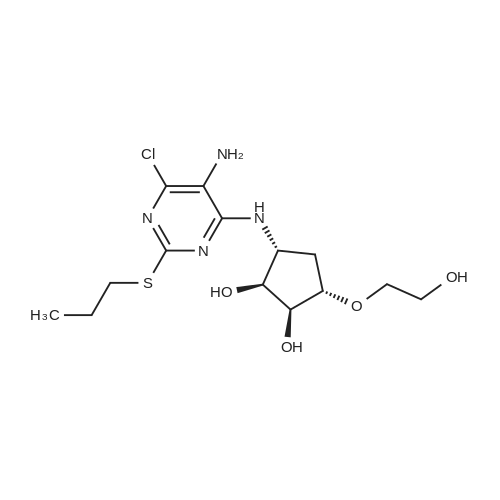
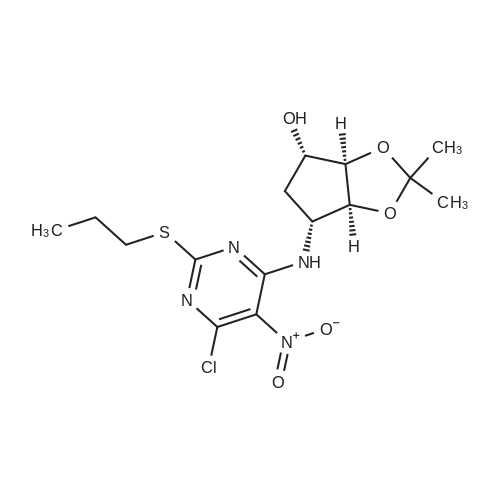
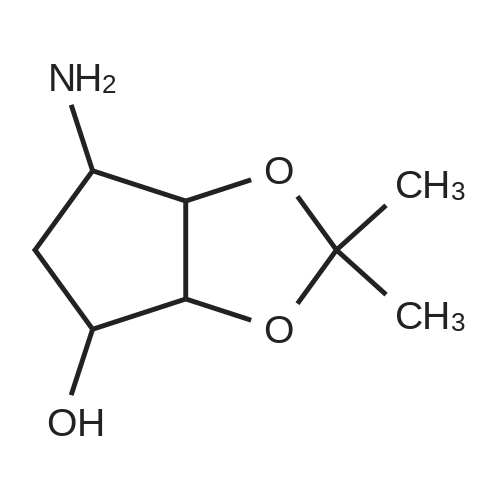
(3aR,4S,6R,6aS)-6-(5-amino-6-chloro-2-(propylthio)pyrimidin-4-ylamino)-2,2-dimethyltetrahydro-3aH-cyclopenta[d][1,3]dioxol-4-ol (4)
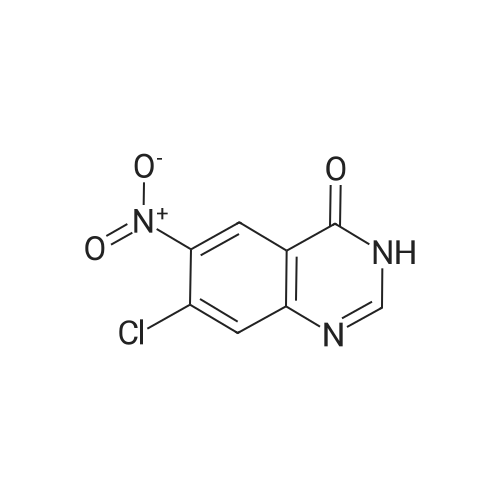
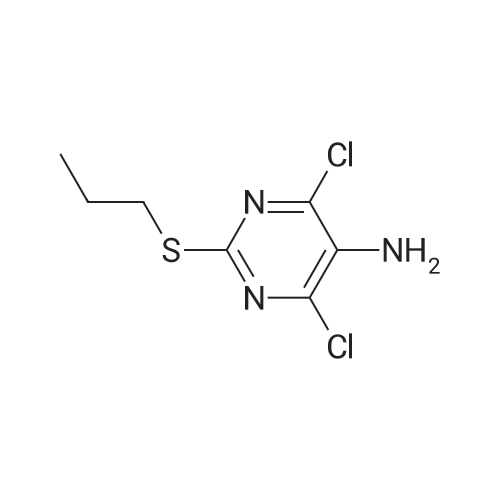
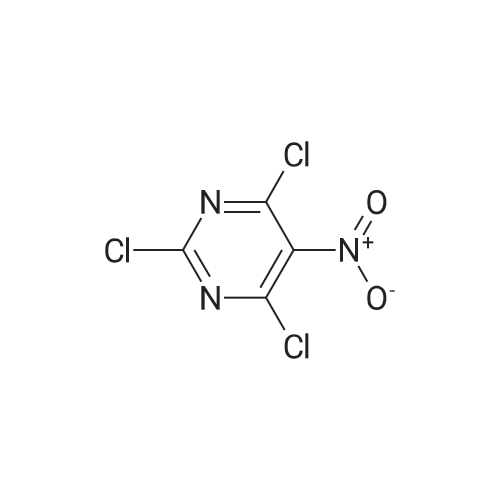
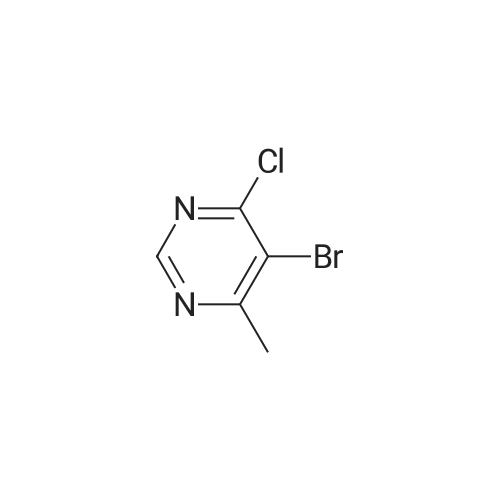
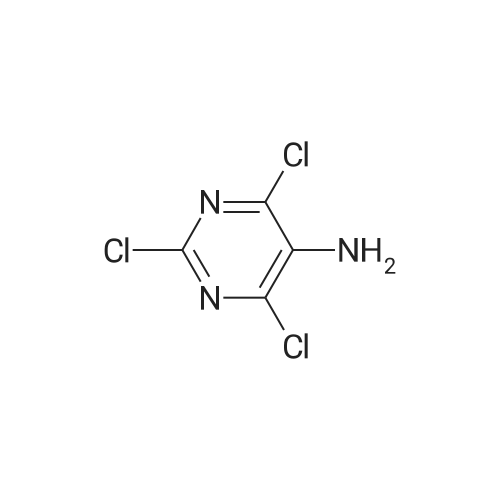
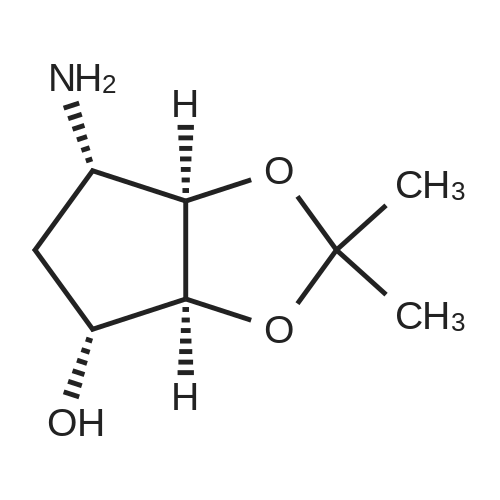
To a stirred solution of pyrimido dichloride 3 (16.10 g, 67.67 mmol) in 1,4-dioxane (300 mL), amino alcohol mandelic acid salts 2 (20.03 g, 61.47 mmol) and N,N-diisopropyl-N-ethylamine (23.83 g, 184.40 mmol) were added in one portion, respectively, while the colour of the reaction mixture changed to brown. The reaction mixture was stirred at 100°C for 16 h, and then water (300 mL) was added to the reaction mixture, then the aqueous phase was separated and extracted twice with ethyl acetate. The combined organic phases were washed with brine and dried over MgSO4. The organic solvent was removed under reduced pressure and residue was purified by silica gel column chromatography (ethyl acetate/hexane 1:5) to give brown oil 4 ( 20.97 g, yield 91%). |
| 90% |
In methanol at 100℃; for 12h; Sealed tube; |
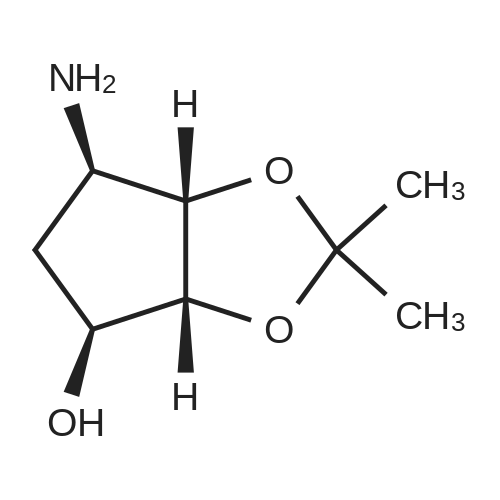
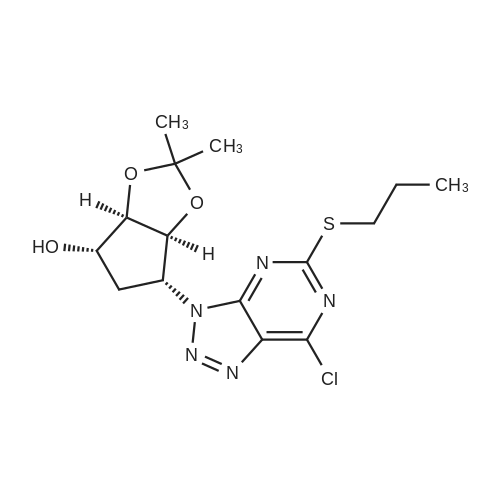
19 (3a/?,45,6/?,6a5)-6-((5-Amino-6-chloro-2-(propylthio)pyrimidin-4-yl)amino)-2,2- dimethyltetrahydro-3a/-/-cyclopenta[ /][l,3]dioxol-4-ol (19a)
4,6-Dichloro-2-(propylthio)pyrimidin-5-amine (0.5 g, 2.1 mmol) was dissolved in methanol (2 mL) and supplemented with (3a/?,45,6/?,6a5)-6-amino-2,2-dimethyltetrahydro-3a/-/- cyclopenta[ /][l,3]dioxol-4-ol (1.1 g, 6.3 mmol). The reaction mixture was introduced in a sealed vessel and heated at 100°C for 12 h. After concentration of the reaction mixture to dryness under vacuum, the residue was purified by silica gel column chromatography.Yield: 90%.Melting point: ND.H NMR (DMSO -d6) d 0.96 (t, J=7.4 Hz, 3H, SCH2CH2CH3), 1.21 (s, 3H, C(CH3)2), 1.36 (s, 3H,C(CH3)2), 1.64 (h, J=7.4 Hz, 2H, SCH2CH2CH3), 1.71 (m, 1H, S'-H), 2.22 (m, 1H, S'-H), 2.98 (t, J=7.2 Hz, 2H, SCH2CH2CH3), 4.06 (bs, 1H, 4’-H), 4.26 (bs, 1H, S'-H), 4.41 (d, J=5.9 Hz, 1H, 3a’-H), 4.51 (d, J=6.0 Hz, 1H, Sa'-H), 4.70 (s, 2H, NH2), 5.27 (d, J=3.1 Hz, 1H, OH), 6.63 (d, J=7.1 Hz, 1H, NH). 13C NMR (DMSO -d6) d 13.3 (SCH2CH2CH3), 22.9 (SCH2CH2CH3), 24.1 (C(CH3)2), 26.5 (C(CH3)2), 32.1 (SCH2CH2CH3), 35.9 (C-5'), 57.2 (C-6'), 75.3 (C-4'), 84.5 (C-6a'), 85.7 (3a'), 109.7 (C(CH3)2), 119.7 (C-5), 136.6 (C-6), 152.4 (C-4), 155.9 (C-2). |
| 90% |
In methanol at 100℃; for 12h; Sealed tube; |
19 (3a/?,4S,6/?,6aS)-6-((5-Amino-6-chloro-2-(propylthio)pyrimidin-4-yl)amino)-2,2- dimethyltetrahydro-3a/-/-cyclopenta [c/] [l,3]dioxol-4-ol (19a)
4,6-Dichloro-2-(propylthio)pyrimidin-5-amine (0.5 g, 2.1 mmol) was dissolved in methanol (2 mL) and supplemented with (3a/?,4S,6/?,6aS)-6-amino-2,2-dimethyltetrahydro-3a/-/- cyclopenta [c/] [l,3]dioxol-4-ol (1.1 g, 6.3 mmol). The reaction mixture was introduced in a sealed vessel and heated at 100°C for 12 h. After concentration of the reaction mixture to dryness under vacuum, the residue was purified by silica gel column chromatography. (0708) Yield: 90%. (0709) Melting point: ND. (0710) XH NMR (DMSO -d6) d 0.96 (t, J=7.4 Hz, 3H, SCH2CH2CH3), 1.21 (s, 3H, C(CH3)2), 1.36 (s, 3H, C(CH3)2), 1.64 (h, J=7.4 Hz, 2H, SCH2CH2CH3), 1.71 (m, 1H, S’-H), 2.22 (m, 1H, S’-H), 2.98 (t, J=7.2 Hz, 2H, SCH2CH2CH3), 4.06 (bs, 1H, A’-H), 4.26 (bs, 1H, 6’-H), 4.41 (d, J=5.9 Hz, 1H, 3a'-H), 4.51 (d, J=6.0 Hz, 1H, 6a’-H), 4.70 (s, 2H, NH2), 5.27 (d, J=3.1 Hz, 1H, OH), 6.63 (d, J=7.1 Hz, 1H, NH). 13C N MR (DMSO -d6) d 13.3 (SCH2CH2CH3), 22.9 (SCH2CH2CH3), 24.1 (C(CH3)2), 26.5 (C(CH3)2), 32.1 (0711) (SCH2CH2CH3), 35.9 (C-5'), 57.2 (C-6'), 75.3 (C-4'), 84.5 (C-6a'), 85.7 (3a'), 109.7 (C(CH3)2), 119.7 (C-5), 136.6 (C-6), 152.4 (C-4), 155.9 (C-2). |

 Chemistry
Chemistry
 Pharmaceutical Intermediates
Pharmaceutical Intermediates
 Inhibitors/Agonists
Inhibitors/Agonists
 Material Science
Material Science














 For Research Only
For Research Only
 110K+ Compounds
110K+ Compounds
 Competitive Price
Competitive Price
 1-2 Day Shipping
1-2 Day Shipping